Properties of Covalent Molecular Substances Properties Depend on

Properties of Covalent (Molecular) Substances

Properties • Depend on strength of forces between “particles” or separate units (Intermolecular Forces) • In covalent substances: – the units are molecules

Intermolecular Forces • Dispersion forces occur between nonpolar molecules. (Van der Waals) • Dipole-dipole forces occur between polar molecules. • Hydrogen bonding occurs between molecules with an H-F, H-O, or H-N bond. Intermolecular forces determine phase!

Weakest intermolecular forces = dispersion - occur between nonpolar molecules ● Monatomic molecules: He, Ne, Ar, Kr, etc. Diatomics with 2 atoms of the same element: O 2, H 2, N 2 ● ● Very symmetric molecules: C 4 H 10 Dispersion forces increase as the size of the molecule increases

Dipole-dipole forces occur between: - molecules with a permanent charge separation (polar molecules) HCl and HBr are examples of polar diatomic molecules
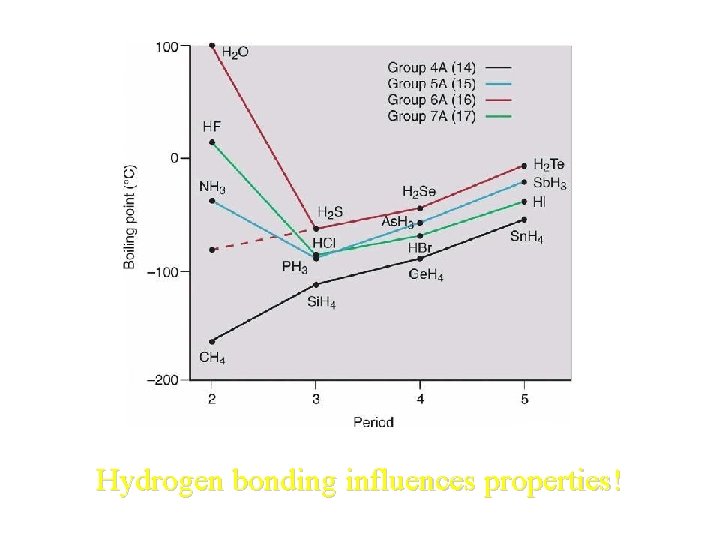
Hydrogen bonding influences properties!
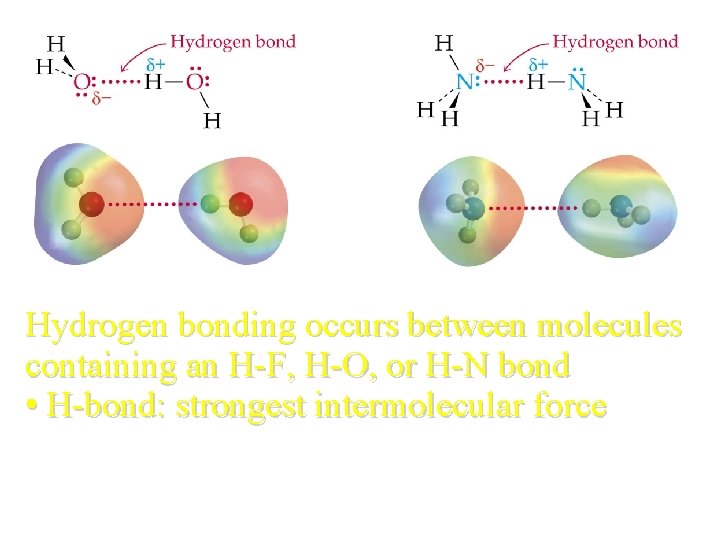
Hydrogen bonding occurs between molecules containing an H-F, H-O, or H-N bond • H-bond: strongest intermolecular force

Properties of Covalent (Molecular) Substances • Poor conductors of heat & electricity in any phase – No charged particles! • Low melting & boiling points – easy to pull molecules apart from each other • Majority of solids are soft • Low Hf and Hv compared to ionic & metallic substances • High vapor pressure compared to ionic & metallic substances
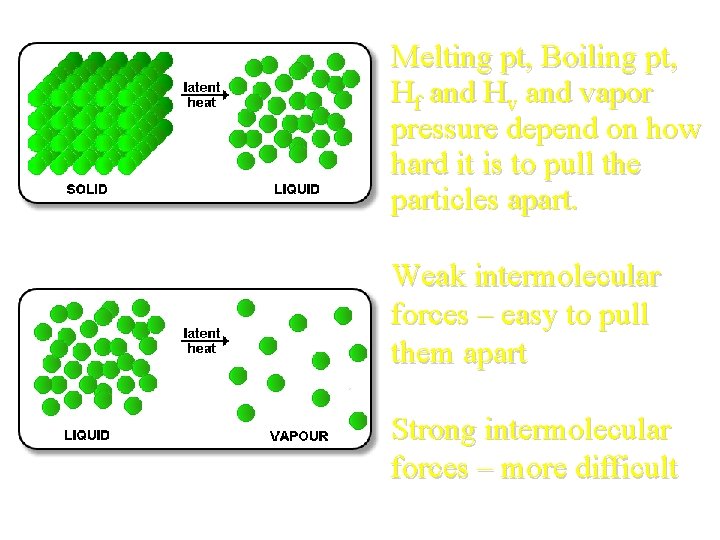
Melting pt, Boiling pt, Hf and Hv and vapor pressure depend on how hard it is to pull the particles apart. Weak intermolecular forces – easy to pull them apart Strong intermolecular forces – more difficult
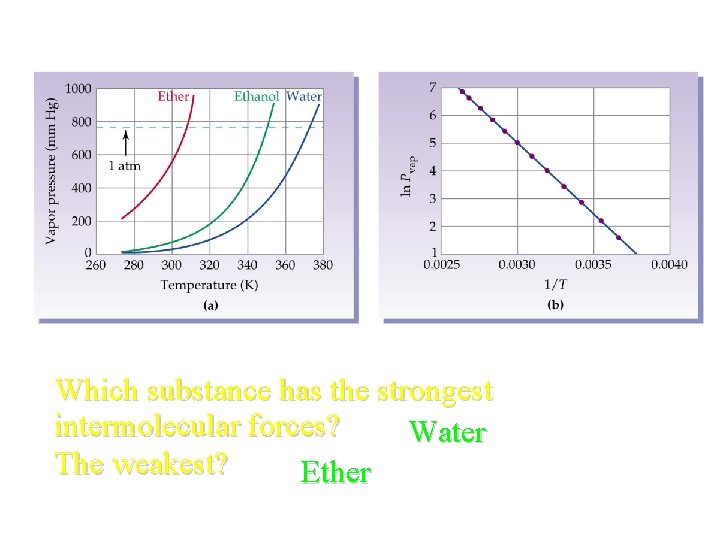
Which substance has the strongest intermolecular forces? Water The weakest? Ether
- Slides: 10