Properties of cell walls prepared using supercritical fluids








- Slides: 19

Properties of cell walls prepared using supercritical fluids Paul Callaghan (VUW), Robert Franich, Stefan J. Hill, and Roger Newman (Scion)

Presentation overview: • Wood cell walls polymers water • Fibre-saturation point • Supercritical CO 2 • Water extraction • Wood material properties • Summary

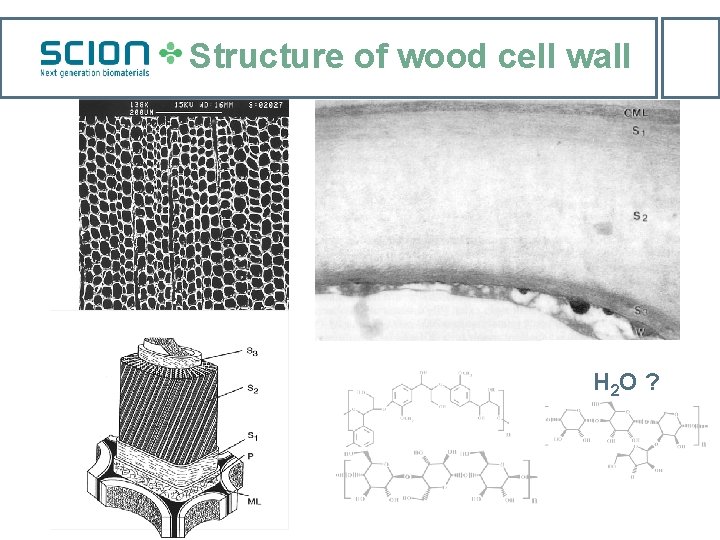
Structure of wood cell wall H 2 O ?

Fibre-saturation point -a chemical phenomenon at molecular/supramolecular scales Cells from green wood – full lumens & fully-water-swollen cell walls – xylem sap FSP – empty lumens & fully-water-swollen cell walls • FSP- independent of scale –log/fibres • Discrete water binding sites - OH • Exothermic wood cell wall hydration – a molecular chemical interaction Stamm et al, 1935 -1971: cell water as gas, liquid, ‘solid solution’ phases

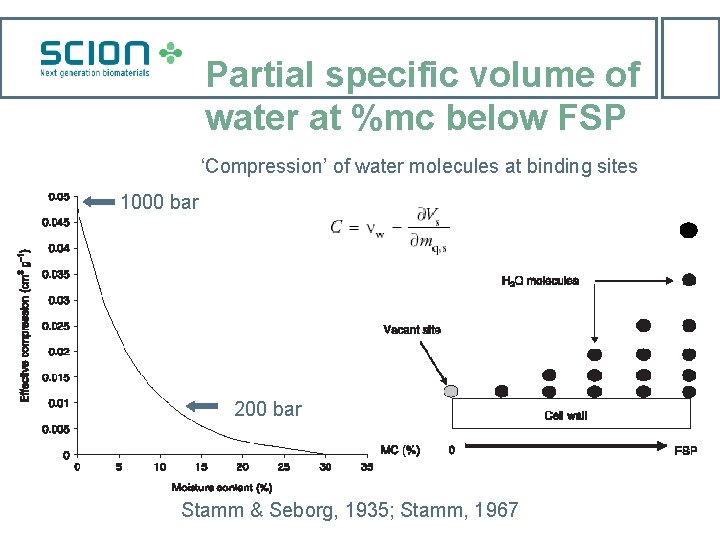
Partial specific volume of water at %mc below FSP ‘Compression’ of water molecules at binding sites 1000 bar 200 bar Stamm & Seborg, 1935; Stamm, 1967

Wood material dynamics Wood at variable moisture content – dimensional, conformational & Mo. E change Water an integral part of the cell wall supramolecular cellulose-hemicellulose-lignin nanocomposite dynamics- ‘Velcro’ mechanics Where is water located and how structured? Difficulty in preparing wood specimens for study of FSP & material dynamics – heating, solvents, azeotropes, critical-point, high-pressure

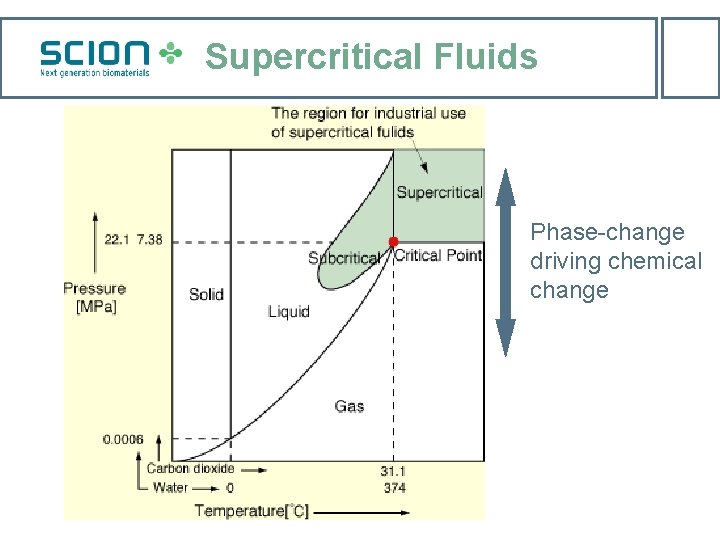
Supercritical CO 2 water extraction Theory: Physical-chemical interaction of cell water and carbon dioxide according to Henry’s Law, the Le Chatelier Principle and the Phase Rule. Variables: P, T, phase (gas, liquid, supercritical) [CO 2] P, 1/T CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 - F=C–P+2 H+ + CO 32 -

The reactant’s properties Rel static permittivity 80. 36 ε 0 at 20 °C 1. 60 ε 0 at 0 °C, 50 B Dipole moment D 1. 85 0 Enthalpy evap k. J/mol 40. 68 15. 33 at – 57. 5°C Entropy evap J/(mol·K) 108. 9 70. 8

Supercritical Fluids Phase-change driving chemical change

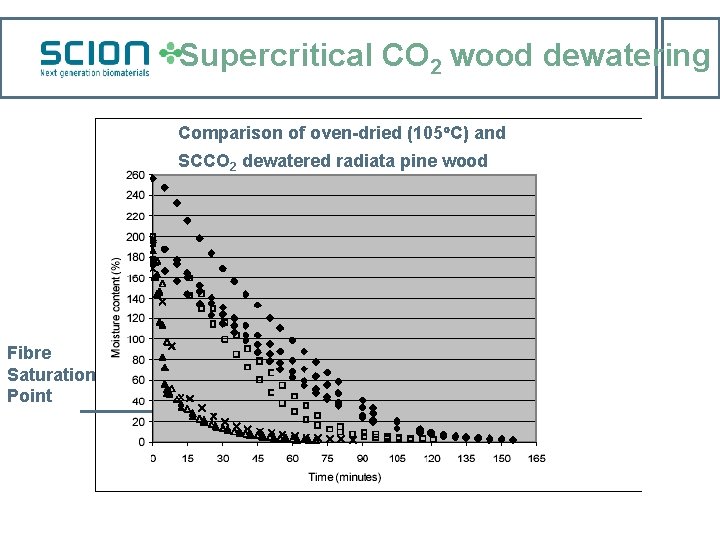
Supercritical CO 2 wood dewatering Comparison of oven-dried (105 C) and SCCO 2 dewatered radiata pine wood Fibre Saturation Point

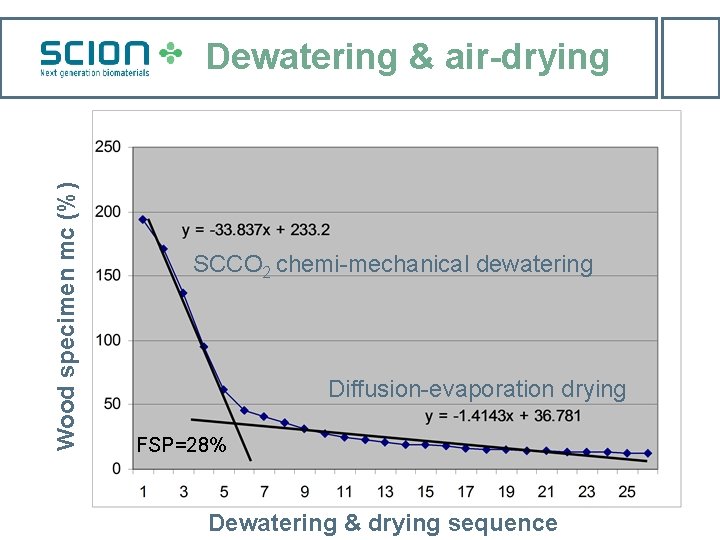
Wood specimen mc (%) Dewatering & air-drying SCCO 2 chemi-mechanical dewatering Diffusion-evaporation drying FSP=28% Dewatering & drying sequence

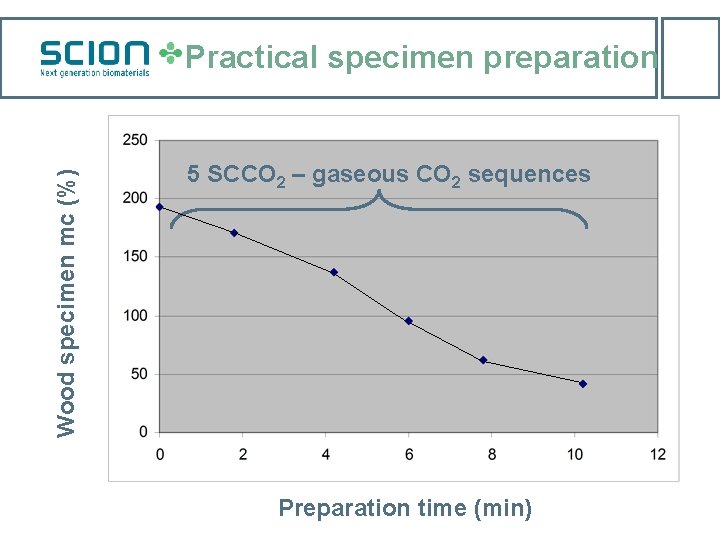
Wood specimen mc (%) Practical specimen preparation 5 SCCO 2 – gaseous CO 2 sequences Preparation time (min)

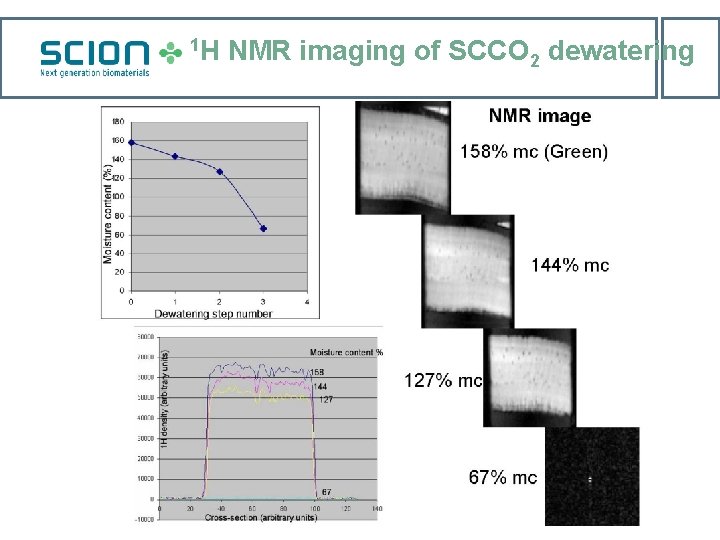
1 H NMR imaging of SCCO 2 dewatering

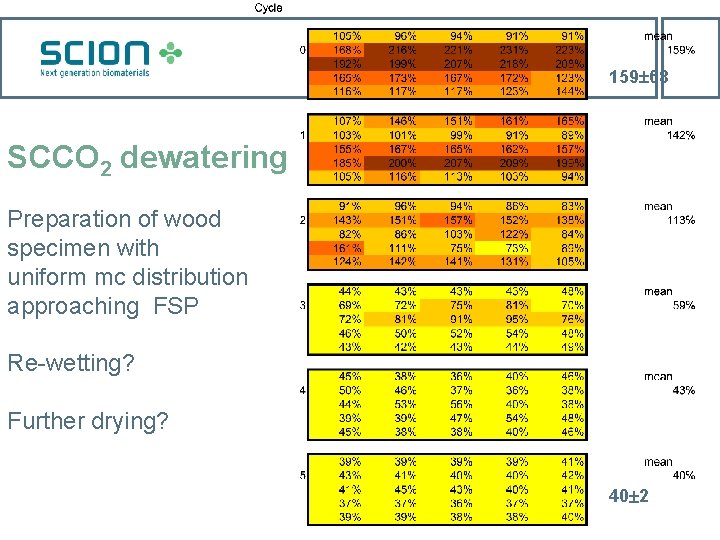
159 68 SCCO 2 dewatering Preparation of wood specimen with uniform mc distribution approaching FSP Re-wetting? Further drying? 40 2

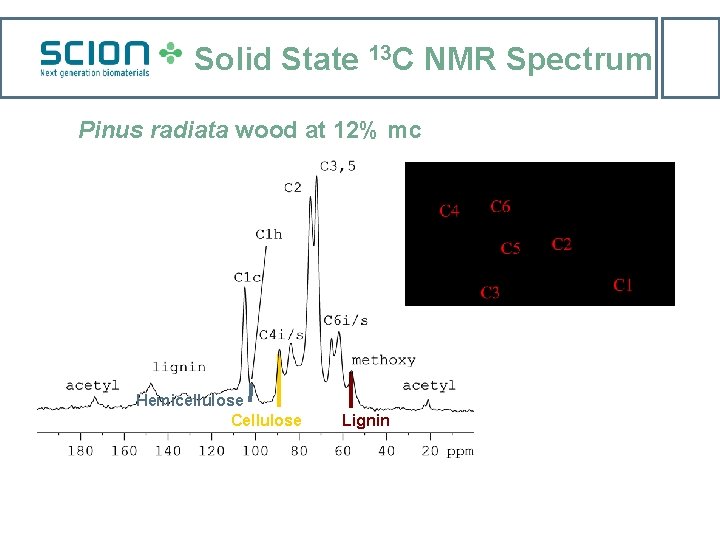
Solid State 13 C NMR Spectrum Pinus radiata wood at 12% mc Hemicellulose Cellulose Lignin

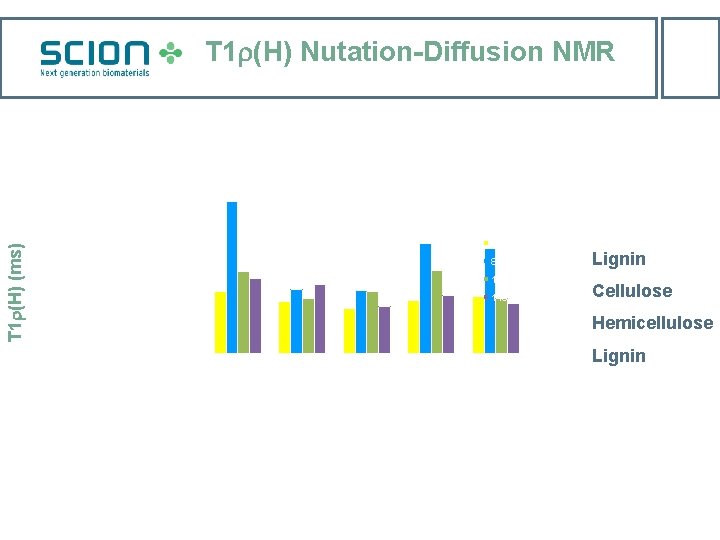
T 1 (H) Nutation-Diffusion NMR 30 25 T 1 (H) (ms) 20 56 ppm 89 ppm 15 102 ppm 10 115 ppm Lignin Cellulose Hemicellulose 5 0 NDW air 105 o. C rewet sc. CO 2 rewet Lignin

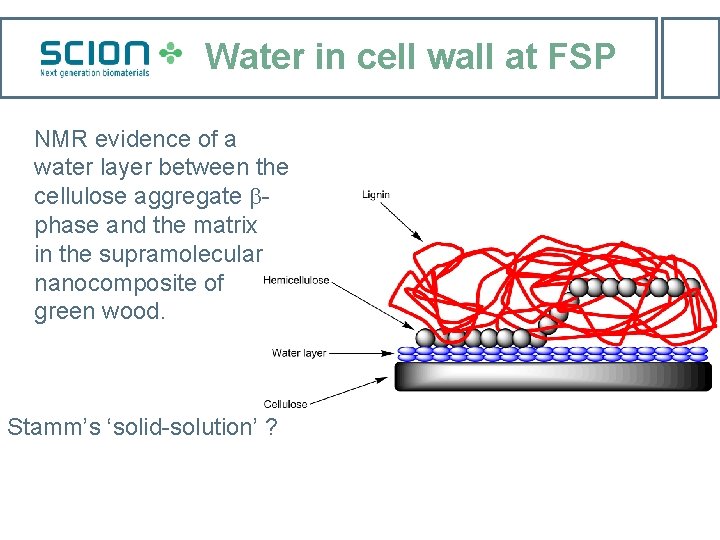
Water in cell wall at FSP NMR evidence of a water layer between the cellulose aggregate phase and the matrix in the supramolecular nanocomposite of green wood. Stamm’s ‘solid-solution’ ?

SCCO 2 dewatering process - summary · Reaction of lumen water with SCCO 2 with reversal of chemistry in gas phase · Bound water bond strength higher than enthalpy of SCCO 2 reaction – no change · Wood material derived approaching FSP from green · Material suitable for wood-water dynamics & cell wall studies – Green to FSP & below

Acknowledgements · Hank Kroese, Suzanne Gallagher, Bernard Dawson & Meeta Patel · FRST – ‘Wood Products for the Future’ Contract C 04 X 0205