Properties of Acids and Bases Property Acid Base

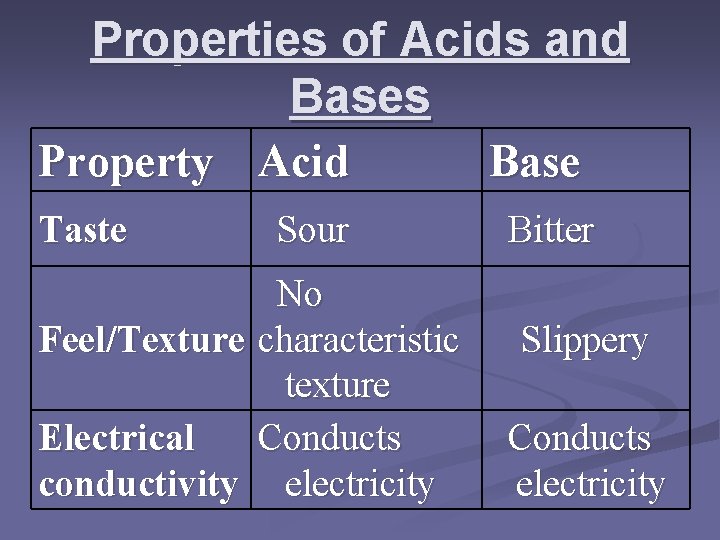
Properties of Acids and Bases Property Acid Base Taste Feel/Texture Electrical conductivity Sour No characteristic texture Conducts electricity Bitter Slippery Conducts electricity
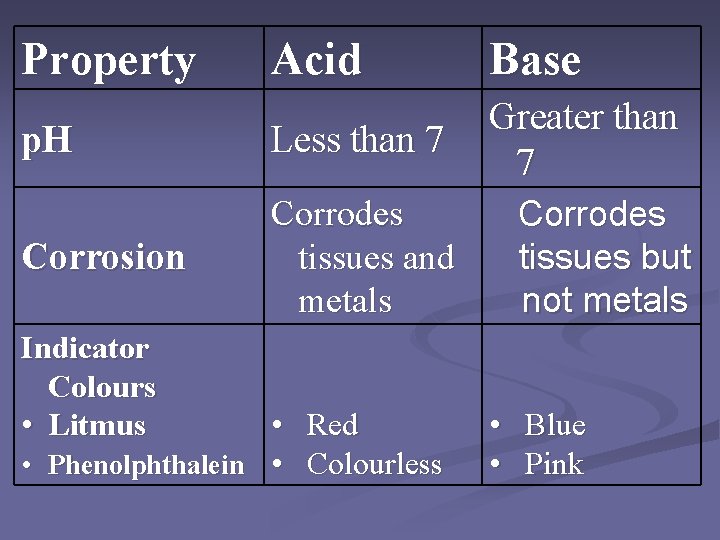
Property Acid Base p. H Less than 7 Greater than 7 Corrosion Corrodes tissues and metals Indicator Colours • Litmus • Red • Phenolphthalein • Colourless Corrodes tissues but not metals • Blue • Pink
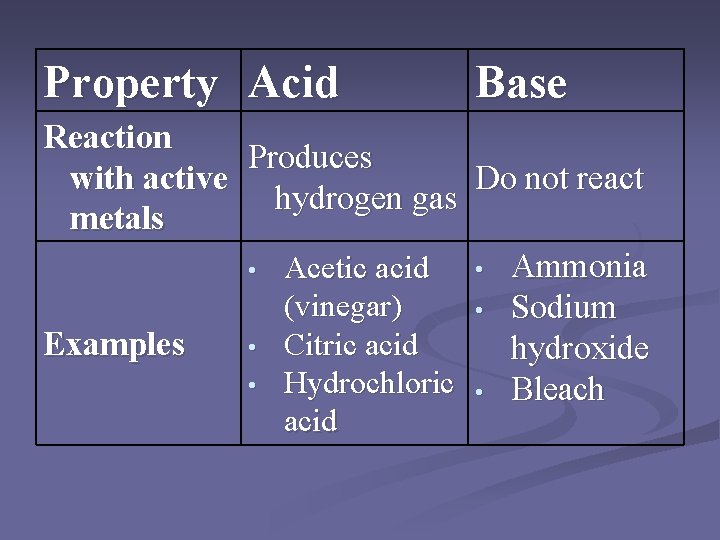
Property Acid Base Reaction Produces with active Do not react hydrogen gas metals • Ammonia • Acetic acid (vinegar) • Sodium • Citric acid Examples hydroxide • Hydrochloric • Bleach acid

Amphiprotic or Amphoteric n. A type of substance that can act as an acid or as a base in different chemical reactions. n Water is the most common amphiprotic compound

n In Acids the late 1800 s, the Swedish scientist Svante Arrhenius proposed that water can dissociate many ionic compounds by separating them into their individual ions n Arrhenius suggested that acids are ionic compounds that contain hydrogen and can dissociate in water to release hydrogen ions into solution (H+1(aq))

n Note that in acids, hydrogen is considered to be a metal n Examples of acids: n hydrochloric acid (HCl) n hydrobromic acid (HBr) n sulfuric acid (H 2 SO 4) n nitric acid (HNO 3) n All acids typically contain hydrogen

n Hydrofluoric (HF) acid is used mainly for industrial purposes (e. g. glass etching, metal cleaning, electronics manufacturing). HF acid may also be found in home rust removers. HF burns can be very severe because dilute solutions deeply penetrate before dissociating, thus causing delayed

n When hydrofluoric acid is added to water (like the water in your blood) it dissociates as follows: n HF(aq) n Note H+(aq) + F-(aq) the production of a hydrogen ion

Bases n Arrhenius defined bases as ionic compounds that dissociate in water to release hydroxide ions (OH-) into solution n Examples of bases: n Lithium hydroxide (Li. OH) n Magnesium hydroxide (Mg(OH)2)

n Sodium hydroxide (Na. OH) is a base commonly used in class and is widely used in industry, mostly as a strong chemical base in the manufacturing of pulp and paper, textiles, drinking water, and detergents. n Sodium hydroxide can cause chemical burns, permanent injury or scarring, and blindness.

n When sodium hydroxide is added to water it dissociates as follows: n Na. OH(aq) Na+(aq) + OH(aq) n Note the production of the hydroxide ion (OH (aq))

n Arrhenius's theory explains why all acids have similar properties to each other (and, conversely, why all bases are similar): because + all acids release H into solution (and all bases release OH-)

Strong and Weak Acids and Bases

n Acids and bases can be divided into two groups; strong or weak n Remember that both acids and bases start as ionic compounds n When these ionic compounds are added to water, they

n Acids produce H+ ions, and bases produce OH- ions n If a compound dissociates completely, it is considered a strong acid or base n HCl(aq) is an example of a strong acid n Therefore, a 1. 0 M solution of HCl(aq) contains virtually no HCl units because they have all separated into H+ and Cl-

n If more than 50% of the compound remains undissociated, it is considered a weak acid or base n Vinegar or acetic acid is a weak acid n Therefore, a 1. 0 M solution of Acetic Acid (CH 3 COOH) contains only some H+ and CH 3 COO- ions

Strong and Weak Acids n Equation n. HA(aq) for a strong acid + H (aq) + A (aq) for a weak acid H+(aq) + A-(aq)

Strong and Weak Bases n Equation for a strong base n. BOH(aq) n Equation B+(aq) + OH-(aq) for a weak base n. BOH(aq) + B (aq) + OH (aq)

Examples of Strong Acids n Hydroiodic acid (HI) n Hydrobromic acid (HBr) n Hydrochloric acid (HCl) n Perchloric acid (HCl. O 4) n Sulfuric acid (H 2 SO 4) n Nitric acid (HNO 3)

Examples of Weak Acids n Phosphoric acid (H 3 PO 4 ) n Hydrofluoric acid (HF) n Acetylsalicylic acid (C 6 H 4 OCOCH 3 CO 2 H) n Acetic Acid (CH 3 COOH)

Examples of Strong Bases n Potassium hydroxide (KOH) n Barium hydroxide (Ba(OH)2) n Cesium hydroxide (Cs. OH) n Sodium hydroxide (Na. OH) n Strontium hydroxide (Sr(OH)2) n Calcium hydroxide (Ca(OH)2) n Lithium hydroxide (Li. OH)

Examples of Weak Bases n Ammonia (NH 3) n Magnesium (Mg(OH)2 ) hydroxide

n Page 300 # 2 and 3
- Slides: 24