Properties of acids and bases l l l

Properties of acids and bases l l l l Sour/tart taste Conduct electricity Litmus turns red Release H+ into water p. H < 7 Neutralize a base Usually start with H Most food items l l l l Slippery to the touch Bitter taste Conduct electricity Litmus turns blue Release OH- into water Neutralize an acid p. H > 7 Most cleaning items

An acid is a compound that gives H+ (or H 3 O+) in water. Acids generally begin with H. HCl + H 2 O H 3 O+1 + Cl-1 HCl H+1 + Cl– 1 A base is a compound that gives OH- in water. Bases usually end in –OH. Na. OH Na+1 + OH-1 An indicator is a colored substance that can exist in either an acid or base solution.

Two common indicators: Litmus paper: red blue 6. 8 7. 3 Phenolphthalein: clear pink 8. 3 8. 4
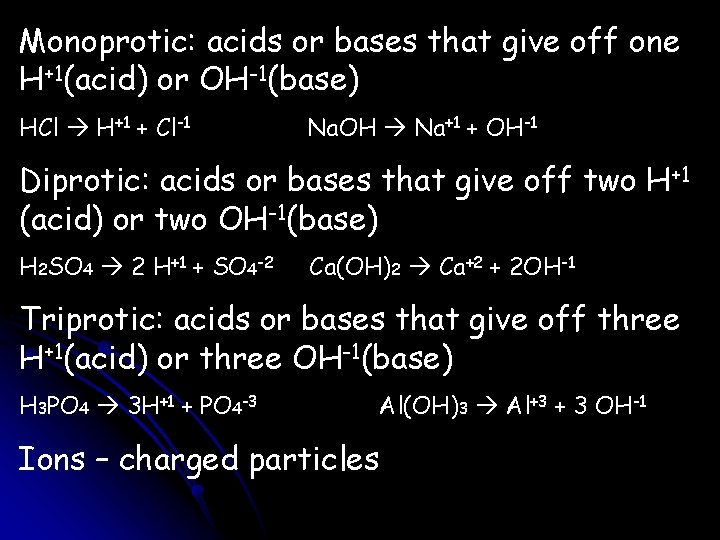
Monoprotic: acids or bases that give off one H+1(acid) or OH-1(base) HCl H+1 + Cl-1 Na. OH Na+1 + OH-1 Diprotic: acids or bases that give off two H+1 (acid) or two OH-1(base) H 2 SO 4 2 H+1 + SO 4 -2 Ca(OH)2 Ca+2 + 2 OH-1 Triprotic: acids or bases that give off three H+1(acid) or three OH-1(base) H 3 PO 4 3 H+1 + PO 4 -3 Al(OH)3 Al+3 + 3 OH-1 Ions – charged particles

Naming acids Common acids normally begin with H. When you see a compound that begins with H, it should be named as an acid. The name of the acid depends on the negative ion: • If the name of the anion ends in –ide Hydro (anion without –ide) –ic acid HCl – hydrochloric acid HBr – hydrobromic acid H 2 S – hydrosulfuric acid

• If the anion ends in –ate, everything is the same except do not add the prefix hydro(anion name without –ate) –ic acid HNO 3 – nitric acid HCl. O 3 – chloric acid H 2 SO 4 – sulfuric acid

Common acids and bases l l l l HCl HNO 3 H 2 SO 4 HC 2 H 3 O 2 HBr H 2 CO 3 H 3 PO 4 l l l Na. OH KOH Ca(OH)2 Mg(OH)2 Ba(OH)2

Bronsted Acid-Bases Acid – a substance that gives up H+ Conjugate Base – what is left after the acid gives up H+ Base – a substance that gains H+ Conjugate Acid – what is left after the base gains H+ Plain acids and bases are on the left side of a reaction, the conjugate acid and base are on the right side of a reaction.
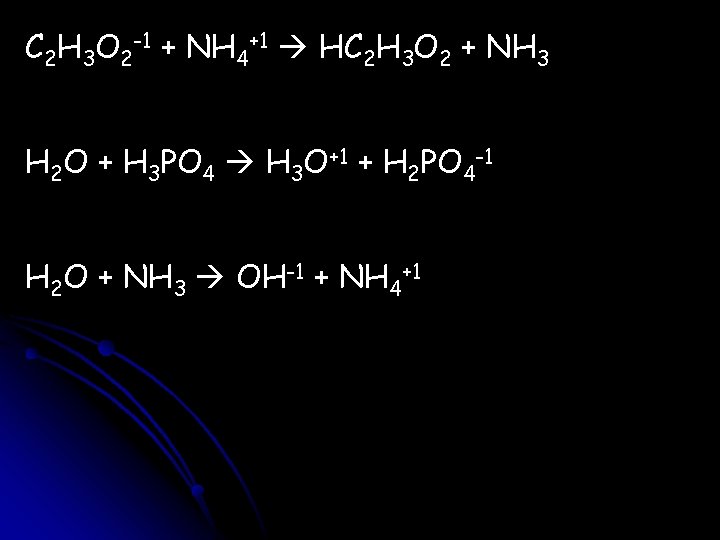
C 2 H 3 O 2 -1 + NH 4+1 HC 2 H 3 O 2 + NH 3 H 2 O + H 3 PO 4 H 3 O+1 + H 2 PO 4 -1 H 2 O + NH 3 OH-1 + NH 4+1
![[ ] = concentration (unit of molarity) In pure water at 25 o. C [ ] = concentration (unit of molarity) In pure water at 25 o. C](http://slidetodoc.com/presentation_image/4fa6b48fd93762c5b498c5ef63fba882/image-10.jpg)
[ ] = concentration (unit of molarity) In pure water at 25 o. C [H+1] = 1. 00 E-7 M [OH-1] = 1. 00 E-7 M [H+1]x[OH-1] = 1. 00 E-7 x 1. 00 E -7 = 1. 00 E-14 One way of expressing the [H+1] or the [OH -1] is the p. H = -log[H+1] p. OH = -log[OH-1]
![Important Formulas: p. H + p. OH = 14. 00 [H+1] x [OH-1] = Important Formulas: p. H + p. OH = 14. 00 [H+1] x [OH-1] =](http://slidetodoc.com/presentation_image/4fa6b48fd93762c5b498c5ef63fba882/image-11.jpg)
Important Formulas: p. H + p. OH = 14. 00 [H+1] x [OH-1] = 1. 00 E-14 p. H = -log[H+1] p. OH = -log[OH-1] p. H Scale 0 6. 99 acid 7. 00 neutral 7. 01 14. 00 base

To calculate the p. H (or the p. OH) 1. (-) 2. log 3. [H+1] (or [OH-1] To calculate [H+1] (or [OH-1]) 1. 10 x (2 nd log) 2. (-) 3. p. H (or p. OH)

Neutralization Reactions An acid will react with a base and neutralize each other. The result when they are mixed is ALWAYS salt and water. Acid + Base Salt + Water HA + BOH BA + HOH HCl + Na. OH Na. Cl + HOH

To predict the products of neutralization reactions: 1. Recognize that it is a double replacement reaction 2. Pair up the new products: one will always be HOH (or water, H 2 O) 3. You can get the correct charges from H+1 or OH-1 4. Get the correct formulas by crossing the charges 5. Balance the reaction

H 2 SO 4 + Al(OH)3 • H 2 SO 4 + Al(OH)3 HOH + Al. SO 4 (pair up new products) • H 2 SO 4 + Al(OH)3 HOH + Al 2(SO 4)3 (get the correct formulas) • 3 H 2 SO 4 + 2 Al(OH)3 6 HOH + Al 2(SO 4)3 (balance the reaction)

Titration – a solution of a known concentration is reacted with a known volume of a solution of unknown concentration. At the endpoint, an indicator will change colors. At that point, the [H+1] = [OH-1]. From this information, the unknown concentration can be determined.

1. Write a balanced reaction 2. Label all numbers 3. Use unit analysis 4. Start with the volume of the compound that you know both volume and concentration 5. vol moles A moles B conc B

27. 5 milliliters of H 2 SO 4 is exactly neutralized by 39. 3 milliliters of 0. 437 M Na. OH. What is the concentration of the acid? H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 HOH Acid: 27. 5 ml Base: 39. 3 ml 0. 0393 L Na. OH 0. 437 M 0. 437 mol Na. OH 1 mol H 2 SO 4 1 L Na. OH 2 mol Na. OH 1 = 0. 0275 L H 2 SO 4 0. 312 M H 2 SO 4

There is an alternate way to do titration calculations that does not involve writing balanced reactions or unit analysis: PLHABTSM® M 1 V 1 P 1 = M 2 V 2 P 2 M 1 = conc of acid M 2 = conc of base V 1 = volume of acid V 2 = volume of base P 1 = protacticity P 2 = protacticity

Protacticity P 1 = # of H+1 per molecules P 2 = # of OH-1 per molecules 1. Read the problem 2. Label all of the numbers 3. Identify the unknown 4. Use M 1 V 1 P 1 = M 2 V 2 P 2 5. Solve for the unknown

27. 5 milliliters of H 2 SO 4 is exactly neutralized by 39. 3 milliliters of 0. 437 M Na. OH. What is the concentration of the acid? M 1 = M 2 = V 1 = V 2 = P 1 = P 2 =
- Slides: 21