Properties Chemicalexhibited by matter as it undergoes changes

Properties: Chemical-exhibited by matter as it undergoes changes in composition 2 Mg + O 2 -----> 2 Mg. O Physical-exhibited by matter without a change in composition. Color, state, density, melting point, boiling point, etc. Extensive-depends on the amount of matter (i. e. mass, heat capacity. . . ) Intensive-independent of the amount of matter (color, state, density, chemical properties, . . . ) Physical & Intensive:

Chemical Change-one or more substances are either used up or formed and energy is absorbed or released. (Rxn’s) 2 Mg + O 2 2 Mg. O Physical Change-change in physical properties without a change in composition.
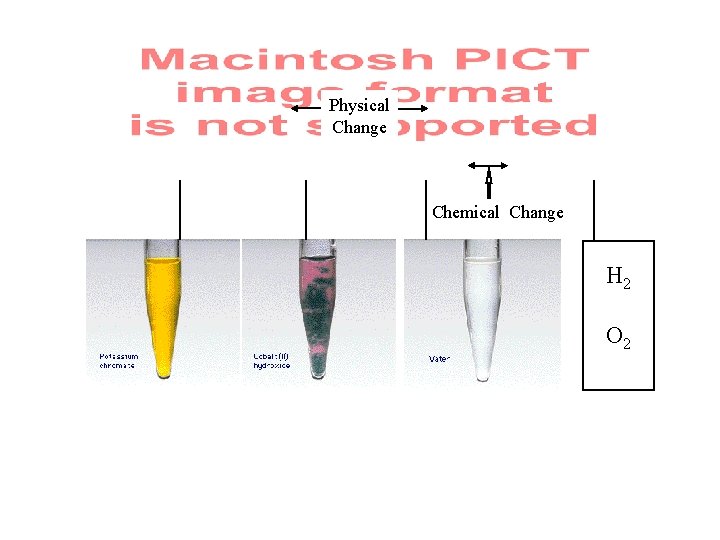
Physical Change Chemical Change H 2 O 2

SI=International System of Units Length = meter Volume = m 3 Mass = kg Temperature = K (Kelvin) Time = s (seconds) Energy = J (Joule) K=o. C+273. 15

% Composition %A (mass)= Density (density)= Specific =Sp. Gr. = (substance) Gravity (water) Specific Heat = Parts of A (by mass) 100 parts mixture (by mass) mass (g) volume (ml) (water)=1. 00 g/ml @ 25 o. C (Amount of heat in J) (mass of substance in g)(Temperature change in K/o. C) Heat Capacity= (Csp)(mass) units=(J/ o. C) Purity is given by the % Composition

Stoichiometry Composition Stoichiometry-Quantitative relationship among elements in a compound. (H 2 O; 2 H atom and 1 O atom per 1 H 2 O molecule) Reaction Stoichiometry-Quantitative relationship among substances as they undergo chemical changes. (P 4+6 Cl 2 4 PCl 3; 1 P 4 molecule reacts with 6 Cl 2 molecules to produce 4 PCl 3 molecules) Dalton’s Atomic Theory: 1) An element is composed of extremely small, indivisible particles called atoms 2) All atoms of a given element have identical properties 3) Atoms cannot be created, destroyed or transformed into atoms of another element. 4) Compounds are formed when atoms of different elements combine with one another in small whole-number ratios. 5) The relative numbers and kinds of atoms are constant in a given compound.

Atom-the smallest particle of an element that maintains its chemical identity through all chemical and physical changes and are composed 3 fundamental particles-electrons, protons and neutrons Molecule-the smallest particle of an element or compound that can have a stable independent existence. Diatomic molecules - O 2, N 2, H 2, F 2, I 2, Br 2, Cl 2. . . (Know these) Formula units: Ionic Compounds-consists of an extended array of ions Know Table 2 -3 in the book Cation=positively charged ion anion=negatively charged ion

Know how to calculate the MW/FW of any molecule/formula unit example: MW of water = 18. 0 amu/molecule = 18. 0 g/mol 2*AW(H)+AW(O)=MW (2*1. 01)+(16. 0)=18. 0 FW sodium chloride = 58. 4 amu/formula unit=58. 4 g/mol AW(Na) + AW(Cl)=FW 23. 0 + 35. 4 = 58. 4 1 mol= 6. 022*1023 particles g MW mole NA just a unit conversion Stoich. particles mole X mol Y

Empirical (Simplest) Formula - The smallest whole-number ratio of atoms present in a compound. Molecular Formula of Caffeine = C 8 O 2 N 4 H 10 Empirical Formula of Caffeine = C 4 ON 2 H 5 The Empirical Formula is found by determining the % composition experimentally. The Molecular Formula is determined from the Empirical Formula and the experimentally determine Molecular Weight. n= MW EW

You should be able to determine if a chemical reaction is balanced and if it isn’t, you should be able to balance it. You should be able to answer questions like: given the mass of CS 2 and excess O 2 how many grams of CO 2 or SO 2 will be produces? given the mass of CS 2 and the mass of O 2 how many grams of CO 2 or SO 2 will be produced? Which reactant is the limiting reagent (reactant)?
- Slides: 10