Properties and Theories of Acids and Bases Chapter

Properties and Theories of Acids and Bases Chapter 14 Sections 1 and 2

Acids • Aqueous solutions of acids have a ____ taste (never taste chemicals) • Will change the color of acid-base _____ – blue litmus changes from ____to _____ • Some acids react with active metals and release ________ gas • React with ______to produce salts and water • Acidic solutions can conduct _______ (varies with strength)

Naming Acids • ______: contains only two different elements; hydrogen and one other more electronegative element (nonmetal) – The name of a binary acid begins with the prefix _______ – The root of the name of the second nonmetal element follows this prefix – The name then ends with the suffix ______

Binary Acids • A binary acid is an aqueous solution of a compound containing hydrogen and a nonmetal. • The formula of an acid always begins with H: – HCl(aq) • Binary acids are named by using the prefix hydro– before the element stem and adding the suffix –ic acid. – HF(aq) is hydrofluoric acid; HI(aq) is hydroiodic acid. ***See Table 1 on page 468*** © 2014 Pearson Education, Inc. Chapter 6

Naming Oxyacids • _____: an acid that is a compound of hydrogen, oxygen, and a third element, usually a nonmetal – Usually hydrogen and a ________ – Also use the term _____acid

Ternary Oxyacids • Ternary oxyacids are aqueous solutions of a compound containing hydrogen and an oxyanion. • If the acid is derived from an oxyanion ending in ____, the suffix is changed to _______acid. – HNO 3(aq) is nitric acid (from NO 3–, nitrate ion). • If the acid is derived from an oxyanion ending in ____, the suffix is changed to _______acid. – HNO 2(aq) is nitrous acid (from NO 2–, nitrite ion). © 2014 Pearson Education, Inc. Chapter 6

Oxyanions and Oxyacids • Below is an example of ternary oxyacid and ternary compound naming. Ternary Oxyacid Ternary Compound HCl. O(aq) hypochlorous acid Na. Cl. O sodium hypochlorite HCl. O 2(aq) chlorous acid Na. Cl. O 2 sodium chlorite HCl. O 3(aq) chloric acid Na. Cl. O 3 sodium chlorate HCl. O 4(aq) perchloric acid Na. Cl. O 4 sodium perchlorate ***See Table 2 on page 469. *** © 2014 Pearson Education, Inc. Chapter 6

Bases • Aqueous solutions of bases taste ____ (never taste chemicals) • Bases change the color of acid-base ____ – Red litmus changes from red to ____ • Dilute aqueous solutions of bases feel ______ • Bases react with _____to produce salts and water • Bases can conduct _____current (varies)

Arrhenius Acid and Base Theory • ___________: a chemical compound that increases the concentration of hydrogen ions, H+, in aqueous solution • ___________: a substance that increases the concentration of hydroxide ions, OH-, in aqueous solution
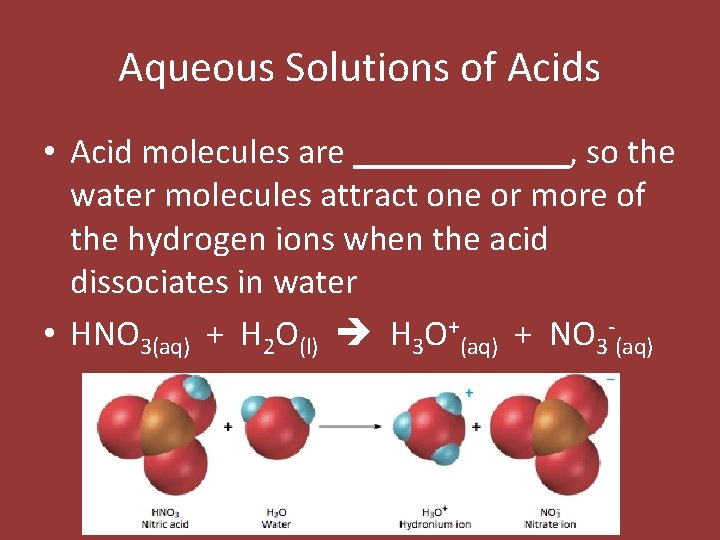
Aqueous Solutions of Acids • Acid molecules are ______, so the water molecules attract one or more of the hydrogen ions when the acid dissociates in water • HNO 3(aq) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq)

Strength of Acids • _________: one that dissociates completely in aqueous solutions; strong electrolyte! • Concentration is not linked to strength • ________: one that releases few hydrogen ions in aqueous solution • When a weak acid is dissolved in water, the reverse reaction occurs simultaneously (to a certain extent)

Weak Acid Solution • HCN(aq) + H 2 O(l) H 3 O+(aq) + CN-(aq) • The number of the hydrogen atoms in the formula does ____ indicate acid strength • Organic acids, that contain COOH are usually weak acids


Aqueous Solutions of Bases • Many bases are ionic compounds containing a metal cation and the hydroxide anion, so they dissociate in water:

Strength of Bases • ________: one that completely dissociates in solution • Strong bases are also strong electrolytes (as well as strong acids) • __________: a substance that dissolves in water to give a solution that conducts an electric current

Strength of Bases • Bases that are not very soluble do not produce a large number of hydroxide ions when added to water • They are __________. • Some bases, like NH 3, are soluble, but do not form many OH- ions, so they are weak


Brønsted-Lowry Theory • Named for 2 chemists J. N. Brønsted and T. M. Lowry who independently expanded the Arrhenius theory • ____________: a molecule or ion that is a proton donor Ø(H+ is a proton) • ____________: a molecule or ion that is a proton acceptor

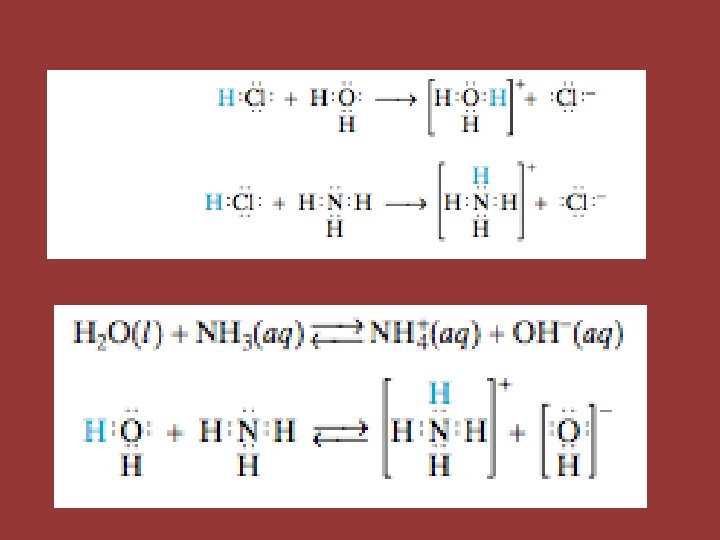
Brønsted-Lowry Acid • Any Arrhenius acid also qualifies as a Brønsted-Lowry acid, but not vice-versa • HCl + NH 3 NH 4+ + Cl • A proton is donated by the hydrogen chloride molecule even though it is not aqueous • Water can also act as a Brønsted-Lowry acid

Monoprotic and Polyprotic Acids • ________acid: an acid that can donate only one proton (hydrogen ion) per molecule – Examples: HCl and HNO 3 • ________acid: an acid that can donate more than one proton per molecule – Examples: H 2 SO 4 and H 3 PO 4 • Specifically: – Diprotic acid: an acid that donates two protons

Lewis Theory of Acids and Bases • __________: an atom, ion, or molecule that accepts an electron pair to form a coordinate covalent bond • __________: an atom, ion, or molecule that donates an electron pair to form a coordinate covalent bond

• An example of a Lewis acid-base reaction is the reaction between boron trifluoride and ammonia

Summary of Theories

Acid-Base Reactions Section 14. 3

Conjugate Base • __________: the species that remains after a Brønsted-Lowry acid has given up a proton Acid conjugate base

Conjugate Acid • ____________: the species that is formed when a Brønsted-Lowry base gains a proton base conjugate acid

Acid-Base Reactions • Usually, acid-base reactions are __________systems. • The forward and reverse reactions occur. • They involve conjugate acid-base pairs. CH 3 COOH(aq) + H 2 O(l) acid base H 3 O+(aq) + CH 3 COO - (aq) conjugate acid base

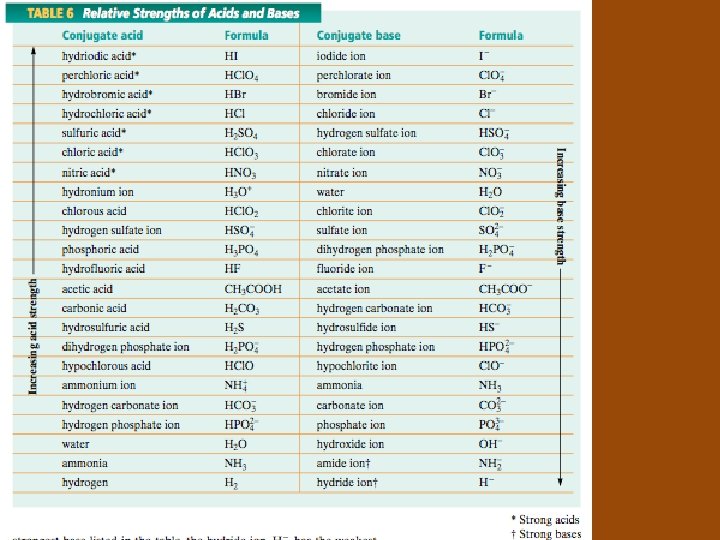
Strength of Conjugate Acids and Bases • The stronger an ______ is, the weaker its ___________. • The stronger a _______is, the weaker its ___________.

Amphoteric Compounds • __________: any species that can react as either an acid or a base • Example: H 2 O can act as either an acid or a base • H 2 SO 4(aq) + H 2 O(l) H 3 O+(aq) + HSO 4 -(aq) • NH 3(g) + H 2 O(l) NH+4(aq) + OH-(aq)

Neutralization Reactions • A strong acid and a strong base react to form a ____and _____. • Aqueous HCl completely dissociates in water to form ____ and ______. • Aqueous Na. OH completely dissociates in water to form ____ and ______.
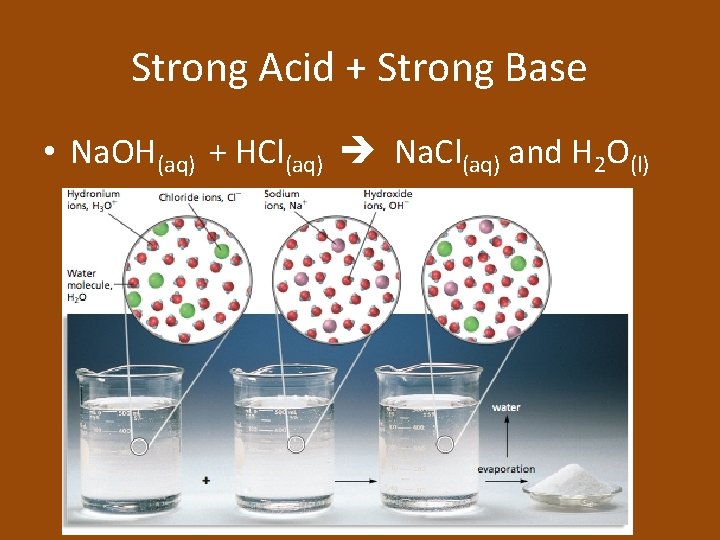
Strong Acid + Strong Base • Na. OH(aq) + HCl(aq) Na. Cl(aq) and H 2 O(l)

Vocabulary • ____________: (in aqueous solutions) the reaction of hydronium ions and hydroxide ions to form water molecules • ________: an ionic compound composed of a cation from a base and an anion from an acid

Acid Rain • Pure rain water is slightly acidic because of dissolved carbon dioxide that forms carbonic acid with a p. H = 5. 6 • Acid rain is defined as rain with a p. H < 5. 6 • Other acids cause rain to have a lower p. H

More about Acid Rain • Main “bad” acids in rain are sulfuric acid and nitric acid • Caused by the reaction of oxides of sulfur and oxides of nitrogen • Can be deposited in rain water, fog, or snow • Drastically affects lakes, streams, ponds and soil
- Slides: 36