PROlonged Bivalirudin Infusion Versus Intraprocedural only Random Ized

PROlonged Bivalirudin Infusion Versus Intraprocedural only Random. Ized study. The PROBI VIRI study. Bernardo Cortese, MD Ospedale della Misericordia Grosseto Italy

Disclosures Consultant (TMC, 2008).

BACKGROUND Why do we use Bivalirudin? Short and long term follow up data from recent RCT.

Acuity trial HD White et Al. , JACC 52; 807 GW Stone et Al. , NEJM 2006; 355: 2203
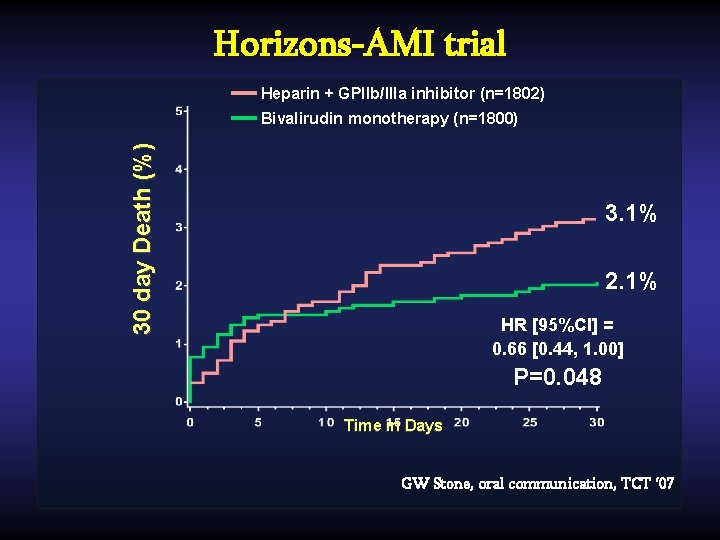
Horizons-AMI trial Heparin + GPIIb/IIIa inhibitor (n=1802) 30 day Death (%) Bivalirudin monotherapy (n=1800) 3. 1% 2. 1% HR [95%CI] = 0. 66 [0. 44, 1. 00] P=0. 048 Time in Days GW Stone, oral communication, TCT ‘ 07

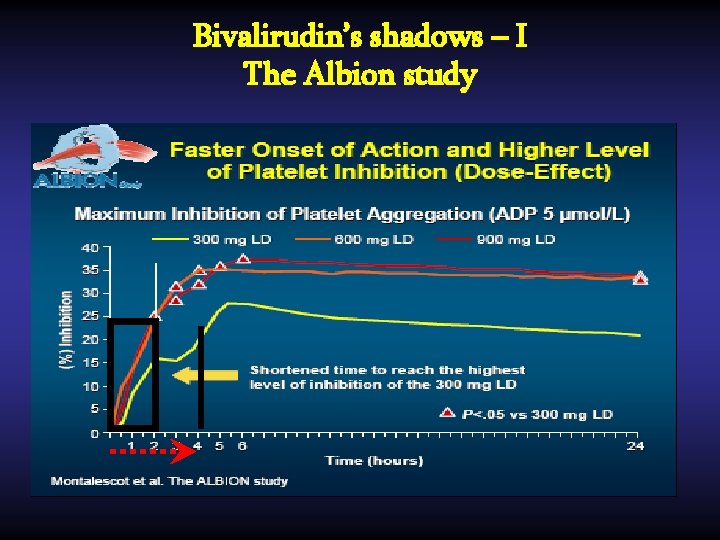
Bivalirudin’s shadows – I The Albion study
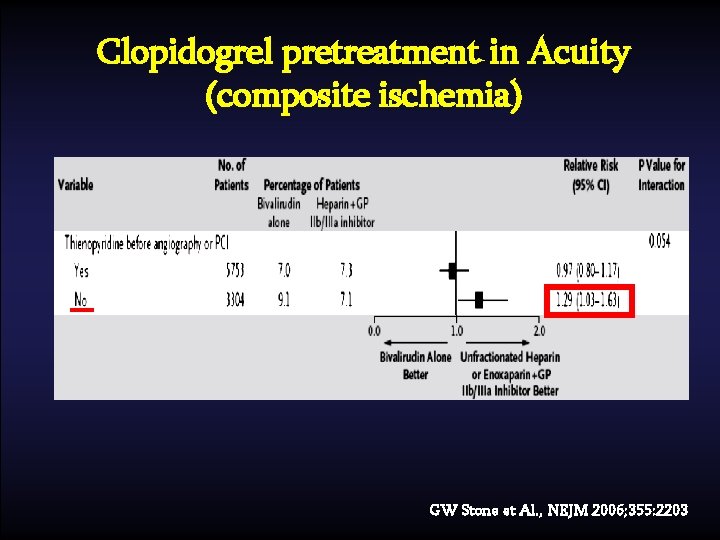
Clopidogrel pretreatment in Acuity (composite ischemia) GW Stone et Al. , NEJM 2006; 355: 2203

Bivalirudin’s shadows – II Isar React-3 trial A Kastrati et Al. , NEJM 2008; 359: 688

Bivalirudin’s shadows – III Horizons-AMI trial GW Stone et Al. , NEJM 2008; 358: 2218

Hypothesis: a prolonged infusion of bivalirudin after PCI would overcome these flaws? ?
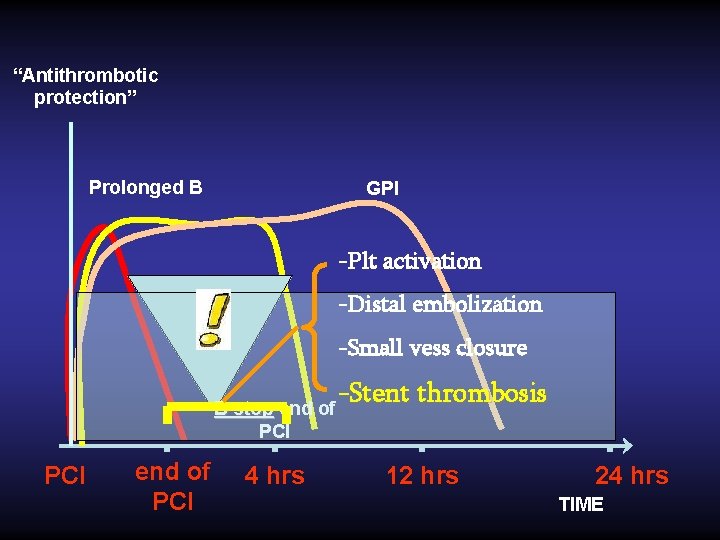
“Antithrombotic protection” Prolonged B GPI -Plt activation -Distal embolization -Small vess closure B stop end of PCI 4 hrs -Stent thrombosis 12 hrs 24 hrs TIME
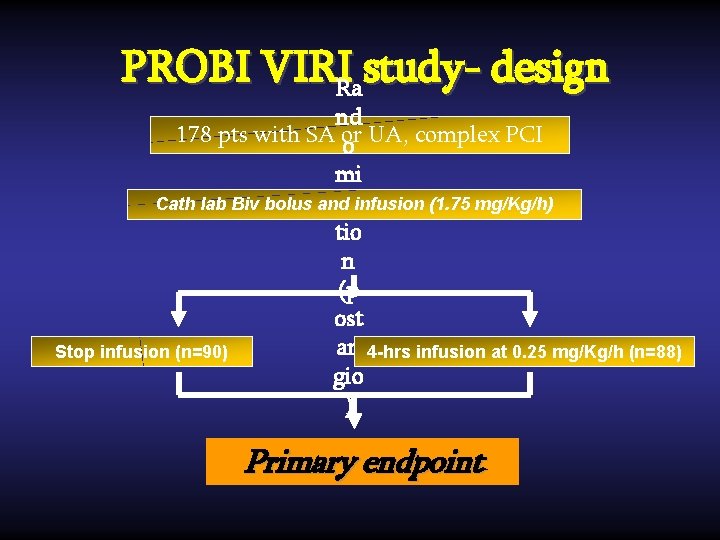
PROBI VIRIRa study- design nd 178 pts with SA or o UA, complex PCI mi Cath lab Biv bolus and zainfusion (1. 75 mg/Kg/h) tio n (p ost an 4 -hrs infusion at 0. 25 mg/Kg/h (n=88) Stop infusion (n=90) gio ) Primary endpoint

Inclusion/Exclusion criteria Stable or unstable angina and at least one of the following: Ø at least one B 2/C lesion (ACC/AHA classification) or Ø planned MV PCI any elevation of pre-procedural CK-MB or Tn, creatinine clearance <30 ml/min, previous use of heparin (2 hrs) or GPI, INR>1. 5

Study endpoints Primary: Primary incidence of procedural-related MI (CK-MB ≥ 3 ULN). Secondary: Secondary in-hospital bleedings; 30 -days and 6 months MACEs.

Statistical Methodology • • randomization after angiography; analysis by intention-to-treat; superiority for P. E. non-inferiority for S. E. • power analysis: to reduce post PCI MI incidence from 12% (control group) to 6% (prol biv group), a sample size of 160 pts would provide study 80% power to meet P. E. . Planned population: 175 pts.
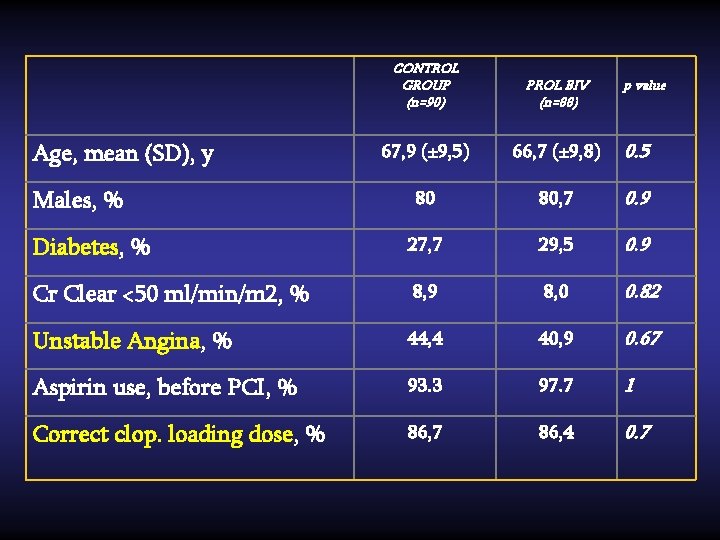
CONTROL GROUP (n=90) PROL BIV (n=88) 67, 9 (± 9, 5) 66, 7 (± 9, 8) 0. 5 80 80, 7 0. 9 Diabetes, % 27, 7 29, 5 0. 9 Cr Clear <50 ml/min/m 2, % 8, 9 8, 0 0. 82 Unstable Angina, % 44, 4 40, 9 0. 67 Aspirin use, before PCI, % 93. 3 97. 7 1 Correct clop. loading dose, % 86, 7 86, 4 0. 7 Age, mean (SD), y Males, % p value
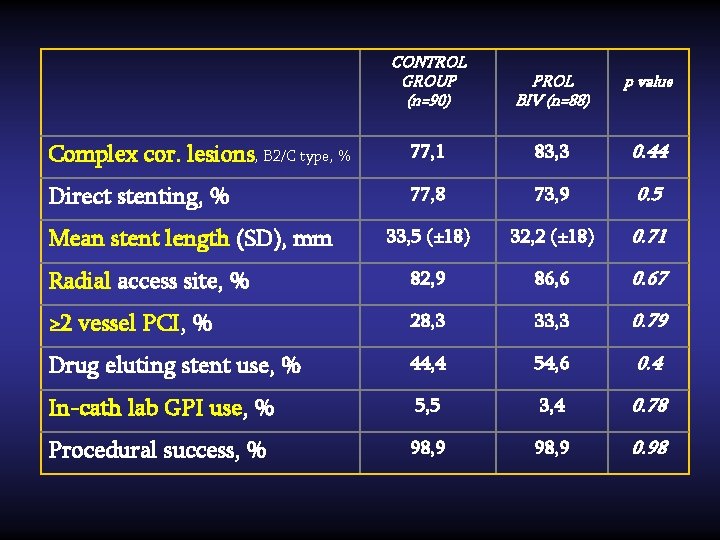
CONTROL GROUP (n=90) PROL BIV (n=88) p value Complex cor. lesions, B 2/C type, % 77, 1 83, 3 0. 44 Direct stenting, % 77, 8 73, 9 0. 5 33, 5 (± 18) 32, 2 (± 18) 0. 71 Radial access site, % 82, 9 86, 6 0. 67 ≥ 2 vessel PCI, % 28, 3 33, 3 0. 79 Drug eluting stent use, % 44, 4 54, 6 0. 4 In-cath lab GPI use, % 5, 5 3, 4 0. 78 Procedural success, % 98, 9 0. 98 Mean stent length (SD), mm
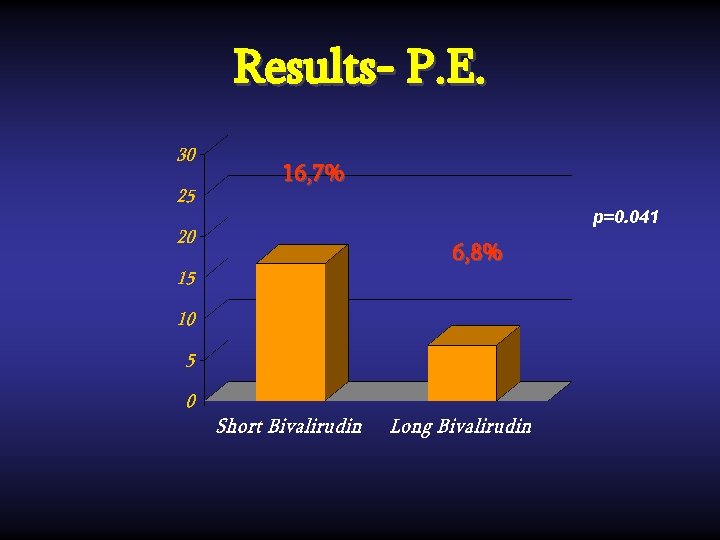
Results- P. E. 16, 7% p=0. 041 6, 8%
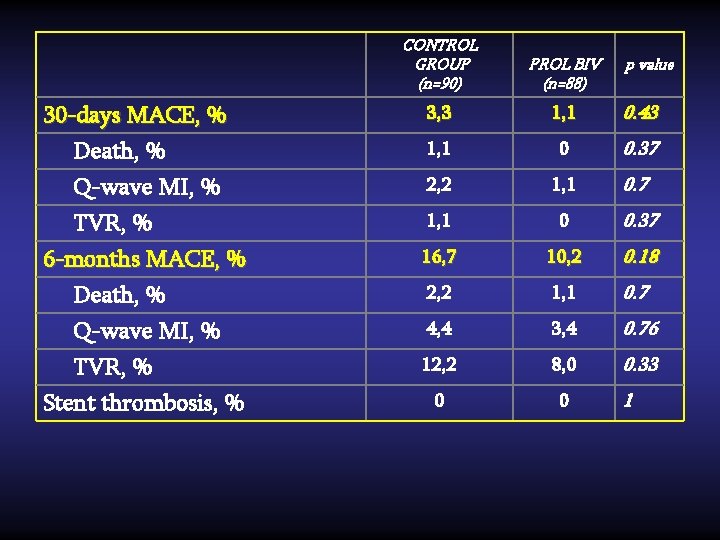
CONTROL GROUP (n=90) PROL BIV (n=88) 30 -days MACE, % Death, % 3, 3 1, 1 0. 43 1, 1 0 0. 37 Q-wave MI, % TVR, % 6 -months MACE, % Death, % 2, 2 1, 1 0. 7 1, 1 0 0. 37 16, 7 10, 2 0. 18 2, 2 1, 1 0. 7 4, 4 3, 4 0. 76 12, 2 8, 0 0. 33 0 0 Q-wave MI, % TVR, % Stent thrombosis, % p value 1

CONTROL GROUP (n=90) PROL BIV (n=88) 86, 6% radial In-hospital Major Bleedings, % 1, 1 In-hospital Minor Bleedings, % 3, 3 p value 1, 1 0. 87 3, 4 0. 96 access!

Limitations of the study Øpost PCI bivalirudin infusion rate. Øthe last word about safety (radial access)?

CK MB increase after PCI IS. REACT 3 ARMYDA 2 Briguori et Al. (EHJ 04; 25: 1822) PROBI VIRI (stop Biv arm) Periproc MI 5. 4% 15% (>2 ULN) 16. 4% 16. 7% Any CK MB incr. above ULN n. a. 26% n. a. 28. 9% Preproc. CK MB increase 0% 0% clop 100% 86. 7% Complex lesions (B 2/C) 67. 5% 81% 64% 77. 1% Stent length, mm 22. 8 (± 11) 20. 8 (± 10) 21 (± 10) 33, 5 (± 18) n. a. 9% 10% 28. 3% Correct loading MV PCI

Conclusions In a complex PCI setting, bivalirudin prolonged infusion after PCI seems a promising choice to reduce myocardial injury. Urgent or Emergent PCI

Bivalirudin’s “Appendix”
- Slides: 25