Project 1 Acid Gas Cleanup CBE 160 February

Project 1: Acid Gas Cleanup CBE 160 February 16, 2017 Group 5 Shivya Bansal Ilyssa Evans Paige Liu Thomas Wu
Executive Summary Introduction Process & Calculations ◆ Problem Statement and Objectives ◆ Overall Material Balance ◆ Acid Gas Removal Process ◆ Equipment List and Sizing ◆ Technical Challenges Conclusions ◆ Alternatives ◆ Future Work

Problem & Objectives Motivation Use Natural Gas to create 1000 metric tons of Ammonia / day Problem Natural Gas is contaminated with H 2 S, CO 2, and C 2 H 6

Problem & Objectives Motivation Use Natural Gas to create 1000 metric tons of Ammonia / day Problem Natural Gas is contaminated with H 2 S, CO 2, and C 2 H 6 Process Objectives ◇ Design a process and size major equipment to meet the specifications ◆ Clean enough Natural Gas to create 1000 metric tons of Ammonia per day ◆ Sweet Gas must have less than 10 ppm H 2 S and less than 10 ppm CO 2
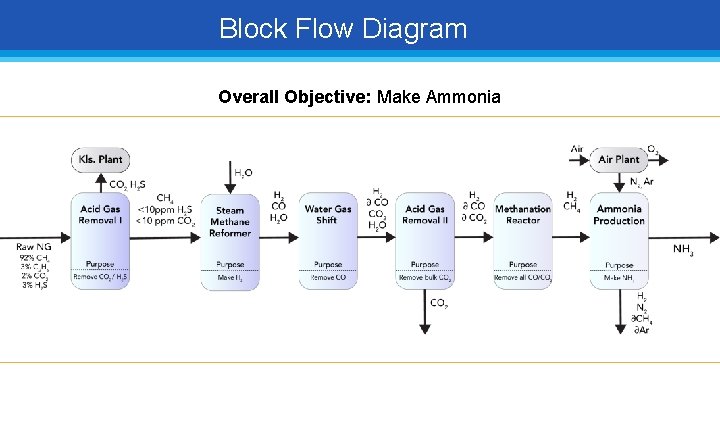
Block Flow Diagram Overall Objective: Make Ammonia
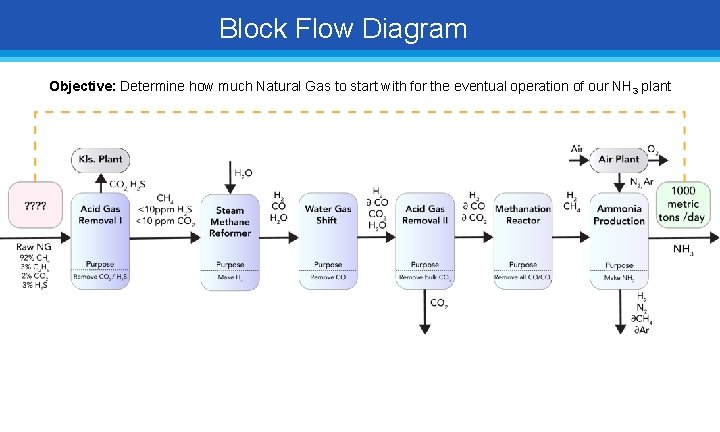
Block Flow Diagram Objective: Determine how much Natural Gas to start with for the eventual operation of our NH 3 plant
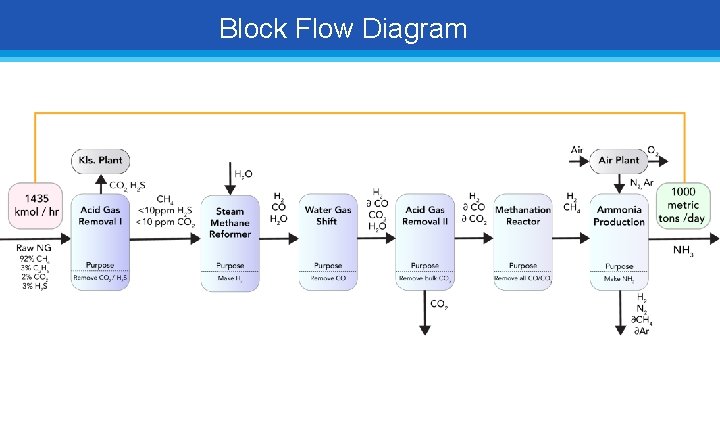
Block Flow Diagram
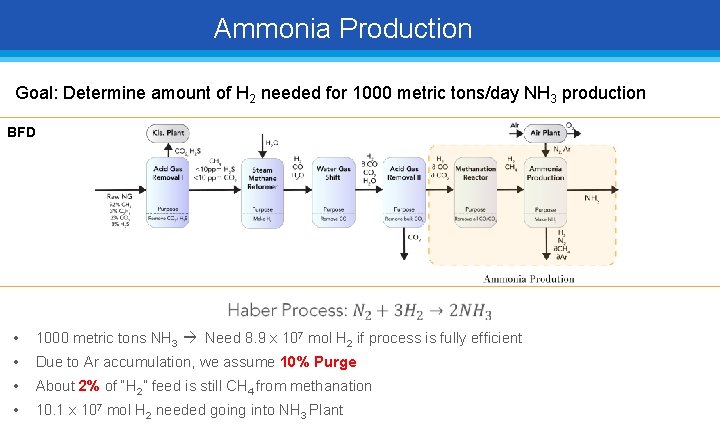
Ammonia Production Goal: Determine amount of H 2 needed for 1000 metric tons/day NH 3 production BFD • 1000 metric tons NH 3 Need 8. 9 x 107 mol H 2 if process is fully efficient • Due to Ar accumulation, we assume 10% Purge • About 2% of “H 2” feed is still CH 4 from methanation • 10. 1 x 107 mol H 2 needed going into NH 3 Plant
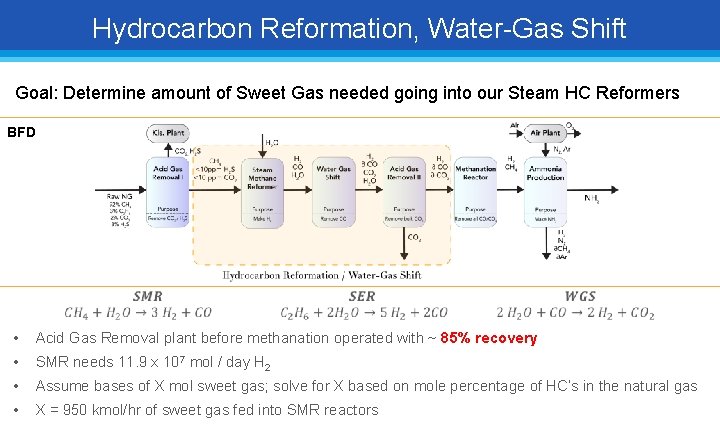
Hydrocarbon Reformation, Water-Gas Shift Goal: Determine amount of Sweet Gas needed going into our Steam HC Reformers BFD • Acid Gas Removal plant before methanation operated with ~ 85% recovery • SMR needs 11. 9 x 107 mol / day H 2 • Assume bases of X mol sweet gas; solve for X based on mole percentage of HC’s in the natural gas • X = 950 kmol/hr of sweet gas fed into SMR reactors
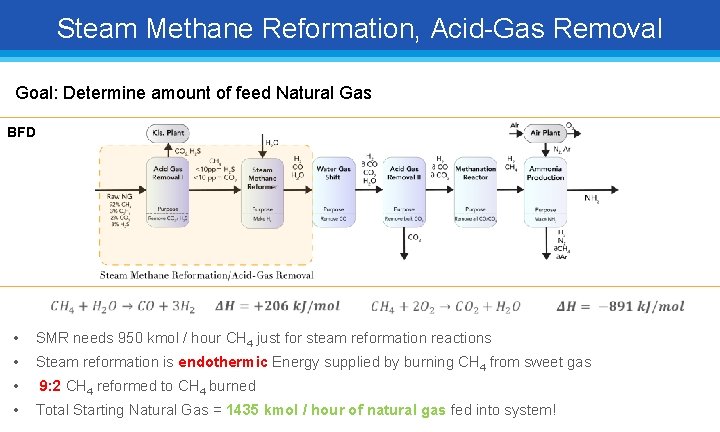
Steam Methane Reformation, Acid-Gas Removal Goal: Determine amount of feed Natural Gas BFD • SMR needs 950 kmol / hour CH 4 just for steam reformation reactions • Steam reformation is endothermic Energy supplied by burning CH 4 from sweet gas • 9: 2 CH 4 reformed to CH 4 burned • Total Starting Natural Gas = 1435 kmol / hour of natural gas fed into system!
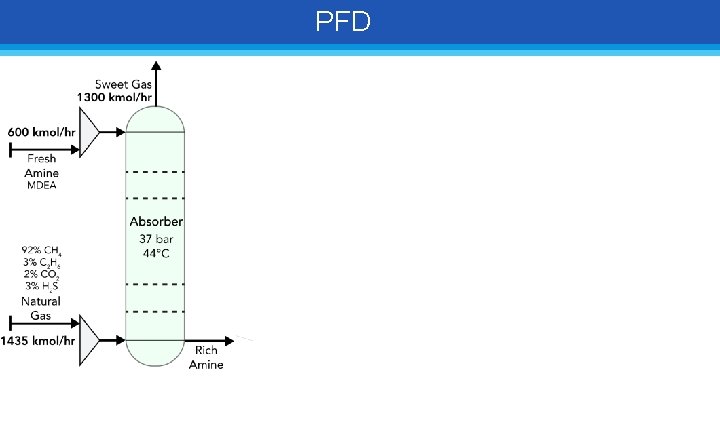
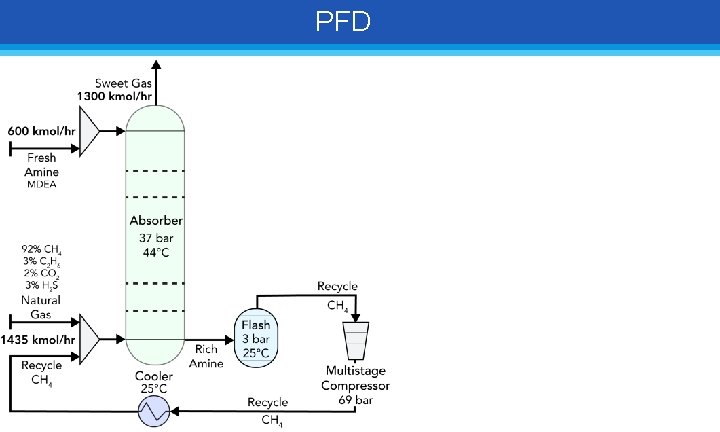
PFD
PFD
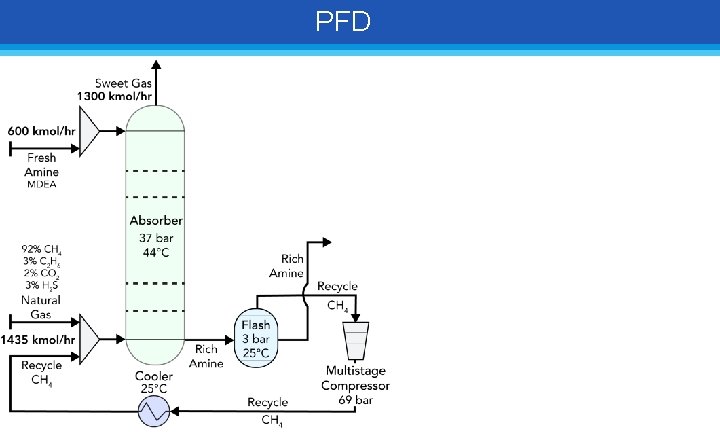
PFD
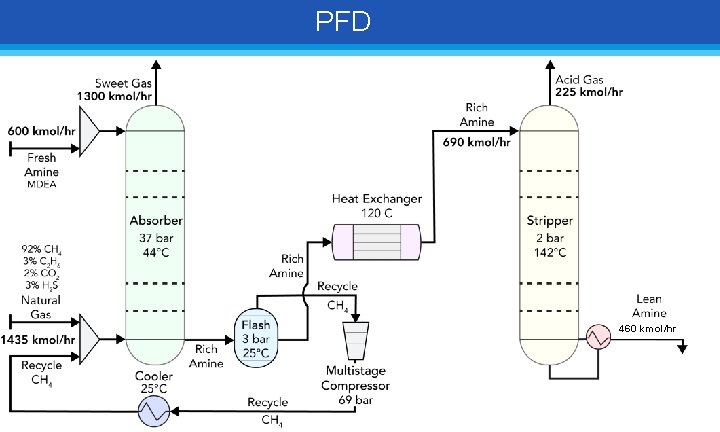
PFD 460 kmol/hr
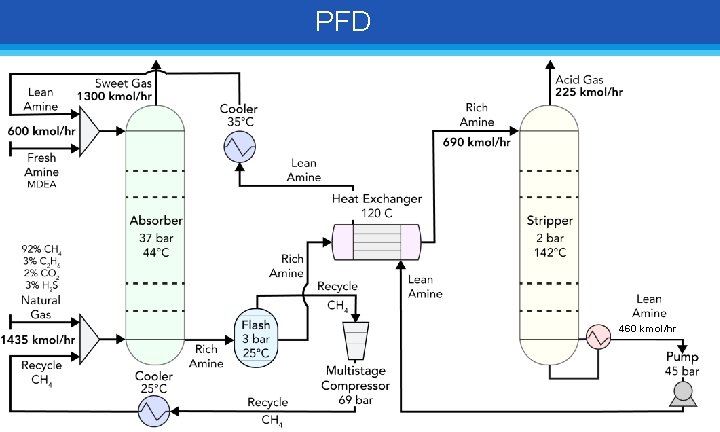
PFD 460 kmol/hr
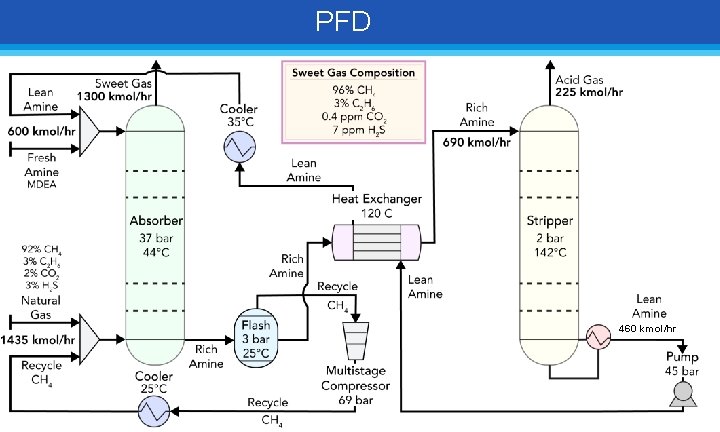
PFD 460 kmol/hr
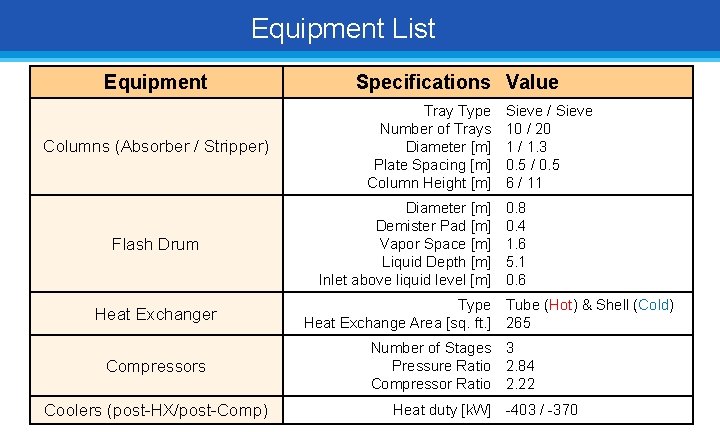
Equipment List Equipment Columns (Absorber / Stripper) Flash Drum Heat Exchanger Compressors Coolers (post-HX/post-Comp) Specifications Value Tray Type Number of Trays Diameter [m] Plate Spacing [m] Column Height [m] Diameter [m] Demister Pad [m] Vapor Space [m] Liquid Depth [m] Inlet above liquid level [m] Sieve / Sieve 10 / 20 1 / 1. 3 0. 5 / 0. 5 6 / 11 0. 8 0. 4 1. 6 5. 1 0. 6 Type Tube (Hot) & Shell (Cold) Heat Exchange Area [sq. ft. ] 265 Number of Stages 3 Pressure Ratio 2. 84 Compressor Ratio 2. 22 Heat duty [k. W] -403 / -370

Technical Challenges Convergence ◇ Build up columns from smaller tray numbers ◇ Iterations in open loop before closing recycle Aspen Methods ◇ Needed to account for binary interactions

Alternatives Methane from Flash ◇ Current: Recycle back to Absorber ◇ Alternatives: ◆ Combine with sweet gas and send forward in process ◆ Burn it Solvent Selection ◇ Current: Chemical Solvent - MDEA ◇ Alternatives: ◆ Physical Solvents (Methanol, Rectisol Process) ◆ Selectivity of solvent for H 2 S (composition specific) Solvent Recycle ◇ Possibility of reusing solvent without cleaning it (not economically viable)
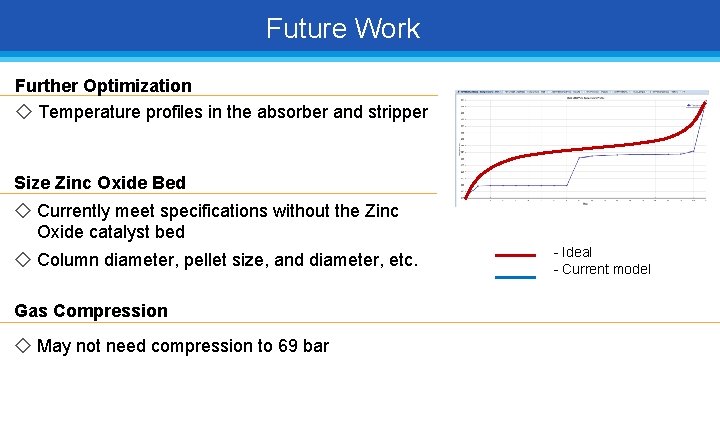
Future Work Further Optimization ◇ Temperature profiles in the absorber and stripper Size Zinc Oxide Bed ◇ Currently meet specifications without the Zinc Oxide catalyst bed ◇ Column diameter, pellet size, and diameter, etc. Gas Compression ◇ May not need compression to 69 bar - Ideal - Current model

Questions
- Slides: 21