Professor Martin Tobin Leicester Precision Medicine Institute LPMI

Professor Martin Tobin Leicester Precision Medicine Institute LPMI general information: Tel: 0116 373 6498 email: LPMI@leicester. ac. uk https: //www 2. le. ac. uk/institutes/lpmi Leicester’s Research Live – celebrating our success

LPMI Team • Martin Tobin, Director - martin. tobin@leicester. ac. uk • Jacqui Shaw, Scientific Director - js 39@leicester. ac. uk • Mike Sullivan, Commercial Director (starting May 2018) • Ruth Barber, Operations Manager - rcb 6@leicester. ac. uk • Riddhi Shukla, Business Development Manager – rys 18@leicester. ac. uk • Carl Edwards, Business Development and Technology Transfer Manager ce 133@leicester. ac. uk • Rebecca Ritchie, Administrator - rv 40@leicester. ac. uk • LPMI general information: Tel: 0116 373 6498 email: LPMI@leicester. ac. uk
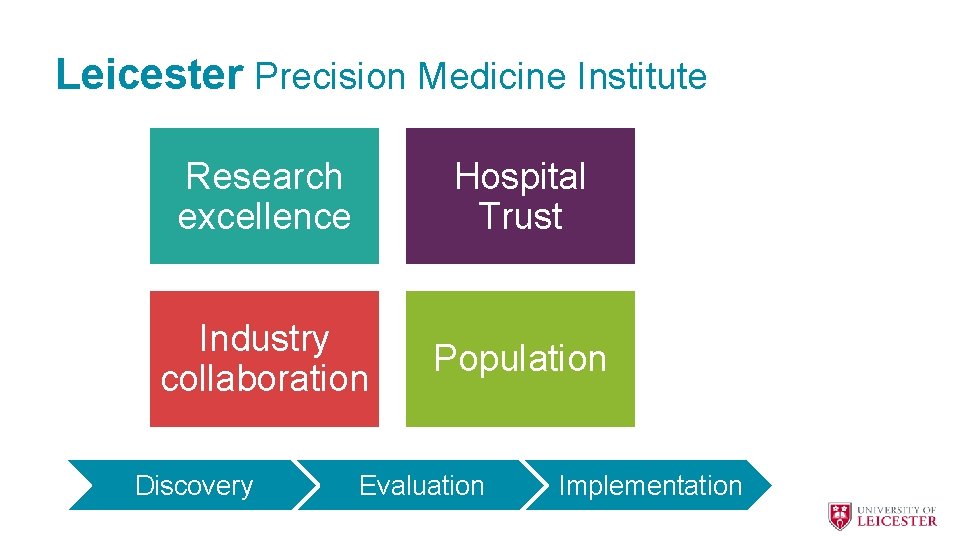
Leicester Precision Medicine Institute Research excellence Hospital Trust Industry collaboration Population Discovery Evaluation Implementation

Flagship projects • Mesothelioma Stratified Therapy (Mi. ST): a multi-arm parallel phase II umbrella trial to evaluate novel targeted therapy in malignant pleural mesothelioma (Professor Dean Fennell) • MRC Molecular Pathology Node in Breathomics (EMBER, Professor Chris Brightling) • UK Biobank study on Biological Ageing (Professor Sir Nilesh Samani) • (Professor Jacqui Shaw) Cancer diagnostics utilising circulating free DNA • Precision Medicine Cohorts (e. g. Gen. Vasc, EXCEED, COPD cohorts)

Am I doing precision medicine? The LPMI definition of Precision Medicine “right drug/treatment, right patient, right time” refers to: • The tailoring or matching of healthcare interventions to particular characteristics of patients. • Targeting interventions at those most likely to benefit. • The classification of individuals into subpopulations that differ in their disease susceptibility, prognosis, or response to individual treatments or care pathways. If your research involves either tailoring treatments to subpopulations or identifying those most likely to benefit from existing treatments, then you are probably doing precision medicine research.

Benefits of engagement with LPMI • Profile of your research • Support for strategic funding applications & developing industry partnerships • Ph. D studentships • Fellowships (supplementing WTISSF) • Capacity-building funding • Events • Support for sandpit events

LMPI support for Ph. D studentships for Autumn 2018 • Precision Medicine projects are a priority area for College of Life Sciences funded studentships: • LPMI/BRC priority studentships: 1 BRC Precision Medicine theme, 2 projects BRC Respiratory theme, 1 BRC Lifestyle theme, 2 BRC Cardiovascular theme • LMPI/Leicester Cancer Research Centre: 2 projects in Cancer precision medicine • LPMI/Informatics: studentship(s) for precision medicine health data research starting 2018 or 2019

Wellcome Trust ISSF fellows • Claire Lawson: cardiovascular risk factor trends and aetiological mechanisms in BME groups leading to Heart Failure. • Ranjit Arnold: role of CMR perfusion imaging in diagnosing and risk stratifying patients with suspected coronary disease. • Joshua Vande Hey: air quality and health, environmental resources for cohort studies. • Chiara Batini: genetic architecture of smoking behaviour in individuals of European, African and Asian ancestries using UK Biobank data. Plus – two Health Data Research UK fellows, Chiara Batini and Tim Beck

LMPI Capacity Building Call 2017 -18 funded projects • Cardiovascular magnetic resonance to individualise management in patients with suspected non-ST elevation acute coronary syndromes - Ranjit Arnold • Lifestyle and trimethylamine N-oxide: Unravelling the potential for personalised therapy - Dr Thomas Yates, Prof Toru Suzuki, Prof Don Jones & Prof Gerry Mc. Cann • Haematological malignancy panel based sequencing for clinical translation - Dr Caroline Cowley, Prof Jacqui Shaw, Prof Catrin Pritchard, Prof Martin Dyer. In collaboration with Dr Linda Barton, Consultant Haematologist & Lara Cresswell FRCPath, Clinical Lead for Molecular Diagnostics, UHL. • A Precision Medicine Cohort goes Multi-ethnic: enhancing medicines discovery in South Asians using EXCEED - Dr Catherine John, Dr Nicola Reeve, Dr Chiara Batini & Prof. Louise Wain • plus two partially funded projects • Total value of funded projects £ 148, 000

LMPI Call 2018…coming soon…… • Call Opening: Early May 2018 • Submission Deadline: Early June 2018 • Project Decision Meeting: Late June 2018 • Project start date: 1 st August 2018 • Project end date: 31 st July 2019 More information about call priority areas and confirmation of dates will be available shortly……. • To contact LPMI please drop an email to LPMI@Leicester. ac. uk

Updates • Successful annual review – research awards £ 21. 7 m in 2016/17 – growth in industry partnerships • 2017 -18 successes so far include: – new industry partnerships – Confidence in Concept, Proximity to Discovery Awards (MRC) – Midlands Health Data Research-UK bid • Plans: – LPMI Executive Committee developing Roadmap – Accelerate ethnic health research – Develop strategic industry partnerships

Questions
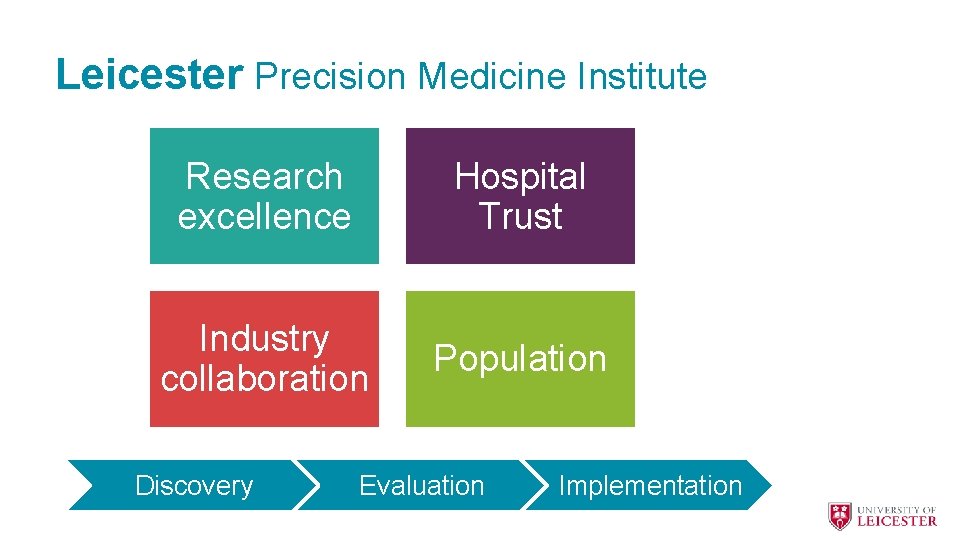
Leicester Precision Medicine Institute Research excellence Hospital Trust Industry collaboration Population Discovery Evaluation Implementation
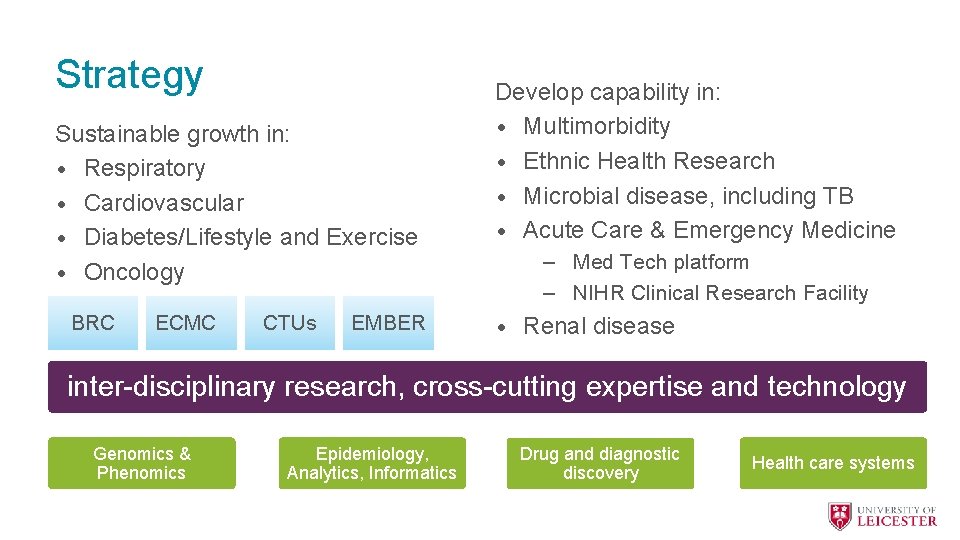
Strategy Sustainable growth in: • Respiratory • Cardiovascular • Diabetes/Lifestyle and Exercise • Oncology BRC ECMC CTUs EMBER Develop capability in: • Multimorbidity • Ethnic Health Research • Microbial disease, including TB • Acute Care & Emergency Medicine – Med Tech platform – NIHR Clinical Research Facility • Renal disease inter-disciplinary research, cross-cutting expertise and technology Genomics & Phenomics Epidemiology, Analytics, Informatics Drug and diagnostic discovery Health care systems
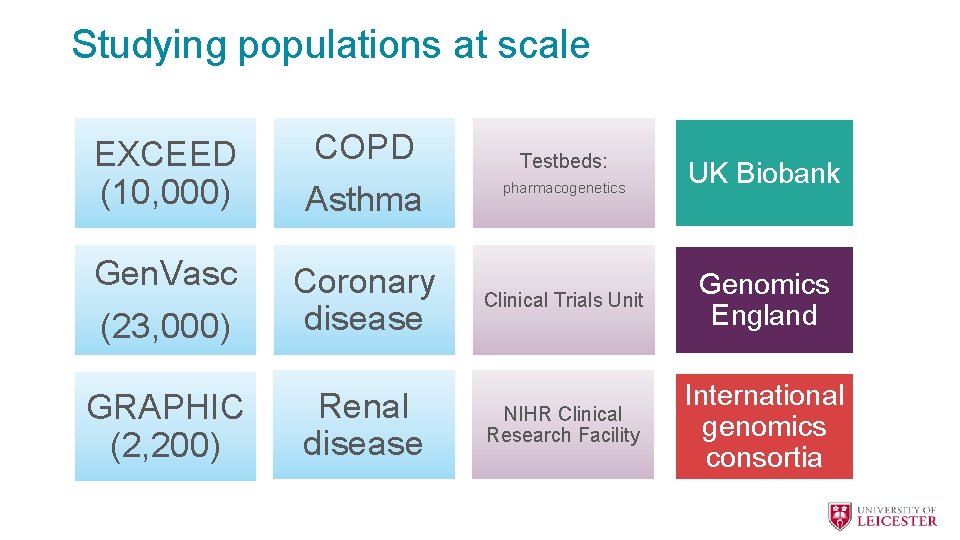
Studying populations at scale EXCEED (10, 000) COPD Asthma Gen. Vasc (23, 000) Coronary disease GRAPHIC (2, 200) Renal disease Testbeds: pharmacogenetics UK Biobank Clinical Trials Unit Genomics England NIHR Clinical Research Facility International genomics consortia
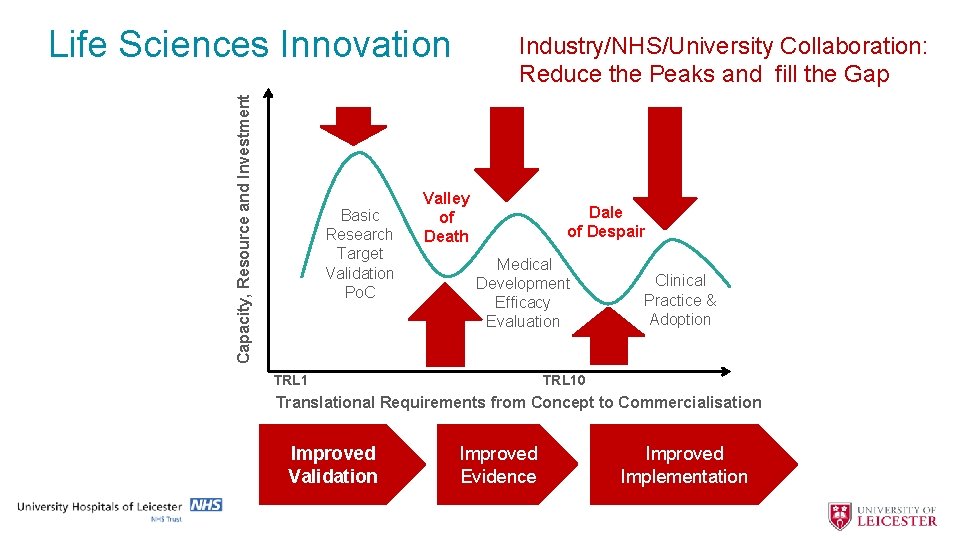
Capacity, Resource and Investment Life Sciences Innovation Basic Research Target Validation Po. C Industry/NHS/University Collaboration: Reduce the Peaks and fill the Gap Valley of Death Dale of Despair Medical Development Efficacy Evaluation TRL 1 Clinical Practice & Adoption TRL 10 Translational Requirements from Concept to Commercialisation Improved Validation Improved Evidence Improved Implementation
- Slides: 16