Prof Mehdi Hasan Mumtaz ACIDBASE BALANCE Terminology Physiologic

Prof. Mehdi Hasan Mumtaz

ACID-BASE BALANCE Terminology. Physiologic Compensation By Body. Pathophysiologic Disturbances. Practical Approach To Assessment. Biochemical Reports & Case Histories.

DEFINITION OF TERMINOLOGY ACID - STANDARD BICARBONATE. BASE - BUFFER BASE & BASE DEFICIT. ALKALI BUFFERING & BUFFER. PH. 24 x PCO 2 (mm. Hg) H+(nmol/L)=- --------------HCO 3 (meq/L) (40 nmol/L)

PRODUCT OF METABOLISM H++ - Anaerobic Metabolism. CO 2 - Aerobic Metabolism.

PHYSIOLOGIC COMPENSATION HYDROGEN IONS. » Incoporation in water. H++HCO 3 H 2 C 3 O CO 2 + H 2 O. » Loss from body. • Kidney – regeneration of HCO 3. » Intestine. CO 2. » Chemoreceptors in hypothalamus. HCO 3. » HCO 3 generation by erythrocytes. » HCO 3 re-absorption in renal tubules. » HCO 3 generation in renal tubules.
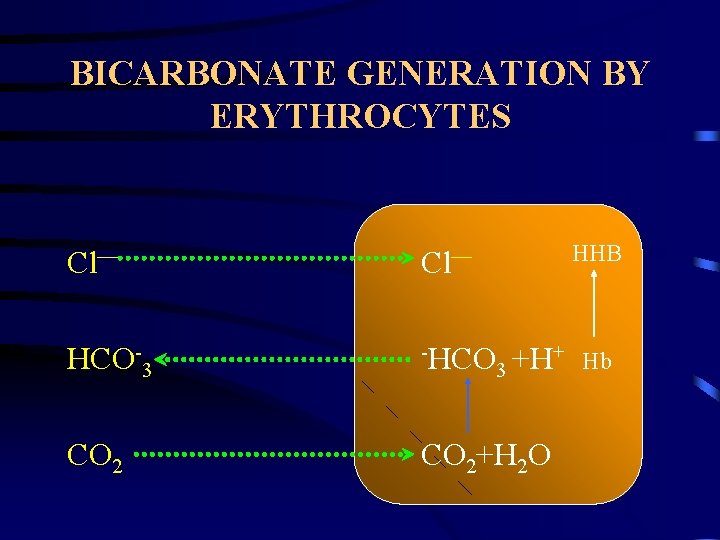
BICARBONATE GENERATION BY ERYTHROCYTES HHB Cl— HCO-3 -HCO CO 2+H 2 O + +H 3 Hb
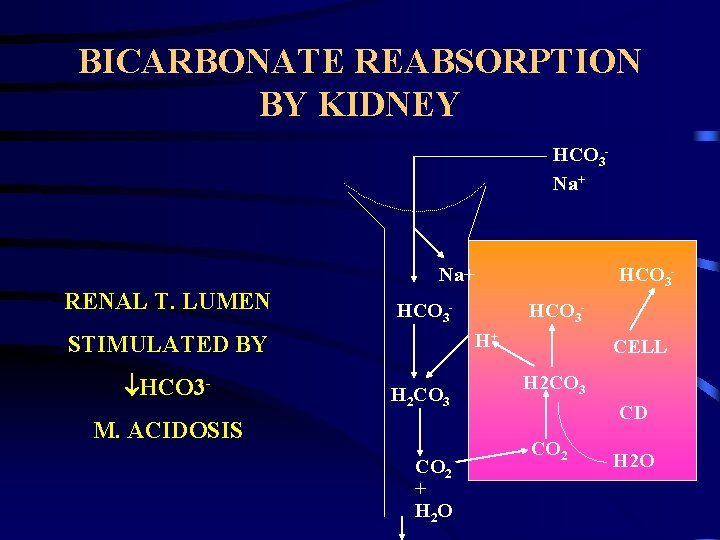
BICARBONATE REABSORPTION BY KIDNEY HCO 3 Na+ RENAL T. LUMEN HCO 3 - HCO 3 H+ STIMULATED BY HCO 3 - H 2 CO 3 M. ACIDOSIS CO 2 + H 2 O CELL H 2 CO 3 CD CO 2 H 2 O
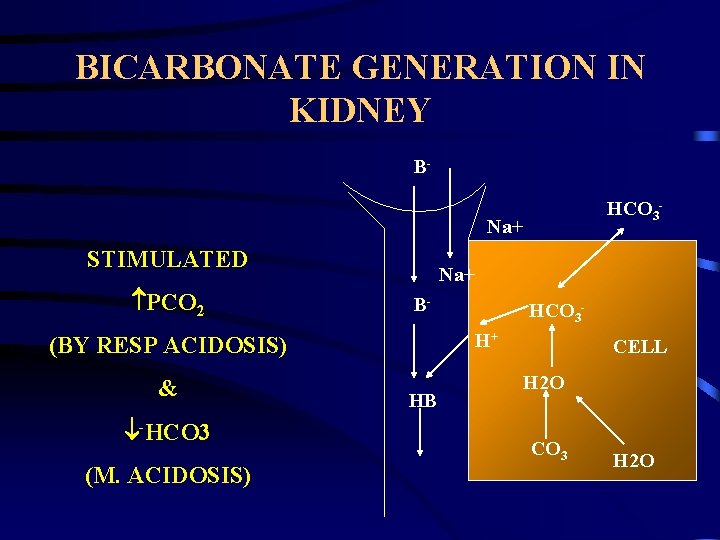
BICARBONATE GENERATION IN KIDNEY BHCO 3 - Na+ STIMULATED PCO 2 Na+ BH+ (BY RESP ACIDOSIS) & -HCO 3 (M. ACIDOSIS) HCO 3 - HB CELL H 2 O CO 3 H 2 O

PATHOPHYSIOLOGIC DISTURBANCES Lungs Disturbances of CO 2= R. Centre Disturbance of H++HCO 3 = Metabolic

Henderson - Hosselbalch EQUATION Proton Acceptor (Base) PH=PK+Log = ----------------Proton Donor (Acid) -HCO (Metabolic) PH=PK+Log = -----------------H 2 CO 3 or PCO 2 x 0. 03 (Respiratory) 3
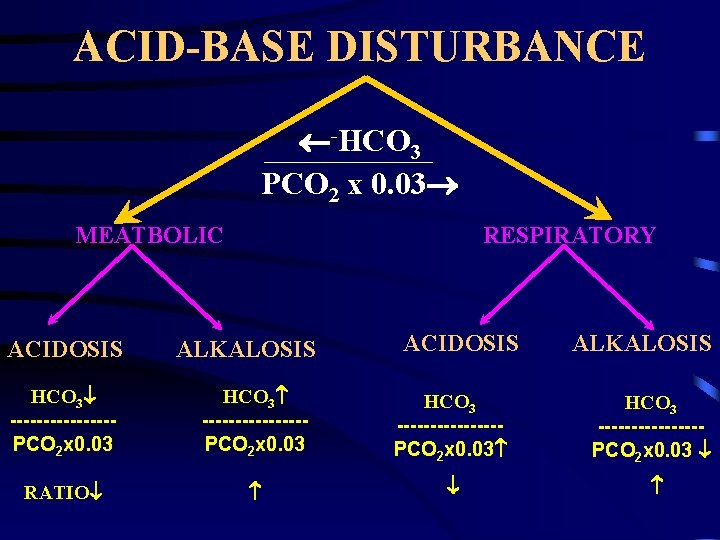
ACID-BASE DISTURBANCE -HCO 3 PCO 2 x 0. 03 MEATBOLIC ACIDOSIS RESPIRATORY ALKALOSIS ACIDOSIS HCO 3 ---------------PCO 2 x 0. 03 HCO 3 --------PCO 2 x 0. 03 RATIO ALKALOSIS HCO 3 --------PCO 2 x 0. 03

Defect Correction Metabolic acidosis = HCO 3 ---------PCO 2 Respiratory acidosis = HCO 3 -----PCO 2 HCO 3 -----PCO 2 Metabolic alkalosis = HCO 3 ---------PCO 2 Respiratory alkalosis = HCO 3 -----PCO 2 HCO 3 -----PCO 2

CAUSES OF M. ACIDOSIS 1. 2. 3. 4. 5. 6. 7. 8. 9. Glomeralar failure. Hyperkalamic M. Acidosis Keto-acidosis. Lactic acidosis. Variable Intestinal loss. R. Tubular failure. Actazolamide therapy. Hyppkalamic Acidosis R. Tubular acidosis. Hyperchloraemic Acidosis Ureteric transplantation. NH 4 Cl el therapy.

CAUSES M. ACIDOSIS A. HYPERKALAEMIC M. ACIDOSIS 1, GLOMERULAR FAILURE 2, KETOACIDOSIS 3, LACTIC ACIDOSIS

CAUSES OF M. ACIDOSIS B. HYPOKALAEMIC M. ACIDOSIS 1. RENAL TUBULAR 2. ACETAZOLAMIDE THERAPY 3. RENAL TUBULAR ACIDOSIS

CAUSES OF M. ACIDOSIS C. HYPERCHLORAEMIC ACIDOSIS 1. ACATAZOLAMIDE THERAPY 2. RENAL TUBULAR ACIDOSIS 3. URETERIC TRASPLANTATION 4. AMMONIUM CHLORIDE

CAUSES OF M. ACIDOSIS D. HYPERCHLORAEMIC HYPOKALAEMIC M. ACIDOSIS 1. ACETAZOLAMIDE THERAPY 2. RENAL TUBULAR ACIDOSIS

SCREENING TESTS METABOLIC ACIDOSIS BLOOD GLUCOSE. URINE/ BLOOD KETONES. SERUM CHLORIDE. SERUM POTASSIUM

RESPIRATORY ACIDOSIS Acute Respiratory Failure. » Erythrocyte Chronic Respiratory Failure. » Renal Generation.

METABOLIC ALKALOSIS Administration of HCO 3. K+ depletion – Generation by kidney. Pyloric Stenosis.
RESPIRATORY ALKALOSIS Hysterical Over-breathing. ICP. Brain Stem Injury. Hypoxia. Pulmonary Oedema. Lobar Pneumonia. Pulmonary Collapse. Excessive Artificial Ventilation.

BALANCE OF ACID-BASE NORMAL VALUES PCO 2 » 30 -50 mm. Hg or 4 -6. 6 k. Pa. » >50 mm. Hg respiratory or 6. 6 k. Pa acidosis » <30 mm. Hg respiratory or 4 k. Pa alkalosis PH » 7. 30 – 7. 50 » >7. 50 alkalaemia. » <7. 30 acidosis

BALANCE OF ACID-BASE RELATIONSHIP PCO 2 and PH. PCO 2 & ventilation. PO 2 and normal range. PO 2 and FIO 2. PCO 2, and temperature.

TERMINOLOGY ACIDAEMIA - PH<7. 30 ALKAEMIA - PH>7. 50. ACIDOSIS - Base Deficit Present. ALKALOSIS - Base Excess Present.

HOW TO ASSESS BLOOD GASES? STEP-1 Assessment of Acid-Base Balance. STEP-2 Assessment of Hypoxaemic State. STEP-3 Assessment of Tissue Oxygenation State.
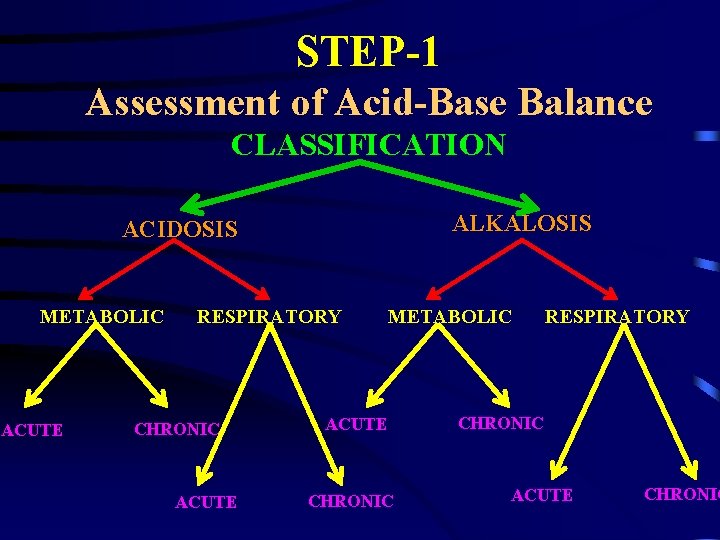
STEP-1 Assessment of Acid-Base Balance CLASSIFICATION ALKALOSIS ACIDOSIS METABOLIC ACUTE RESPIRATORY CHRONIC ACUTE METABOLIC ACUTE CHRONIC RESPIRATORY CHRONIC ACUTE CHRONIC

STEP-1 Assessment of Acid-Base Balance Acute Chronic - Uncompensated. - Compensated. -Fully. - Partially. COMPENSATED PH 7. 30 -7. 50 DIAGNOSIS.

DIAGNOSIS SEQUENCE. PH. PCO 2. HCO 3. PH Normal 7. 4 Compensated 7. 3 -7. 5 PCO 3 Normal 40 mm. Hg (5. 3 k. Pa) Compensated 30 -50 mm. Hg (4 -6. 6 k. Pa)
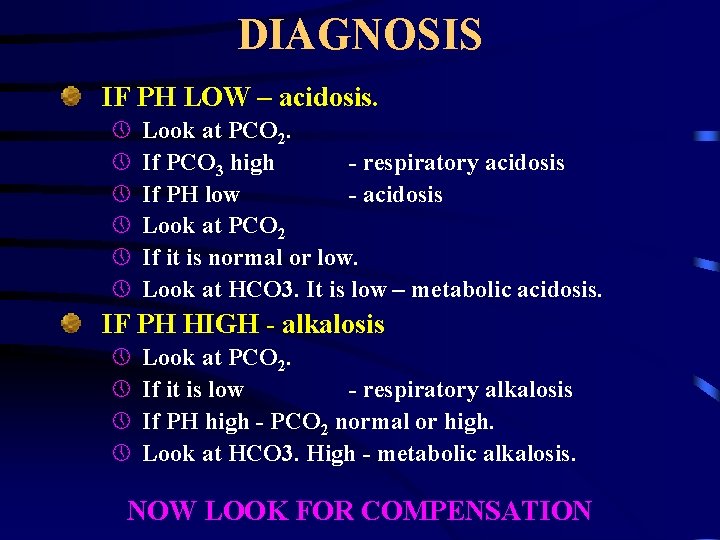
DIAGNOSIS IF PH LOW – acidosis. » » » Look at PCO 2. If PCO 3 high - respiratory acidosis If PH low - acidosis Look at PCO 2 If it is normal or low. Look at HCO 3. It is low – metabolic acidosis. IF PH HIGH - alkalosis » » Look at PCO 2. If it is low - respiratory alkalosis If PH high - PCO 2 normal or high. Look at HCO 3. High - metabolic alkalosis. NOW LOOK FOR COMPENSATION
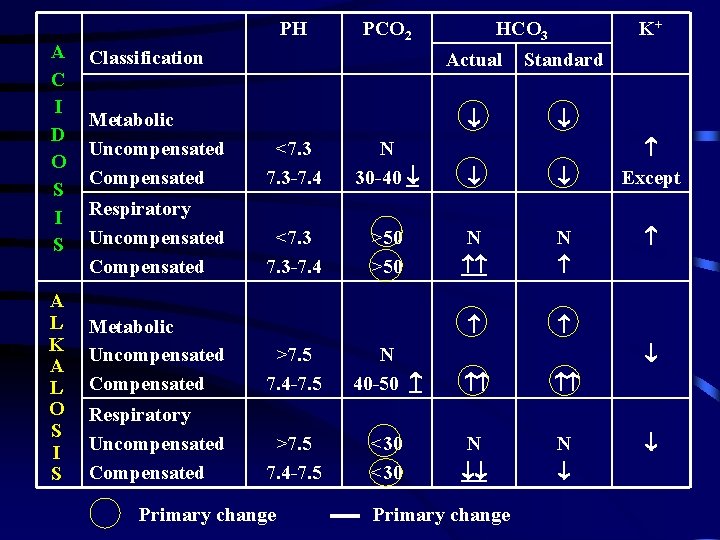
PH A C I D O S I S A L K A L O S I S PCO 2 Classification HCO 3 K+ Actual Standard Metabolic Uncompensated Compensated <7. 3 -7. 4 N 30 -40 Respiratory Uncompensated Compensated <7. 3 -7. 4 >50 N N Metabolic Uncompensated Compensated >7. 5 7. 4 -7. 5 N 40 -50 Respiratory Uncompensated Compensated >7. 5 7. 4 -7. 5 <30 N N Primary change Except Primary change

PRACTICAL EXAMPLES PH 7. 26 7. 38 7. 20 7. 36 7. 60 7. 56 PCO 2 56 76 40 25 25 44 HCO 3 24 24 9 15 24 38

STEP-2 Hypoxaemic State Below 60 years of age: » Normal PO 2 = 97 mm. Hg. (13 Kpa) » Acceptable range = >80 m. Hg. (10. 6 Kpa) » Mild hypoxiaemia = <80 mm. Hg. (10. 6 Kpa) » Moderate hypoxiaemia = <60 mm. Hg. (8) » Severe hypoxiaemia = <40 mm. Hg. (5. 3)

STEP-2 Hypoxaemic State Above 60 years of age: » Subtract 1 mm. Hg from minimal 80 mm. Hg for every year over 60; this means acceptable range: • • 60 years = >80 mm. Hg. =>10. 6 Kpa 70 years = >70 mm. Hg. => 9. 3 Kpa 80 years = >60 mm. Hg. = > 8. 0 Kpa 90 years = >50 mm. Hg. = > 6. 6 Kpa New Born: » Acceptable = 40 -70 mm. Hg. = 5. 3 -9. 3 Kpa

STEP-2 Hypoxaemic State Oxygen Therapy FIO 2 x 5 = Expected PO 2. Uncorrected Hypoxaemia = PO 2<Room Air Acceptable Limit. Corrected Hypoxaemia = PO 2 > Room Air Acceptable Limit. <100 mm. Hg. =13. 3 Kpa Excessively Corrected Hypoxaemia = PO 2>100 mm. Hg(13. 3 Kpa) < minimal predicted.

OXYGENATION STATUS FIO 2 ----PIO 2 PAO 2 = PIO 2 -PACO 2 Pa. O 2 P(A-a)O 2=PAO 2 -Pa. O 2/PAO 2=0. 67 ---------------------------DO 2 520— 720 ml/min/m 2 VO 2 110— 160 ml/min/m 2 Lactate
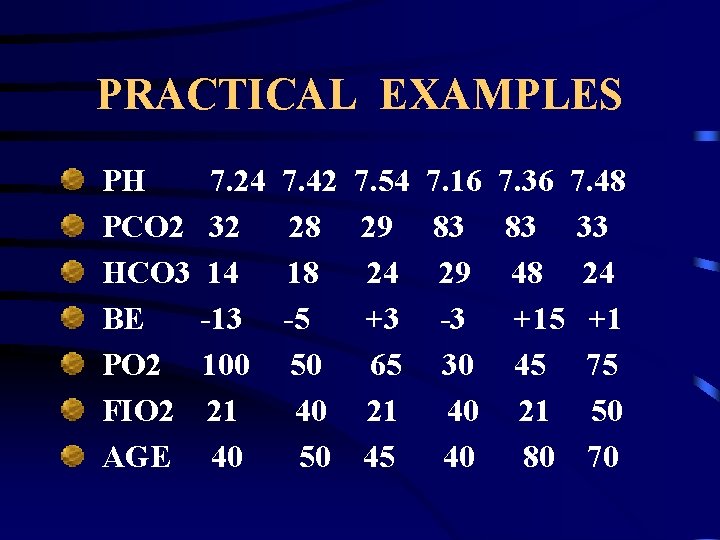
PRACTICAL EXAMPLES PH PCO 2 HCO 3 BE PO 2 FIO 2 AGE 7. 24 32 14 -13 100 21 40 7. 42 28 18 -5 50 40 50 7. 54 29 24 +3 65 21 45 7. 16 7. 36 7. 48 83 83 33 29 48 24 -3 +15 +1 30 45 75 40 21 50 40 80 70
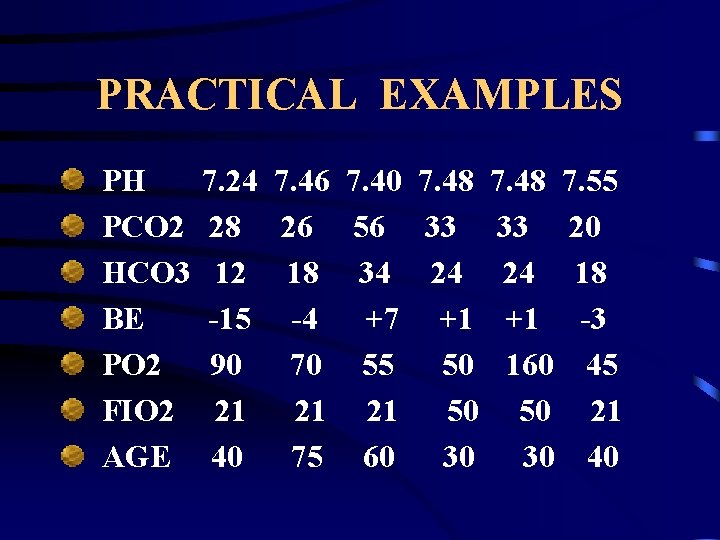
PRACTICAL EXAMPLES PH PCO 2 HCO 3 BE PO 2 FIO 2 AGE 7. 24 7. 46 7. 40 7. 48 7. 55 28 26 56 33 33 20 12 18 34 24 24 18 -15 -4 +7 +1 +1 -3 90 70 55 50 160 45 21 21 21 50 50 21 40 75 60 30 30 40

STEP-3 Assessment of Tissue Oxygenation 1. Cardiac Status. 2. Peripheral Perfusion Status. 3. Blood Oxygen Transport Mechanism. Depends on: » Vital Signs » Physical Examination.
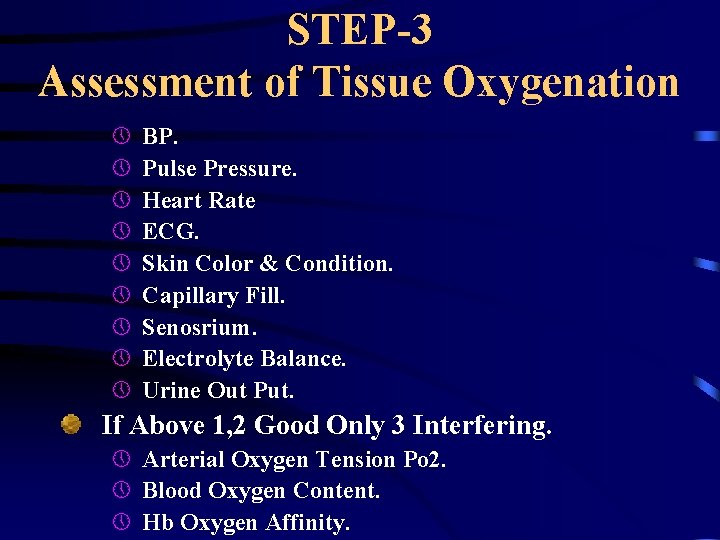
STEP-3 Assessment of Tissue Oxygenation » » » » » BP. Pulse Pressure. Heart Rate ECG. Skin Color & Condition. Capillary Fill. Senosrium. Electrolyte Balance. Urine Out Put. If Above 1, 2 Good Only 3 Interfering. » Arterial Oxygen Tension Po 2. » Blood Oxygen Content. » Hb Oxygen Affinity.

SUMMARY ASSESS ACID/BASE STATUS. ASSESS HYPOXAEMIC STATE ASSESS TISSUE OXYGENATION. TRY TO FIND OUT THE CAUSE. SEE FOR THE NEED OF HCO 3.

SUMMARY Acidosis Metabolic Look at 1. Blood urea If and K+ G. F. 2. Blood Glucose/ ketones If and K+ ketoacidosis. 3. PO 2 If K+ Lactic acidosis 4. Serum HCO 3. If only H/o Therapy 5. If K+ think of NH 4 Cl therapy + G. Transplantation 6. If Cl- K+ Think of actazolamide therapy and R. Tubul Acidosis. 7. If Cl-N K+ Proximal Tubul Failure. OTHERWISE THINK ABOUT GIT INVOLVEMENT
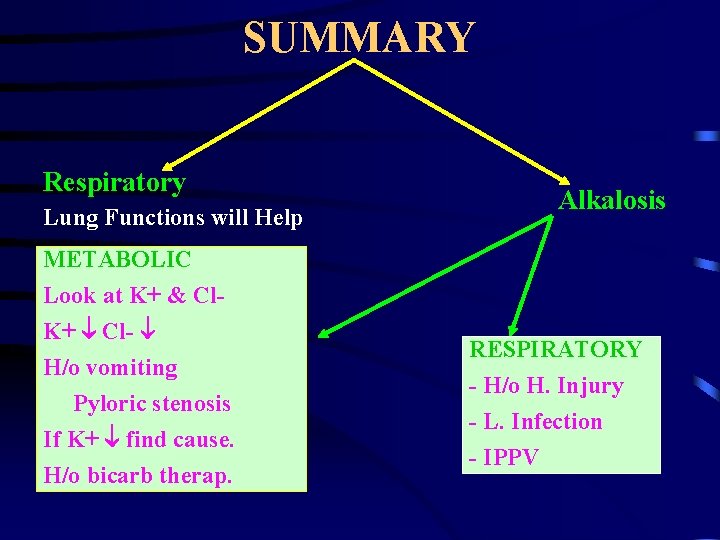
SUMMARY Respiratory Lung Functions will Help Alkalosis METABOLIC Look at K+ & Cl. K+ Cl- H/o vomiting Pyloric stenosis If K+ find cause. H/o bicarb therap. RESPIRATORY - H/o H. Injury - L. Infection - IPPV
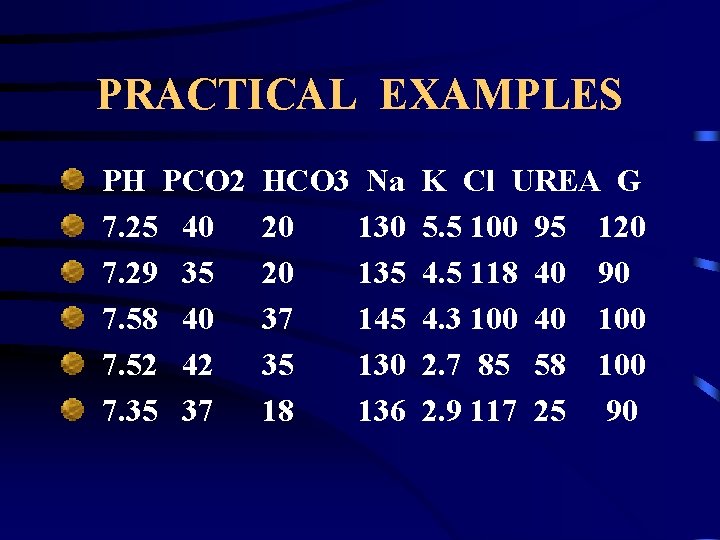
PRACTICAL EXAMPLES PH PCO 2 7. 25 40 7. 29 35 7. 58 40 7. 52 42 7. 35 37 HCO 3 Na 20 130 20 135 37 145 35 130 18 136 K Cl UREA G 5. 5 100 95 120 4. 5 118 40 90 4. 3 100 40 100 2. 7 85 58 100 2. 9 117 25 90

BUFFER BASE/BASE DEFICIT “Sum of the Buffer anions; 44 --55 meq/l PREDICTED RESPIRATORY PH? PCO 2 -- PH RELATIONSHIP PCO 2 20 mm. Hg = 0. 1 PH. PCO 2 10 mm. Hg = 0. 1 PH

BASE EXCESS/ DEFICIT Predicted Respiratory PH 1. Calculate difference between measured PCO 2 and 40 mm. Hg. Move decimal 2 places to left. 2. If PCO 2 > 40 subtract ½ difference from 7. 4. 3. If PCO 2 < 40 add the difference to 7. 40. PH 7. 21 PCO 2 90 » 90 -40 = 50 = 0. 50 x ½ = 0. 25 » 7. 40 -0. 25 =7. 15 PH 7. 47 PCO 2 18 » 40 -18 = 22= 0. 22

METABOLIC COMPONANT METABOLIC PH CHANGE = Measured PH -- Predicted Respiratory PH

DETERMINATION OF METABOLIC COMPONENT Determine predicted PH Determine difference between measured and predicted PH Move two decimal point to the right Multiply by 2/3 Base excess if measured Ph > predicted PH Base deficit if measured PH<predicted PH

EXAMPLE PH 7. 04 PCO 2 76 Predicted PH= 76 -40=36 =. 36=1/2*36 = 18=7. 4 -. 18=7. 22 Base Excess/deficit=7. 22 -7. 04=0. 18= 18*2/3=12 As measured PH is less than predicted, so it is Base deficit

Pa. CO 2/HCO 3 Relationship 1. A. Respiratory acidosis. Expected HCO 3=24+0. 1*(Paco 2 -40) 2. Chronic Respiratory acidosis. Expected HCO 3=24+0. 35*(Paco 2 -40) 3. A. Respiratory alkalosis. Expected HCO 3=24 -0. 2*(40 -Paco 2) 4. Chronic respiratory alkalosis. Expected HCO 3=24 -0. 5*(40 -Paco 2)

EXPECTED Pa. CO 2 METABOLIC ACIDOSIS Expected Pa. CO 2=1. 5*(HCO 3) + 8 METABOLIC ALKALOSIS Expected Pa. CO 2=40+0. 6*(HCO 3)
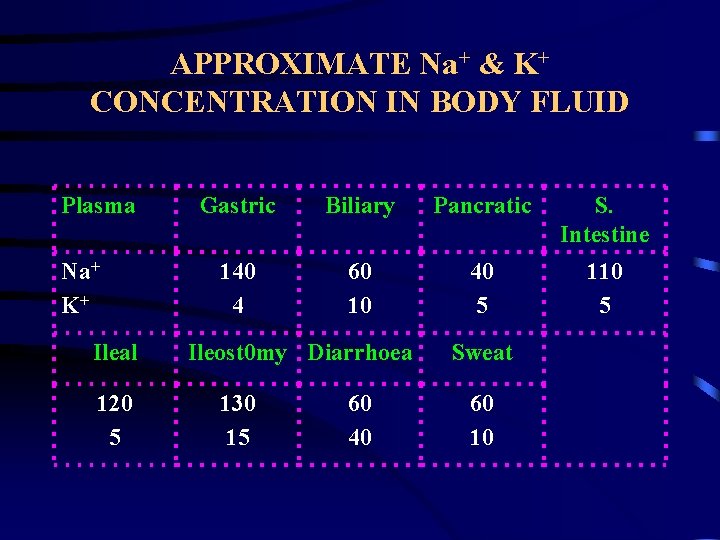
APPROXIMATE Na+ & K+ CONCENTRATION IN BODY FLUID Plasma Na+ K+ Ileal 120 5 Gastric Biliary Pancratic S. Intestine 140 4 60 10 40 5 110 5 Ileost 0 my Diarrhoea 130 15 60 40 Sweat 60 10
DOES TRADITIONAL BLOOD GAS ANALYSIS SERVES THE PURPOSE? PH PCO 2 PO 2 HCO 3

WHAT INFORMATION DOES IT GIVE? OXYGEN UPTAK CO 2 PRODUCTION ACIDITY/ ALKALINITY
WHAT INFORMATION IS REQUIRED FOR THERAPY? UPTAKE TRANSPORT capillaries. RELEASE - O 2 uptake in lungs. - from lungs to - from capillaries to tissues. HOW TO WE GET? DEEP PICTURE OF BLOOD GASES

O 2 UPTAKE MOUTH TO ALVEOLI “Grahams’ Law” of diffusion

O 2 UPTAKE Alveoli to Hb “Henrys’ Law” of diffusion

COMBINE BOTH LAWS Mouth to Alveoli Grahams’ Law of diffusion Alveoli to Hb Henrys’ Law of diffusion.

TRANSPORT TO CAPILLARIES DO 2 “ 520 - 720 ml/min/m 2 ”

O 2 RELEASE TO TISSUE VO 2 “ 110 - 160 ml/min/m 2 ”
WIHAT IS DEEP PICTURE? PCO 2. t. Hb. o. S 2. O 2 Hb. ct. O. p 50. VO 2.

O 2 TRANSPORT AMOUNT OF HB. FRACTION OF OXYGENATED HB. O 2 TENSION.

MAJOR CHALLENGES Balancing O 2 Supply and O 2 Demand

O 2 CARRYING CAPACITY 98% Bound to Hb. 2% in plasma. Forms of hemoglobins. Oxygenated – O 2 Hb. Deoxygenated – RHb. Dyshaemoglobins. » Carboxyhaemoglobin (Co. Hb). » Methaemoglobin (Met. Hb). t. Hb = c. O 2 Hb + c. RHb + c. Co. Hb + c. Met. Hb

DEGREE TO WHICH Hb CARRIES O 2 Expressed in two Different Ways. 1. Fraction of Oxygenated Hb. c. O 2 Hb = -----------------------c. O 2 Hb + c. RHb + c. Co. Hb + c. Met. Hb FRACTIONAL SATURATION 2. O 2 Saturation. c. O 2 Hb s. O 2 = -------------- X 100 c. O 2 Hb + c. RHb

DEGREE TO WHICH Hb CARRIES O 2 “FUNCTIONAL SATURATION” Relationship between Oxygenated Hb (O 2 Hb) and Oxygen Saturation s. O 2) O 2 Hb = s. O 2 x (1 -Co. Hb – c. Met. Hb)

Example: Patient exposed to carbon monoxide t. Hb = 10. 0 mmol/L c. O 2 Hb = 07. 7 mmol/L c. RHb = 0. 3 mmol/L c. Co. Hb = 2. 0 mmol/L 7. 7 mmol/L 0. 77 c. O 2 Hb = -------------- = -----(7. 7+0. 3+2. 0) mmol/L (or 77%) 7. 7 mmol/L s. O 2 = ------------- X 100 = 96. 25% (7. 7+0. 3) mmol/L
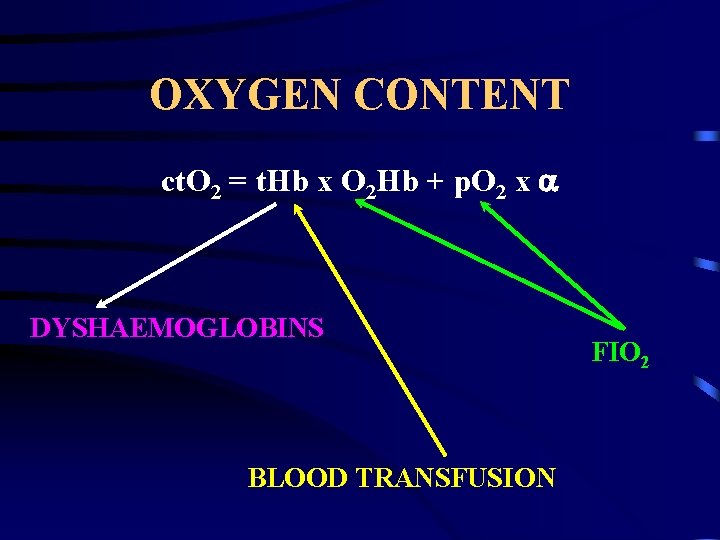
OXYGEN CONTENT ct. O 2 = t. Hb x O 2 Hb + p. O 2 x DYSHAEMOGLOBINS BLOOD TRANSFUSION FIO 2
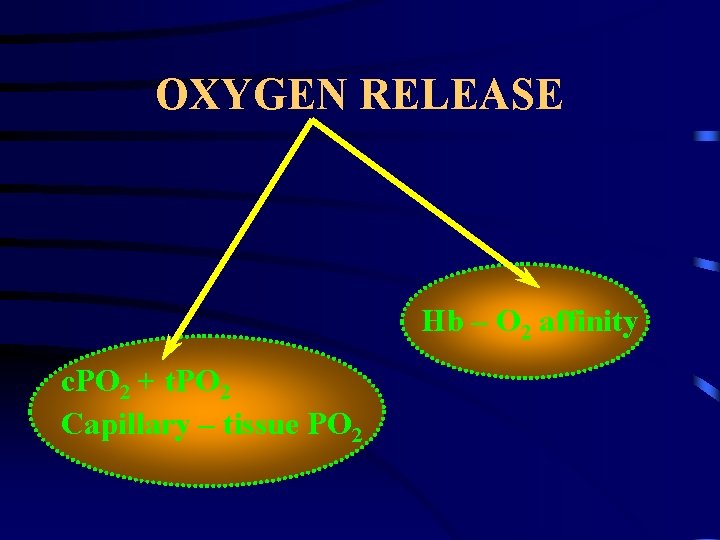
OXYGEN RELEASE Hb – O 2 affinity c. PO 2 + t. PO 2 Capillary – tissue PO 2
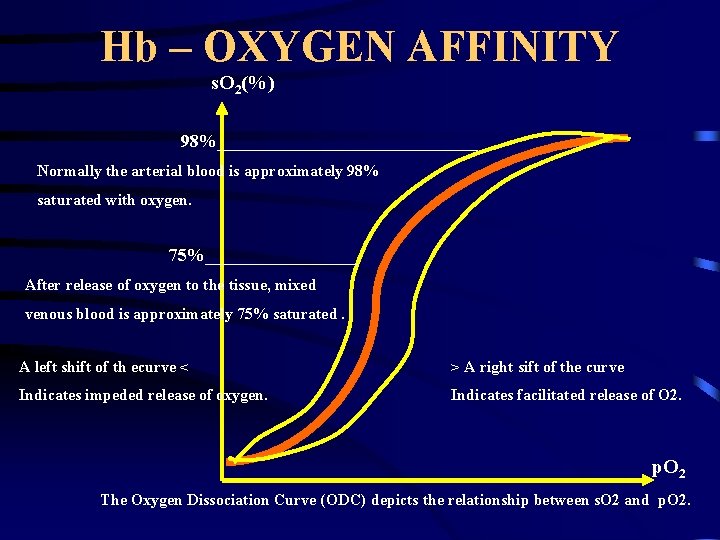
Hb – OXYGEN AFFINITY s. O 2(%) 98%_______________ Normally the arterial blood is approximately 98% saturated with oxygen. 75%_________ After release of oxygen to the tissue, mixed venous blood is approximately 75% saturated. A left shift of th ecurve < > A right sift of the curve Indicates impeded release of oxygen. Indicates facilitated release of O 2. p. O 2 The Oxygen Dissociation Curve (ODC) depicts the relationship between s. O 2 and p. O 2.
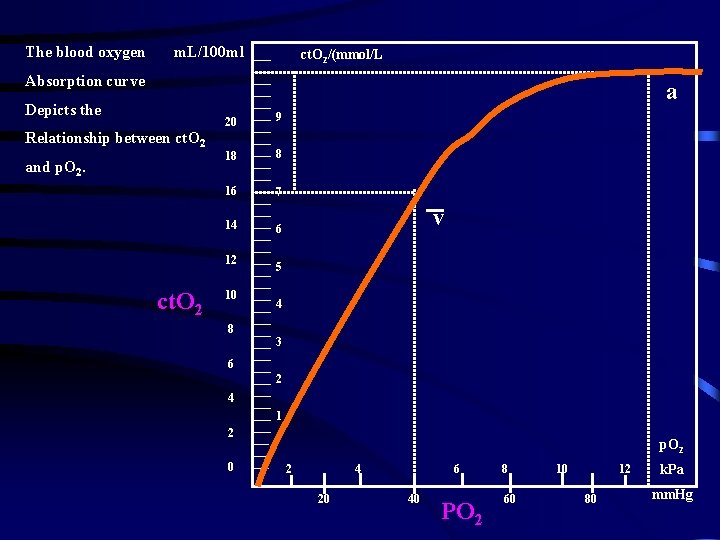
The blood oxygen m. L/100 ml ct. O 2/(mmol/L Absorption curve a Depicts the Relationship between ct. O 2 and p. O 2. 20 9 18 8 16 7 14 6 12 ct. O 2 10 8 v 5 4 3 6 2 4 1 2 0 p. O 2 2 4 20 6 40 PO 2 8 60 10 12 80 k. Pa mm. Hg

DEEP PICTURE CONTAINS INFORMATION on OXYGEN UPTAKE TRANSPORT RELEASE

Deep Picture Contains Information OXYGEN UPTAKE Pa. O 2 = 9. 2 – 15. 5 Kpa QSQT = 2 -6% Optimise Ventilation Optimise Specific Lung Disease (PAO 2 – Pa. O 2) = 5 - 15 mm. Hg

Deep Picture Contains Information RELEASE PO 2 Gradient - Optimise Ventilation - Optimise Factors O 2 Dissociation Curve
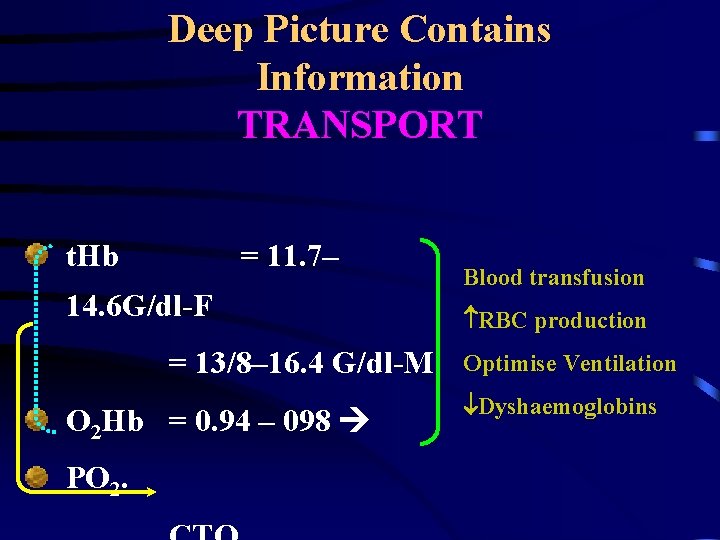
Deep Picture Contains Information TRANSPORT t. Hb = 11. 7– 14. 6 G/dl-F = 13/8– 16. 4 G/dl-M O 2 Hb = 0. 94 – 098 PO 2. Blood transfusion RBC production Optimise Ventilation Dyshaemoglobins

CLINICAL SCENARIO A 60 year old woman presents with pneumonia , shock and resp. distress. ABG; Fio 2 0. 5 PH 7. 15 PCO 2 30/4 HCO 3 10. 3 PO 2 105/14 BE -18. 3 a, Describe; Acid base status? b, Is there problem with O 2 transfer? c, Is intubation likely to be necessary?

ANSWER 1, Metabolic Acidosis +Resp. Acidosis 2, Yes, A-a gradient is raised at 218. Shunt or V/Q mismatch 3, Paco 2 is very high than expected Paco 2 in metabolic acidosis showing imminant respiratory arrest.
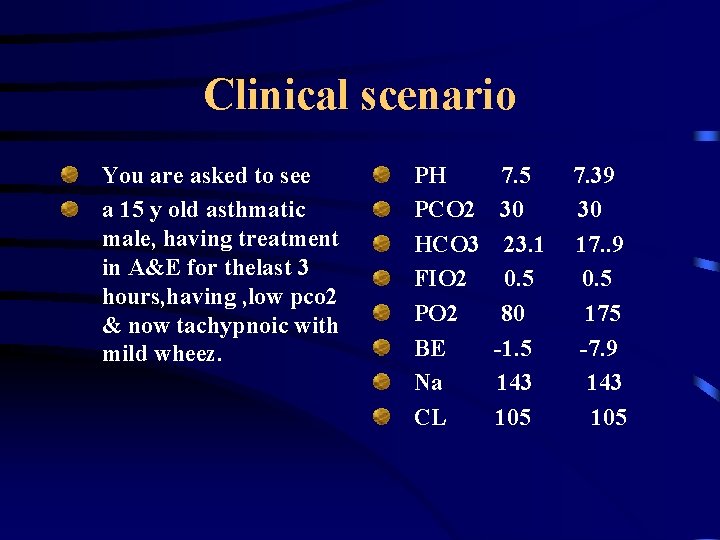
Clinical scenario You are asked to see a 15 y old asthmatic male, having treatment in A&E for thelast 3 hours, having , low pco 2 & now tachypnoic with mild wheez. PH PCO 2 HCO 3 FIO 2 PO 2 BE Na CL 7. 5 30 23. 1 0. 5 80 -1. 5 143 105 7. 39 30 17. . 9 0. 5 175 -7. 9 143 105

QUESTIONS Calculate& interpret A-a gradient. ? Describe acid base status. ? Has his asthma improved , reasons. ? What is the most likely explanation for the unchanging hypocapnoea. ?
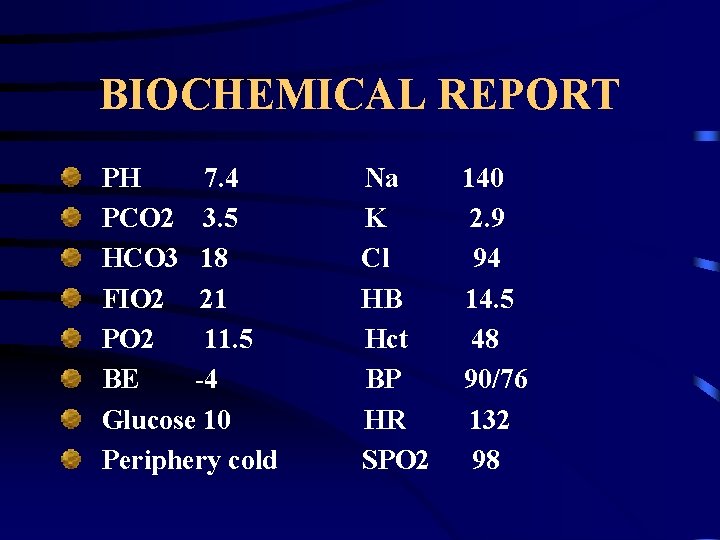
BIOCHEMICAL REPORT PH 7. 4 PCO 2 3. 5 HCO 3 18 FIO 2 21 PO 2 11. 5 BE -4 Glucose 10 Periphery cold Na K Cl HB Hct BP HR SPO 2 140 2. 9 94 14. 5 48 90/76 132 98
- Slides: 79