Prof Dr med Sigmund Silber FESC FACC FAHA

Prof. Dr. med. Sigmund Silber FESC, FACC, FAHA Cardiology Practice and Hospital Munich, Germany Outpatient Practice Heart Center at the Isar


Potential conflicts of interest Speaker's name: Sigmund Silber I have the following potential conflicts of interest to report: Research grant for the RESOLUTE AC trial Honorarium for this presentation

The Problem of Dual Antiplatelet Therapy (DAPT) and Anticoagulation Ø Ø S. P. , 86 y/o male, arterial hypertension / diabetes mellitus Presented with dyspnoea and tachycardia Echo: severely depressed LV-EF ECG: atypical atrial flutter

The Problem of Dual Antiplatelet Therapy (DAPT) and Anticoagulation Ø Ø S. P. , 86 y/o male, arterial hypertension / diabetes mellitus Presented with dyspnoea and tachycardia Echo: severely depressed LV-EF ECG: atypical atrial flutter


The Problem of Dual Antiplatelet Therapy (DAPT) and Anticoagulation Ø Ø Ø S. P. , 86 y/o male, arterial hypertension / diabetes mellitus Presented with dyspnoea and tachycardia Echo: severely depressed LV-EF ECG: atypical atrial flutter Actual Medication: • • • Rivaroxaban 15 mg/d ASA 100 mg/d Ramipril, Bisoprolol, Simvastatin and various diuretics
Patient needs Anticoagulation Ø BMS ? Ø Genous EPC capturing ? Ø DES ?
Ø DES: RESOLUTE 2, 5 / 14 mm

The Problem of Dual Antiplatelet Therapy (DAPT) and Anticoagulation Ø S. P. , 86 y/o male, arterial hypertension / diabetes mellitus Ø Stenting January 21 st 2013 Ø 4 weeks triple therapy with ASS 100 mg/d + Clopidogrel 75 ? S E mg/d and Rivaroxaban 15 mg/d for 4 weeks D e t Ø Then Clopidogrel discontinued u l o s e flutter Ø Did not agree to ablation for atrial R eth h t Ø Telephone follow-up May e 13 2013: free of symptoms s u e w id d y h W
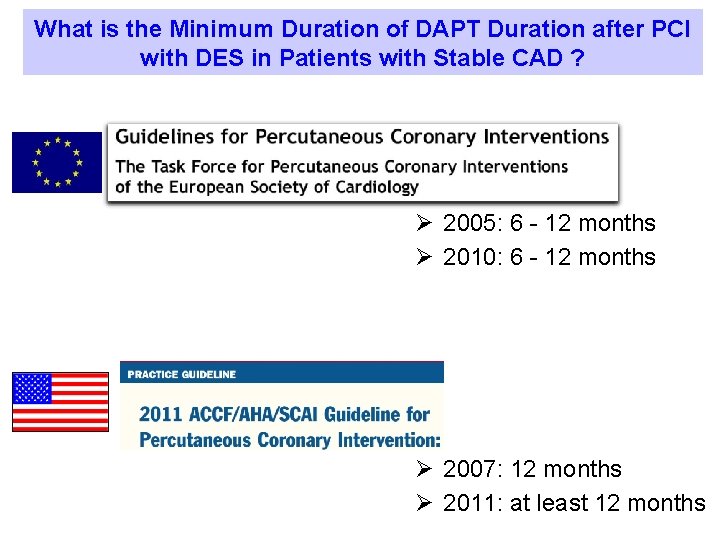
What is the Minimum Duration of DAPT Duration after PCI with DES in Patients with Stable CAD ? Ø 2005: 6 - 12 months Ø 2010: 6 - 12 months Ø 2007: 12 months Ø 2011: at least 12 months

Fully published randomized studies for DAPT duration after the guidelines have been finalized: DAPT Duration Message REAL/ZEST LATE (2010) 1 year vs. 2 years no advantage of 2 years EXCELLENT (2012) 6 months vs. 1 y no signif. difference PRODIGY (2013) 6 months vs. 2 y no advantage of longer DAPT (probably worse)
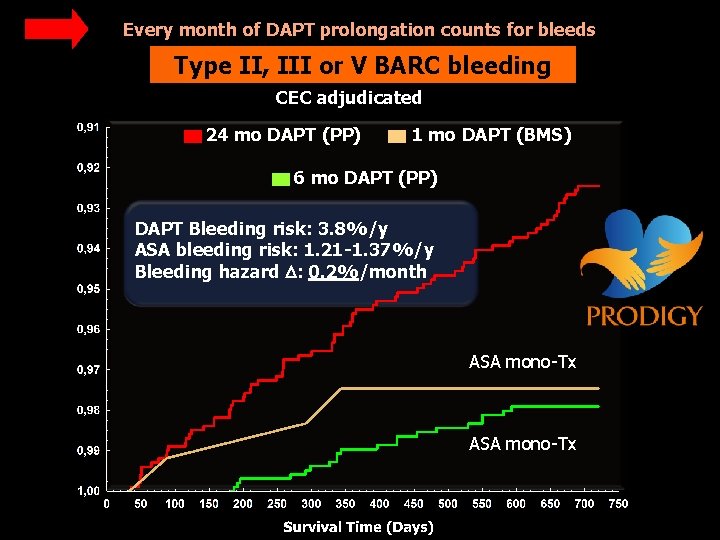
Every month of DAPT prolongation counts for bleeds Type II, III or V BARC bleeding CEC adjudicated 24 mo DAPT (PP) 1 mo DAPT (BMS) 6 mo DAPT (PP) DAPT Bleeding risk: 3. 8%/y ASA bleeding risk: 1. 21 -1. 37%/y Bleeding hazard : 0. 2%/month ASA mono-Tx

March 2013

Background • The European and USA guideline recommendations are based only on observational data and of first generation DES. No randomized trial of 6 months vs. less than 6 months exists. • Observational data suggest that a significant proportion of patients either interrupt or are unable to tolerate DAPT in this period. • It is unclear if earlier interruption and/or discontinuation of DAPT is associated with a higher risk of stent thrombosis (ST), particularly with newer generation DES.
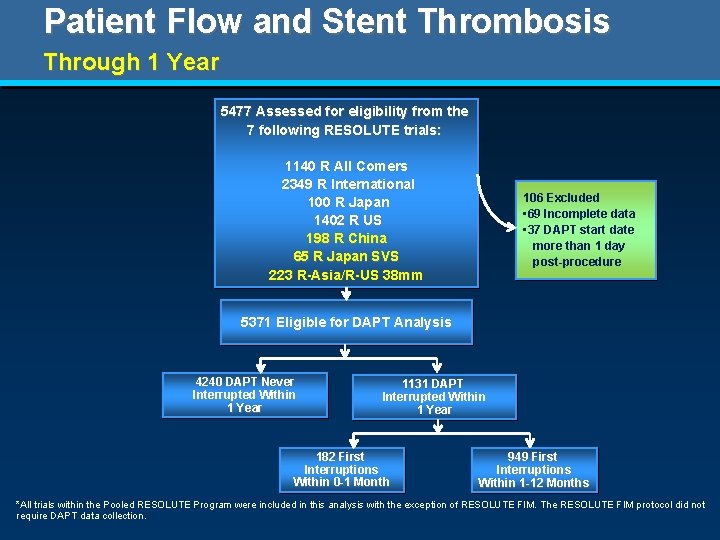
Patient Flow and Stent Thrombosis Through 1 Year 5477 Assessed for eligibility from the 7 following RESOLUTE trials: 1140 R All Comers 2349 R International 100 R Japan 1402 R US 198 R China 65 R Japan SVS 223 R-Asia/R-US 38 mm 106 Excluded • 69 Incomplete data • 37 DAPT start date more than 1 day post-procedure 5371 Eligible for DAPT Analysis 4240 DAPT Never Interrupted Within 1 Year 1131 DAPT Interrupted Within 1 Year 182 First Interruptions Within 0 -1 Month 949 First Interruptions Within 1 -12 Months *All trials within the Pooled RESOLUTE Program were included in this analysis with the exception of RESOLUTE FIM. The RESOLUTE FIM protocol did not require DAPT data collection.
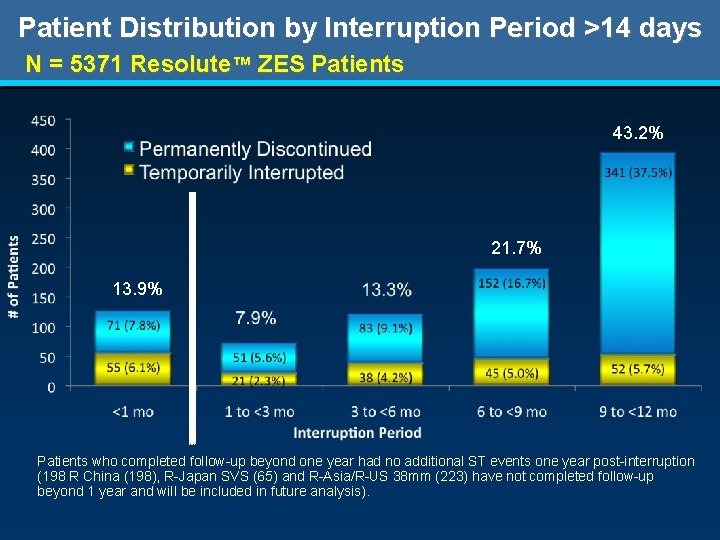
Patient Distribution by Interruption Period >14 days N = 5371 Resolute™ ZES Patients 43. 2% 21. 7% 13. 9% Patients who completed follow-up beyond one year had no additional ST events one year post-interruption (198 R China (198), R-Japan SVS (65) and R-Asia/R-US 38 mm (223) have not completed follow-up beyond 1 year and will be included in future analysis).
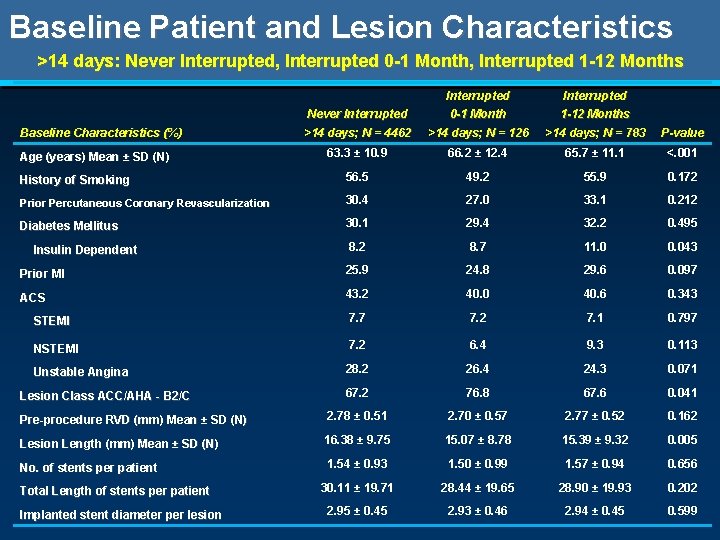
Baseline Patient and Lesion Characteristics >14 days: Never Interrupted, Interrupted 0 -1 Month, Interrupted 1 -12 Months Interrupted Never Interrupted 0 -1 Month 1 -12 Months >14 days; N = 4462 >14 days; N = 126 >14 days; N = 783 P-value 63. 3 ± 10. 9 66. 2 ± 12. 4 65. 7 ± 11. 1 <. 001 History of Smoking 56. 5 49. 2 55. 9 0. 172 Prior Percutaneous Coronary Revascularization 30. 4 27. 0 33. 1 0. 212 Diabetes Mellitus 30. 1 29. 4 32. 2 0. 495 8. 2 8. 7 11. 0 0. 043 Prior MI 25. 9 24. 8 29. 6 0. 097 ACS 43. 2 40. 0 40. 6 0. 343 STEMI 7. 7 7. 2 7. 1 0. 797 NSTEMI 7. 2 6. 4 9. 3 0. 113 Unstable Angina 28. 2 26. 4 24. 3 0. 071 67. 2 76. 8 67. 6 0. 041 Pre-procedure RVD (mm) Mean ± SD (N) 2. 78 ± 0. 51 2. 70 ± 0. 57 2. 77 ± 0. 52 0. 162 Lesion Length (mm) Mean ± SD (N) 16. 38 ± 9. 75 15. 07 ± 8. 78 15. 39 ± 9. 32 0. 005 No. of stents per patient 1. 54 ± 0. 93 1. 50 ± 0. 99 1. 57 ± 0. 94 0. 656 30. 11 ± 19. 71 28. 44 ± 19. 65 28. 90 ± 19. 93 0. 202 2. 95 ± 0. 45 2. 93 ± 0. 46 2. 94 ± 0. 45 0. 599 Baseline Characteristics (%) Age (years) Mean ± SD (N) Insulin Dependent Lesion Class ACC/AHA - B 2/C Total Length of stents per patient Implanted stent diameter per lesion
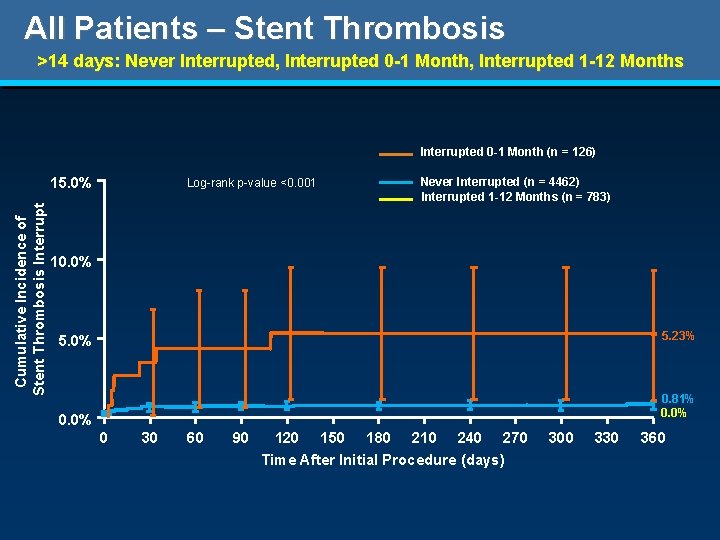
All Patients – Stent Thrombosis >14 days: Never Interrupted, Interrupted 0 -1 Month, Interrupted 1 -12 Months Interrupted 0 -1 Month (n = 126) Cumulative Incidence of Stent Thrombosis Interrupt 15. 0% Log-rank p-value <0. 001 Never Interrupted (n = 4462) Interrupted 1 -12 Months (n = 783) 10. 0% 5. 23% 5. 0% 0. 81% 0. 0% 0 30 60 90 120 150 180 210 240 270 Time After Initial Procedure (days) 300 330 360
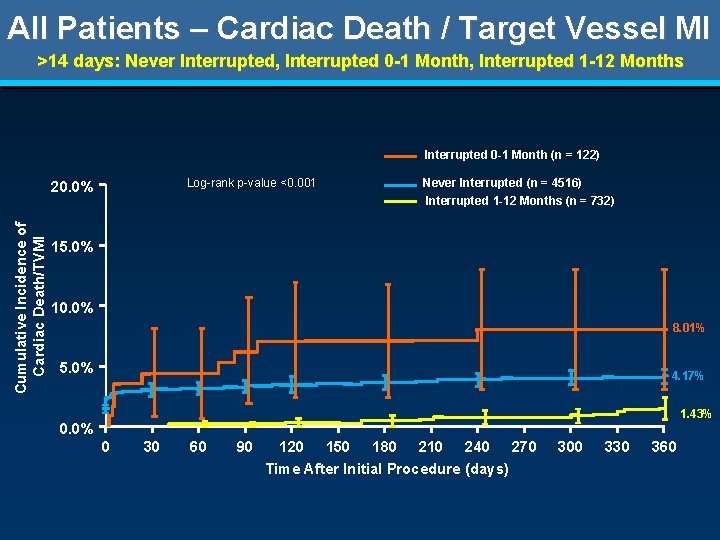
All Patients – Cardiac Death / Target Vessel MI >14 days: Never Interrupted, Interrupted 0 -1 Month, Interrupted 1 -12 Months Interrupted 0 -1 Month (n = 122) Log-rank p-value <0. 001 Cumulative Incidence of Cardiac Death/TVMI 20. 0% Never Interrupted (n = 4516) Interrupted 1 -12 Months (n = 732) 15. 0% 10. 0% 8. 01% 5. 0% 4. 17% 1. 43% 0. 0% 0 30 60 90 120 150 180 210 240 270 Time After Initial Procedure (days) 300 330 360

Strengths and Limitations Strengths: • All data were collected from regulatory trials with 100% monitoring (except for RESOLUTE International with 25% randomly assigned monitoring) and high rates of follow-up. Limitations: • This is a post-hoc analysis of pooled datasets, the generalized application of these results to the entire population demands careful attention given that a larger sample size might be required to provide a definite answer regarding low frequency events such as ST.

Conclusions • One-year data from the RESOLUTE Clinical Program indicate low stent thrombosis rates for those who interrupted or discontinued DAPT after 1 month. • While physicians should adhere to current ESC or ACC/AHA/SCAI Guidelines for PCI, in the event a patient interrupts or discontinues DAPT medication 1 month or more after stent implantation, the incremental risk of stent thrombosis may be low with the Resolute. TM stent, as used in these included trials.

Thank you !
- Slides: 23