Prof Dr med Sigmund Silber FACC FESC Cardiology

Prof. Dr. med. Sigmund Silber FACC, FESC Cardiology Practice and Hospital Munich, Germany Outpatient Practice Heart Center at the Isar


Disclosures I, Sigmund Silber, MD, have the following financial relationships to disclose: Type of relation: None (related to this presentation) *Type of relation: Consultancy, employment, honoraria, other compensation *Level of remuneration: high (over $50, 000 yearly), moderate (10 -50, 000), modest (under 10, 000)

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?

EHJ, 31: 2501 -2555, (2010)


EHJ, 31: 2501 -2555, (2010)

EHJ, 31: 2501 -2555, (2010)

EHJ, 31: 2501 -2555, (2010)

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?

Which Randomized All Comers DES studies do we have ? Ø COMPARE 1 st generation vs. 2 nd generation § Taxus Liberté vs. Xience V Ø LEADERS 1 st generation vs. 2 nd generation § Cypher vs. Biomatrix Ø RESOLUTE ALL COMERS § Xience V vs. Resolute 2 nd generation vs. 2 nd generation Ø TWENTE § Xience V vs. Resolute 2 nd generation vs. 2 nd generation
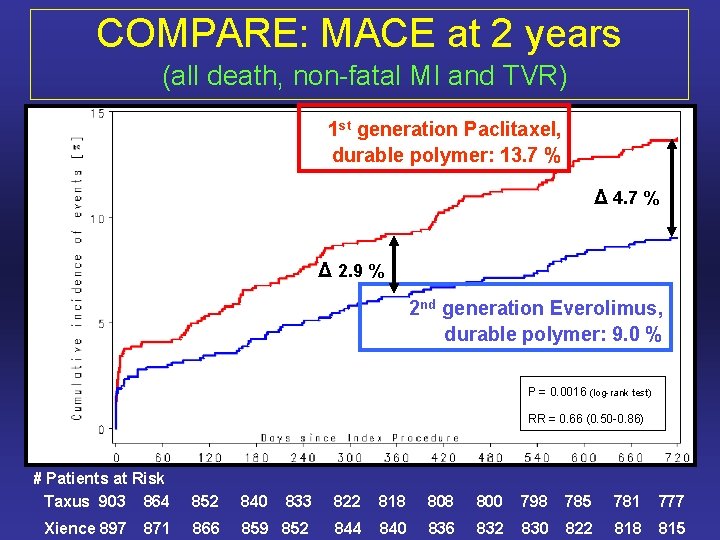
COMPARE: MACE at 2 years (all death, non-fatal MI and TVR) 1 st generation Paclitaxel, durable polymer: 13. 7 % Δ 4. 7 % Δ 2. 9 % 2 nd generation Everolimus, durable polymer: 9. 0 % P = 0. 0016 (log-rank test) RR = 0. 66 (0. 50 -0. 86) # Patients at Risk Taxus 903 864 Xience 897 871 852 840 833 822 818 800 798 785 781 777 866 859 852 844 840 836 832 830 822 818 815
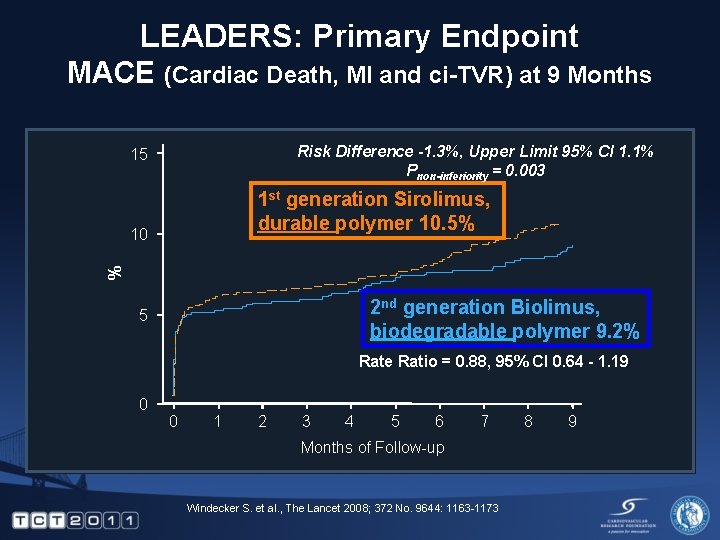
LEADERS: Primary Endpoint MACE (Cardiac Death, MI and ci-TVR) at 9 Months Risk Difference -1. 3%, Upper Limit 95% CI 1. 1% Pnon-inferiority = 0. 003 15 1 st generation Sirolimus, durable polymer 10. 5% % 10 2 nd generation Biolimus, biodegradable polymer 9. 2% 5 Rate Ratio = 0. 88, 95% CI 0. 64 - 1. 19 0 0 1 2 3 4 5 6 7 Months of Follow-up Windecker S. et al. , The Lancet 2008; 372 No. 9644: 1163 -1173 8 9
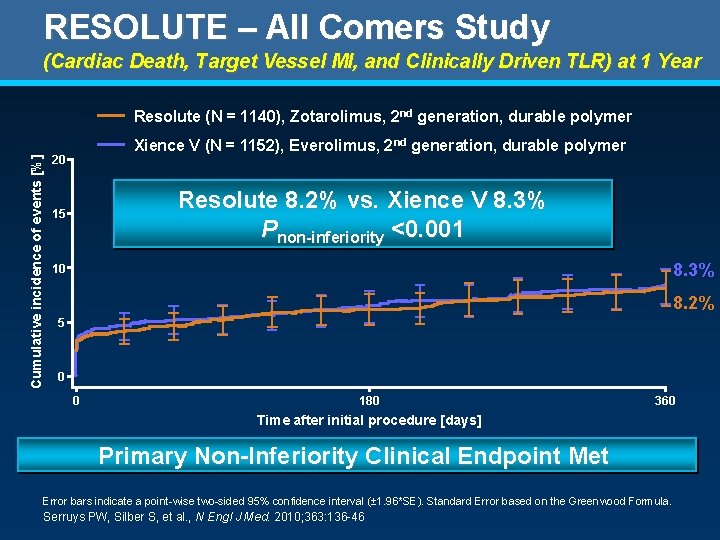
RESOLUTE – All Comers Study (Cardiac Death, Target Vessel MI, and Clinically Driven TLR) at 1 Year Cumulative incidence of events [%] Resolute (N = 1140), Zotarolimus, 2 nd generation, durable polymer Xience V (N = 1152), Everolimus, 2 nd generation, durable polymer 20 Resolute 8. 2% vs. Xience V 8. 3% Pnon-inferiority <0. 001 15 8. 3% 10 8. 2% 5 0 0 180 360 Time after initial procedure [days] No. at risk ZES EES 0 1140 1152 30 1110 1123 60 1084 1088 90 1076 1080 120 1078 150 1062 1074 180 1068 210 1058 1061 240 1051 1047 270 1042 1046 300 1038 Primary Non-Inferiority Clinical Endpoint Met 330 1037 1032 360 1025 1019 Error bars indicate a point-wise two-sided 95% confidence interval (± 1. 96*SE). Standard Error based on the Greenwood Formula. Serruys PW, Silber S, et al. , N Engl J Med. 2010; 363: 136 -46
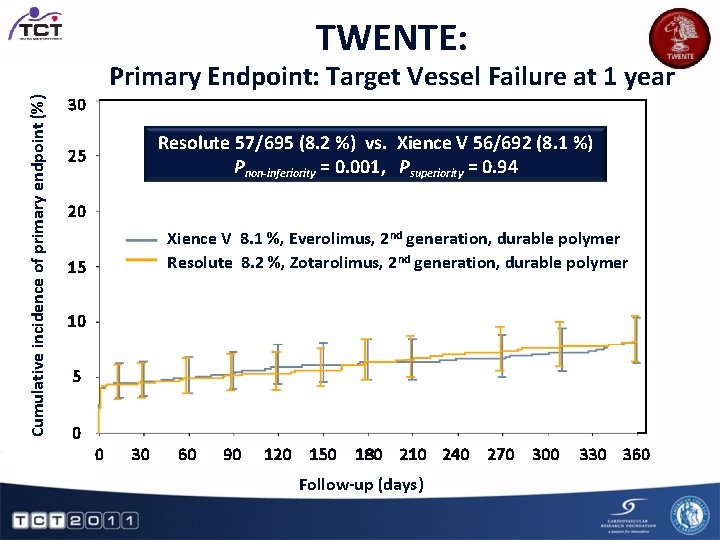
TWENTE: Cumulative incidence of primary endpoint (%) Primary Endpoint: Target Vessel Failure at 1 year 30 Resolute 57/695 (8. 2 %) vs. Xience V 56/692 (8. 1 %) Pnon-inferiority = 0. 001, Psuperiority = 0. 94 25 20 Xience V 8. 1 %, Everolimus, 2 nd generation, durable polymer Resolute 8. 2 %, Zotarolimus, 2 nd generation, durable polymer 15 10 5 0 0 30 60 90 120 150 180 210 240 270 300 Follow-up (days) 330 360
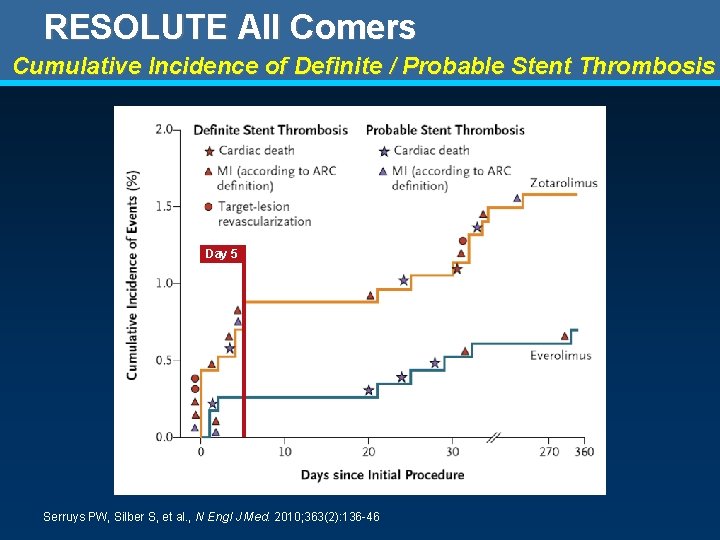
RESOLUTE All Comers Cumulative Incidence of Definite / Probable Stent Thrombosis Day 5 Serruys PW, Silber S, et al. , N Engl J Med. 2010; 363(2): 136 -46
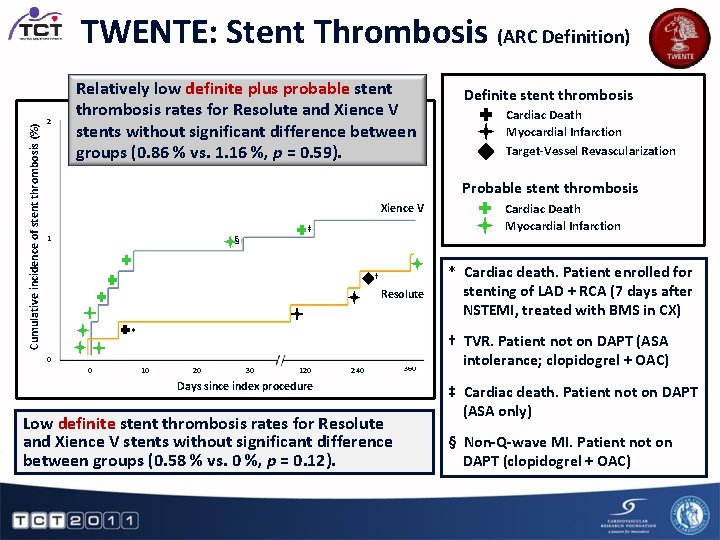
Cumulative incidence of stent thrombosis (%) TWENTE: Stent Thrombosis (ARC Definition) 2 Relatively low definite plus probable stent thrombosis rates for Resolute and Xience V stents without significant difference between Definite Stent Thrombosis Probable Stent Thrombosis groups (0. 86 % vs. 1. 16 %, p Cardiac = 0. 59). Cardiac Death Target-Vessel Revascularization 1 Xience V ‡ § † Resolute * 0 0 10 20 30 120 Cardiac Death Myocardial Infarction Target-Vessel Revascularization Probable stent thrombosis Myocardial Infarction Definite stent thrombosis 240 Days since index procedure Low definite stent thrombosis rates for Resolute and Xience V stents without significant difference between groups (0. 58 % vs. 0 %, p = 0. 12). 360 Cardiac Death Myocardial Infarction * Cardiac death. Patient enrolled for stenting of LAD + RCA (7 days after NSTEMI, treated with BMS in CX) † TVR. Patient not on DAPT (ASA intolerance; clopidogrel + OAC) ‡ Cardiac death. Patient not on DAPT (ASA only) § Non-Q-wave MI. Patient not on DAPT (clopidogrel + OAC)
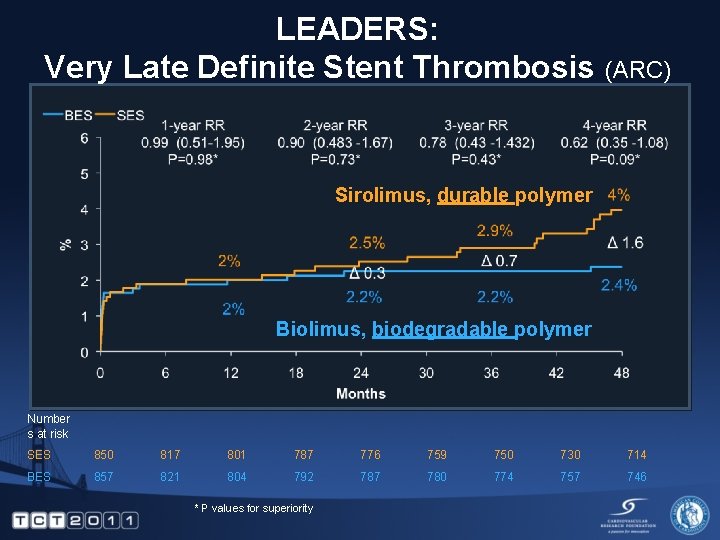
LEADERS: Very Late Definite Stent Thrombosis (ARC) Sirolimus, durable polymer Biolimus, biodegradable polymer Number s at risk SES 850 817 801 787 776 759 750 730 714 BES 857 821 804 792 787 780 774 757 746 * P values for superiority

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?
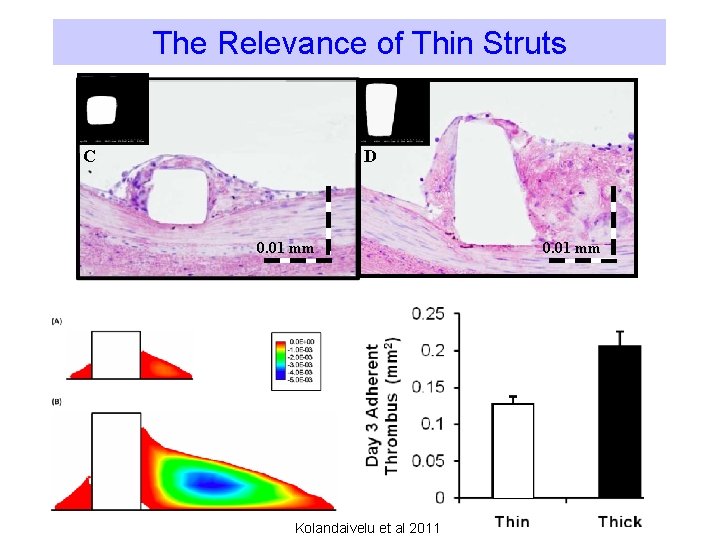
The Relevance of Thin Struts C D 0. 01 mm Kolandaivelu et al 2011 0. 01 mm
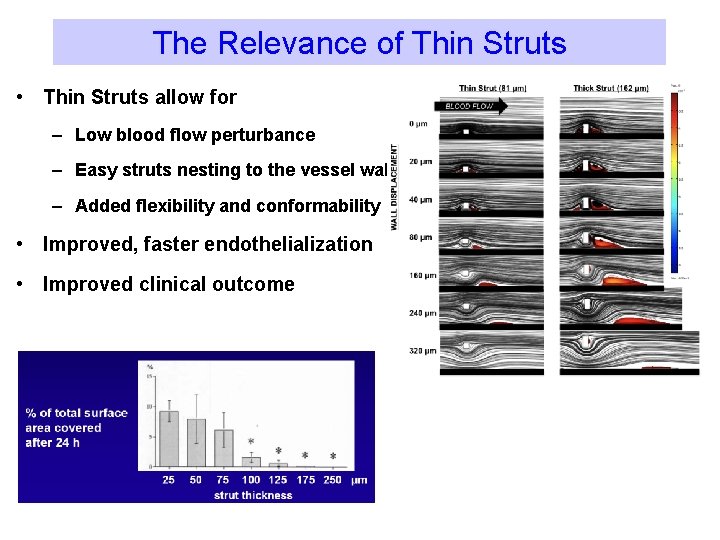
The Relevance of Thin Struts • Thin Struts allow for – Low blood flow perturbance – Easy struts nesting to the vessel wall – Added flexibility and conformability • Improved, faster endothelialization • Improved clinical outcome
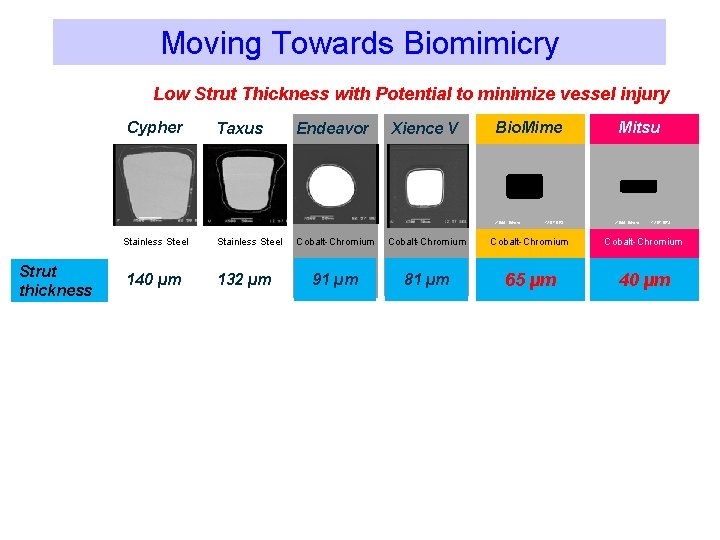
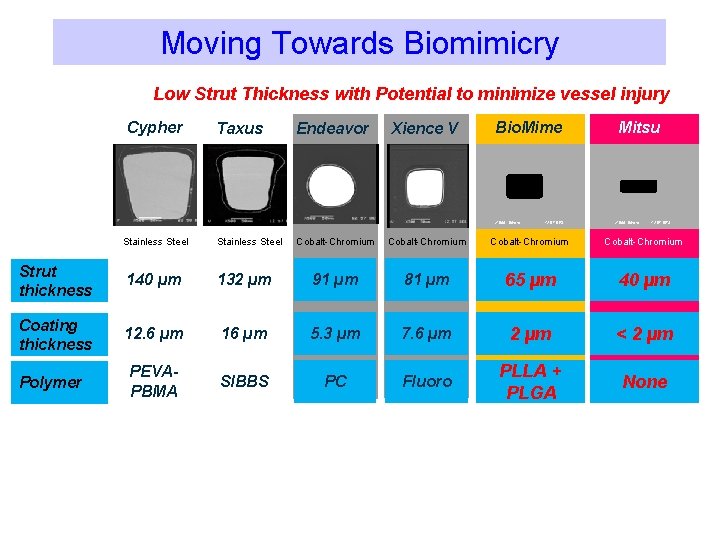
Moving Towards Biomimicry Low Strut Thickness with Potential to minimize vessel injury Cypher Taxus Endeavor Xience V Bio. Mime X 500 50 mm Stainless Steel Strut thickness 140 µm Stainless Steel 132 µm 12 57 DES Mitsu X 500 50 mm 12 57 DES Cobalt-Chromium 91 µm 81 µm 65 µm 40 µm

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?
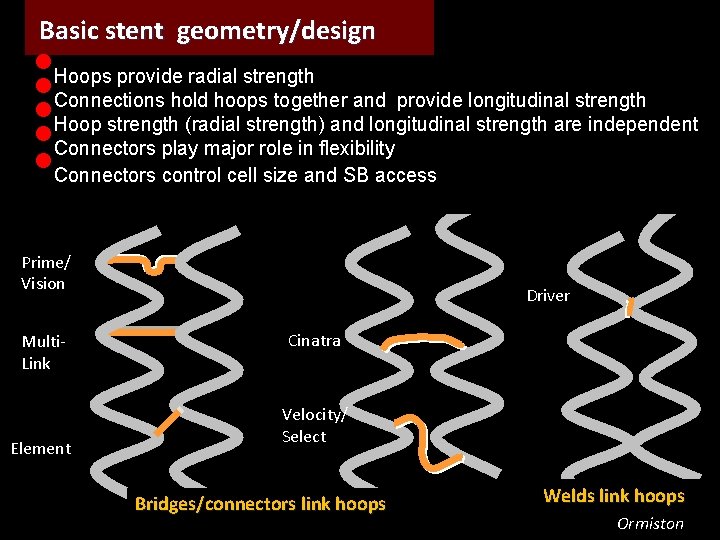
Basic stent geometry/design • • • Hoops provide radial strength Connections hold hoops together and provide longitudinal strength Hoop strength (radial strength) and longitudinal strength are independent Connectors play major role in flexibility Connectors control cell size and SB access Prime/ Vision Multi. Link Element Driver Cinatra Velocity/ Select Bridges/connectors link hoops Welds link hoops Ormiston

How far are stents compressed with 0. 5 N force ? • The Cypher Select did not shorten • The Element shortened 5 mm • Xience 1 mm Ormiston
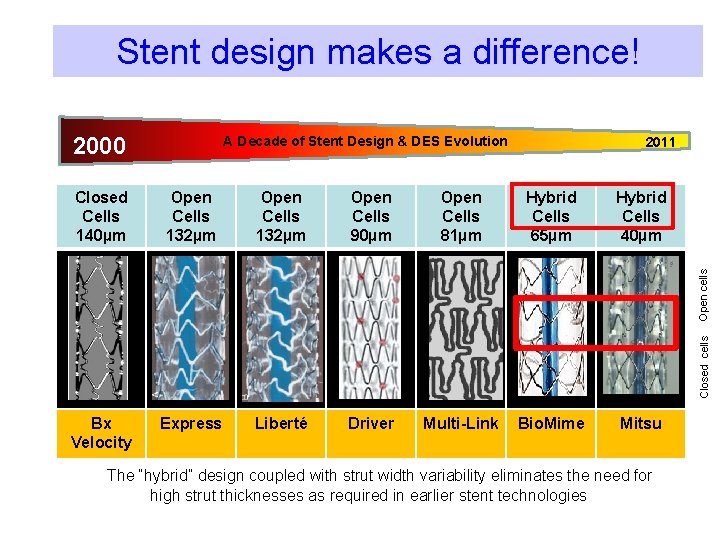
Stent design makes a difference! 2000 A Decade of Stent Design & DES Evolution 2011 Open Cells 132µm Open Cells 90µm Open Cells 81µm Hybrid Cells 65µm Hybrid Cells 40µm Bx Velocity Express Liberté Driver Multi-Link Bio. Mime Mitsu Closed cells Open cells Closed Cells 140µm The “hybrid” design coupled with strut width variability eliminates the need for high strut thicknesses as required in earlier stent technologies
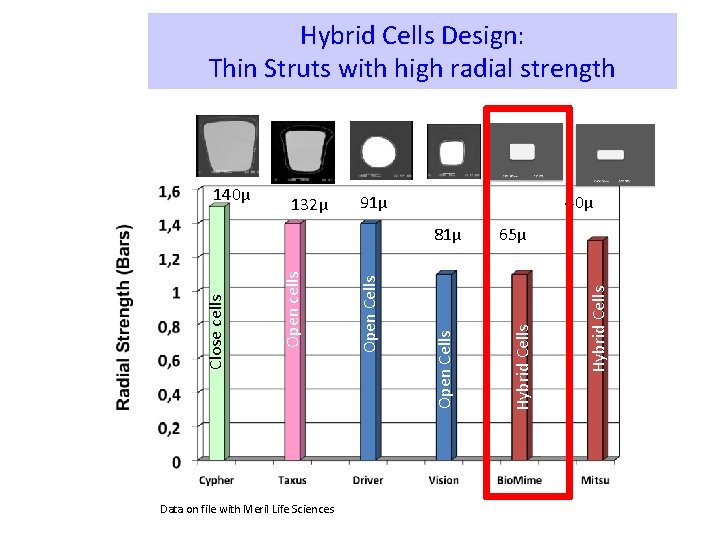
Hybrid Cells Design: Thin Struts with high radial strength 40µ 91µ Data on file with Meril Life Sciences Open Cells Open cells Close cells 81µ 65µ Hybrid Cells 132µ Hybrid Cells 140µ

The Bio. Mime DES: Ø Cobalt chromium (L 605) platform. 65µm strut thickness. Ø Hybrid cell design, an intelligent mix of open and close cells. Ø Excellent radial strength & high flexibility. § <3% recoil and 0. 29% foreshortening Ø Special electro-polishing technique eliminates surface nickel oxides Hybrid design Open cells in mid segment Close cells at edges Strut width variability

Bio. Mime Stent Architecture • Cobalt chromium (L 605) platform. 65µm strut thickness. • Hybrid cell design, an intelligent mix of open and close cells. • Excellent radial strength & high flexibility. ¡ <3% recoil and 0. 29% foreshortening • Special electro-polishing technique eliminates surface nickel oxides Hybrid design Open cells in mid segment Close cells at edges Strut width variability

MERIT-1 Study with the Bio. Mime DES: • Design: Phase IV, prospective, single arm, single centre study. N = 30 • Objective: To evaluate the safety and efficacy of Bio. Mime Sirolimus Eluting Coronary Stent • Inclusion Criteria: Single, Discrete, De novo lesions, Mean Vessel Lumen Diameter 2. 5, 3. 0 and 3. 5 mm. Stent lengths 19 to 24 mm • Exclusion Criteria: CTO’s, Bifurcations, SVG’s, AMI’s, LM disease, LVEF <30 %

MERIT-1 Study with the Bio. Mime DES: In-Stent Late Loss: 0. 15 mm Hierarchical 30 days 8 months 20 months 0 (0%) Cardiac Death 0 (0%) Myocardial Infarction (%) Q-wave MI Non-Q-wave MI 0 (0%) 0 (0%) TLR (%) 0 (0%) TVR excluding TLR (%) 0 (0%) Primary Endpoint: Composite of Death, MI & TLR (%) All Death (%)

MERIT-2 Study with the Bio. Mime DES: Ø Design: Prospective, Non-Randomized, Multi- Centre, Complex, Real world study involving 250 patients Ø Ø Objective: Assess the safety and efficacy of the Bio. Mime™ Sirolimus Eluting Coronary Stent System in Complex Real World patients Inclusion Criteria: To include most lesions (CTO’s included) § Vessel Diameter : >2. 5 and <3. 5 mm § Lesion lengths up to 37 mm treated with maximum stent length of 40 mm Ø Exclusion Criteria: SVG’s, AMI’s, LM disease, LVEF <30%
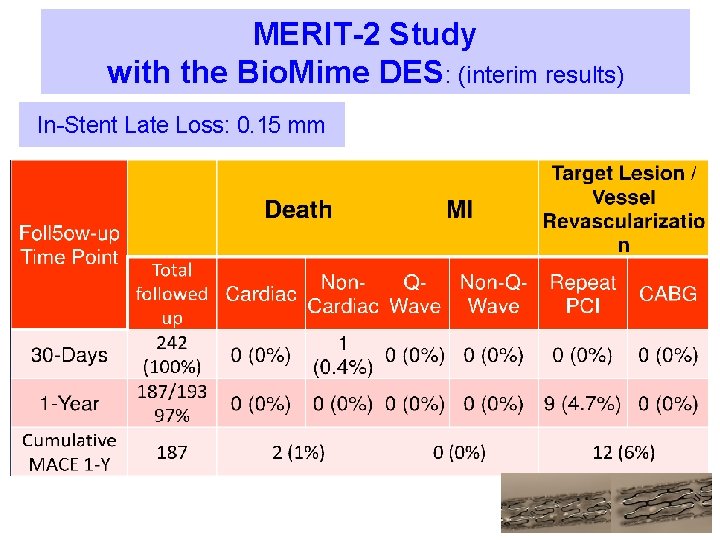
MERIT-2 Study with the Bio. Mime DES: (interim results) In-Stent Late Loss: 0. 15 mm

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?
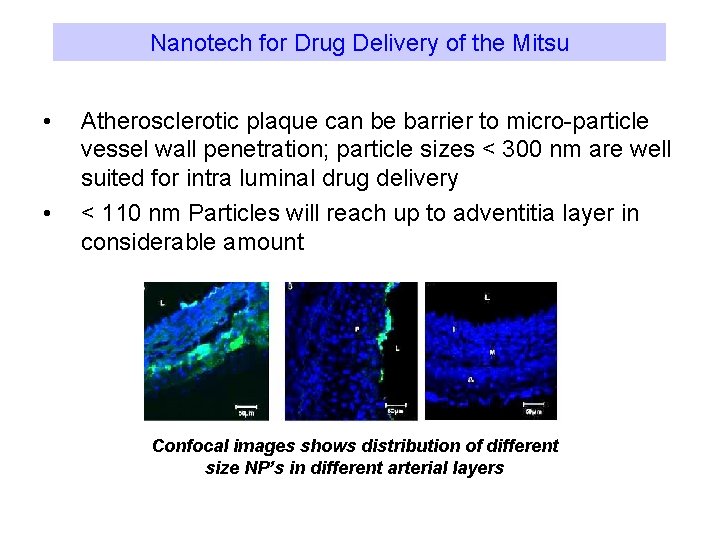
Nanotech for Drug Delivery of the Mitsu • • Atherosclerotic plaque can be barrier to micro-particle vessel wall penetration; particle sizes < 300 nm are well suited for intra luminal drug delivery < 110 nm Particles will reach up to adventitia layer in considerable amount Confocal images shows distribution of different size NP’s in different arterial layers

Nanotech for Drug Delivery of the Mitsu • • Control and target drug release Improved stability Enhanced drug content (compare to other carriers) Feasibility of carrying both lipophilic and hydrophilic drugs Most lipid are biodegradable, SLN have excellent biocompatibility Benefits include lower doses and lower side effects By using different sizes NP’s, drug delivery to each layer and every affected cells can be achieved Because of their small size, they are often taken up by cells where larger/micro particles would be excluded

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?
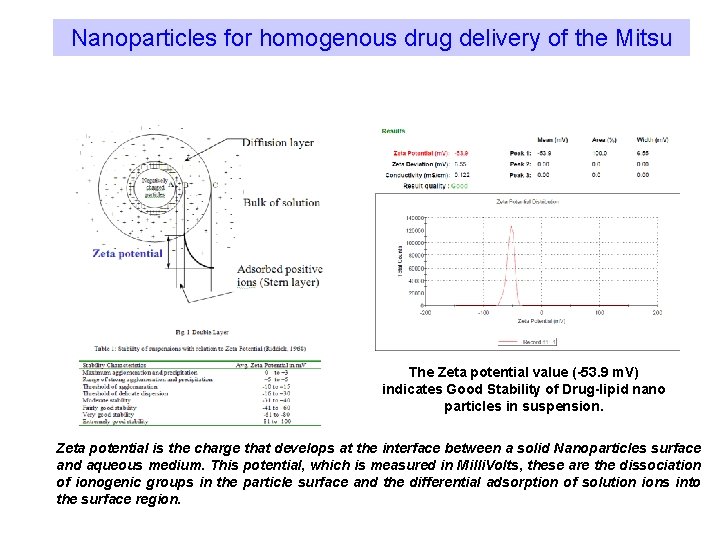
Nanoparticles for homogenous drug delivery of the Mitsu The Zeta potential value (-53. 9 m. V) indicates Good Stability of Drug-lipid nano particles in suspension. Zeta potential is the charge that develops at the interface between a solid Nanoparticles surface and aqueous medium. This potential, which is measured in Milli. Volts, these are the dissociation of ionogenic groups in the particle surface and the differential adsorption of solution ions into the surface region.

Nanoparticles for homogenous drug delivery of the Mitsu • Nanotechnology based • Allows for high coating uniformity and low drug • • • dosage Controlled and reproducible drug release kinetics Rapid release of drug into tissue with drug depot or reservoir effect Renders the stent drug free within a short time § Rapid stent healing (endothelialization) § Rapid tissue absorption (SNP has high tissue diffusion co-efficient) § Formulation ensures drug availability for 1 month
Moving Towards Biomimicry Low Strut Thickness with Potential to minimize vessel injury Cypher Taxus Endeavor Xience V Bio. Mime X 500 50 mm Stainless Steel 12 57 DES Mitsu X 500 50 mm 12 57 DES Cobalt-Chromium Strut thickness 140 µm 132 µm 91 µm 81 µm 65 µm 40 µm Coating thickness 12. 6 µm 16 µm 5. 3 µm 7. 6 µm 2 µm < 2 µm PEVAPBMA SIBBS PC Fluoro PLLA + PLGA None Polymer

Drug-eluting Stents (DES): Current Status and desirable improvements 1. Current Status of DES: a) Indications and Contraindications b) Efficacy and Safety: where are the current limits ? 2. Desirable Improvements of DES to possibly increase efficacy and safety: a) b) c) d) e) Thinner struts ? Different cell design but maintaining the strength ? No polymer ? More homogenous distribution of the drug ? Better drugs ?
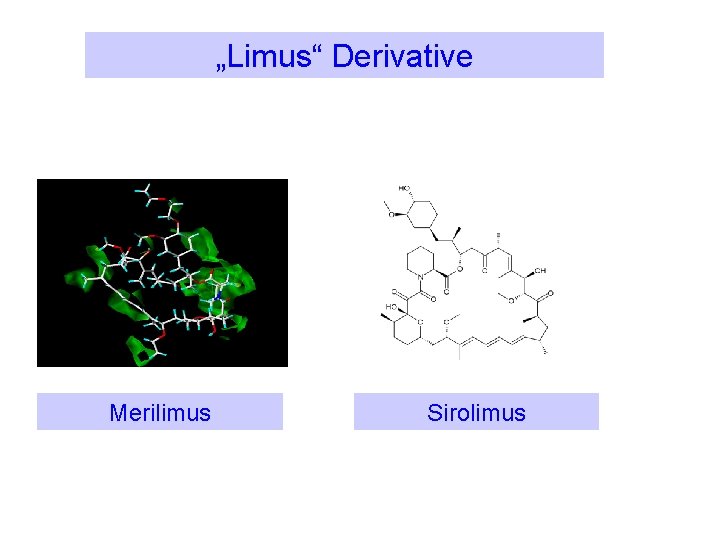
„Limus“ Derivative Merilimus Sirolimus

Merilimus The Anti-proliferative Drug Ø Merilimus is a Meril Life Sciences invention Ø Heterogeneous 5 member ring on the parent limus molecule Ø Better toxicological profile than Sirolimus and a wider therapeutic window (more lipophilic)) fk. BP-12 receptor site adaptation by Merilimus molecule Ø Low drug dosing of 0. 4 µg/mm 2 possible to get optimal antiproliferative effect

Nanotech The Polymer-Free Formulation Unique Formulation - Solid lipid nano-spheres (SLN) consisting of Merilimus + lipid (<300 nm) SLN rapidly leave stent and enter vessel wall with prolonged tissue residence time SEM picture of struts coated with nanoformulation
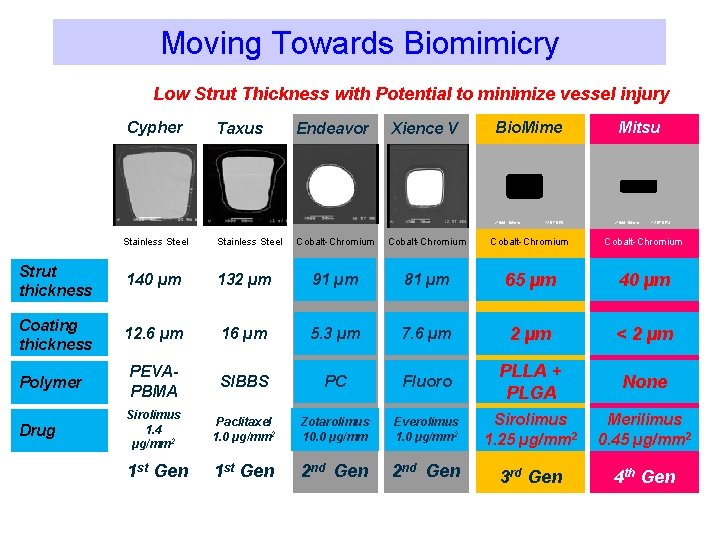
Moving Towards Biomimicry Low Strut Thickness with Potential to minimize vessel injury Cypher Taxus Endeavor Xience V Bio. Mime X 500 50 mm Stainless Steel 12 57 DES Mitsu X 500 50 mm 12 57 DES Cobalt-Chromium Strut thickness 140 µm 132 µm 91 µm 81 µm 65 µm 40 µm Coating thickness 12. 6 µm 16 µm 5. 3 µm 7. 6 µm 2 µm < 2 µm Polymer PEVAPBMA SIBBS PC Fluoro PLLA + PLGA None Drug Sirolimus 1. 4 µg/mm 2 Paclitaxel 1. 0 µg/mm 2 Zotarolimus 10. 0 µg/mm Everolimus 1. 0 µg/mm 2 Sirolimus 1. 25 µg/mm 2 Merilimus 0. 45 µg/mm 2 1 st Gen 2 nd Gen 3 rd Gen 4 th Gen

Mitsu – The New Merilimus-eluting Polymer-free Coronary Stent System New. Stent Design • 40 µm thin stent • Cobalt Chromium L 605 • Hybrid stent design • Variable strut width • Tapered balloon shoulders New Drug New Delivery System • Merilimus – new limus analogue • Novel SLN polymer-free formulation • Cytostatic • Low drug for same effect • Anti-inflammatory • Fast release kinetics • Lipophilic • • Wide therapeutic window Completely biocompatible and anti-inflammatory A unique differentiated new DES with high potentials !

Drug-eluting Stents (DES): Current Status and desirable improvements Take home Message: 1. 2. 3. 4. 5. 6. 7. DES have revolutionized PCI. Nevertheless, the current rate of failure with second generation DES seems to be 8% in the first year. Alhough DES are indicated for the majority of the patients, the key limitation is the need for prolonged dual antiplatelet therapy (DAPT). Although rare with even newer generation DES, the problem of stent thrombosis continues to exist (1% in the first year). It may possibly be related to the polymer, especially regarding the very late events. Improvements are still necessary, regarding strut thickness, stent pattern, polymer-free delivery systems and new drugs. With its hybrid-cell, thin-strut stent design and a biodegradable polymer, the Bio. Mime DES represents a transition to the next generation Mitsu DES. The Merilimus-eluting, hybrid-cell and thin-strut polymer-free Mitsu DES is an innovative concept addressing the current critical issues of available DES. Future studies will evaluate this innovative concept in experimental and clinical studies. Hopefully, the necessary duration of DAPT can be shortened with the Mitsu DES.

The best way to predict the future. . . …is to create it. . . Abraham Lincoln

- Slides: 50