Prof Dr Alam Sher HOD Biochemistry 1 AMINO

Prof. Dr. Alam Sher HOD Biochemistry 1

AMINO ACIDS � CLASSIFICATION AND STRUCTURE OF AMINO ACIDS � Amino acids can be classified into 3 groups depending on their reaction in solution. � A. Neutral � B. Acidic and � C. Basic. 2
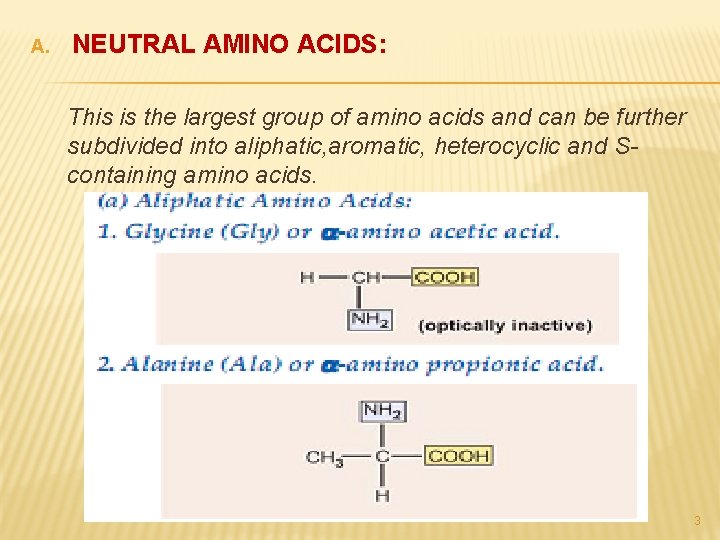
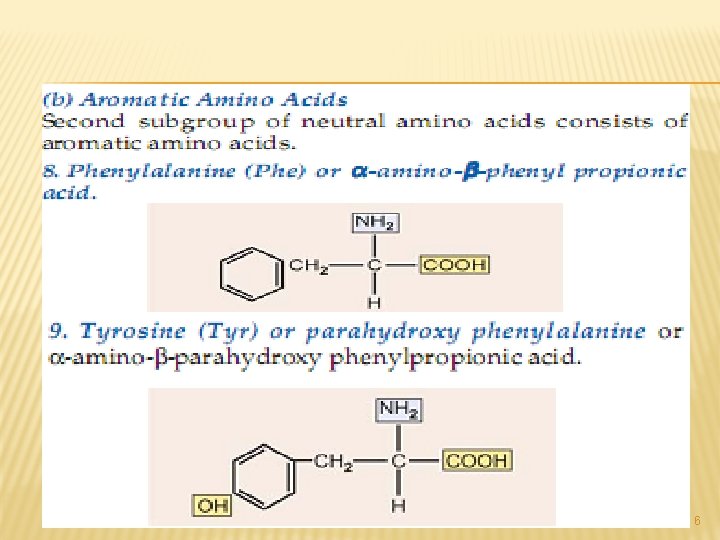
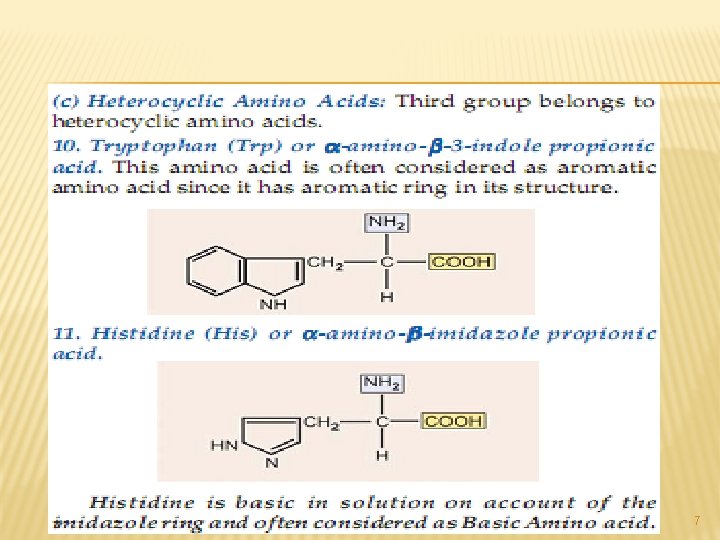
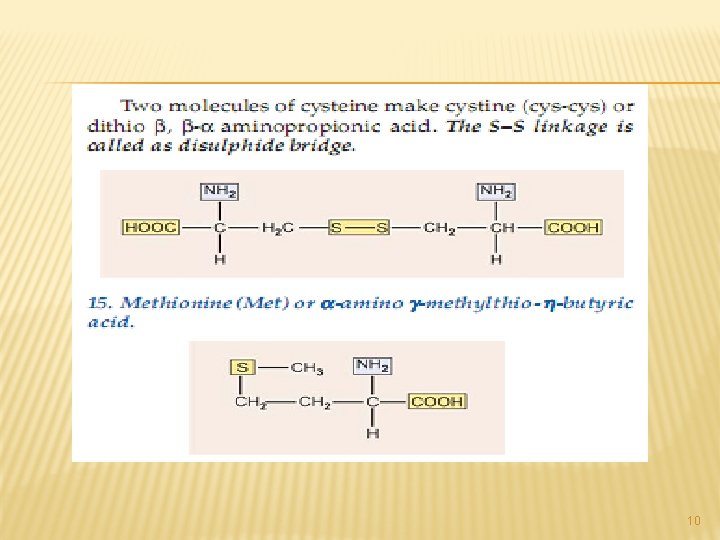
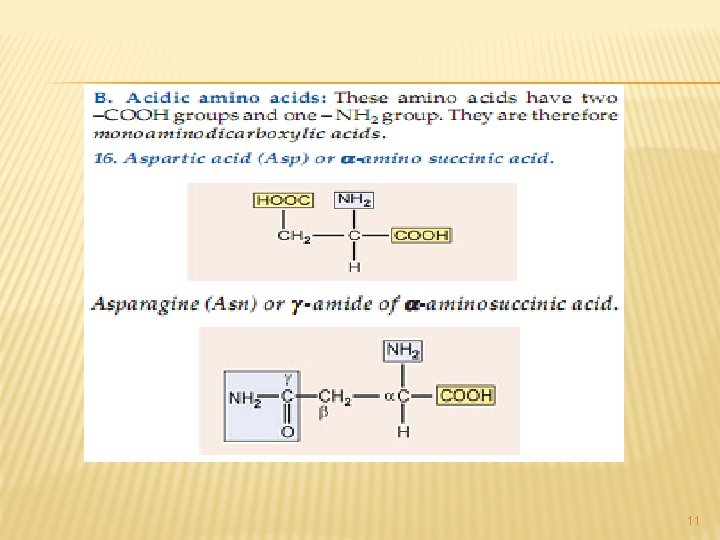
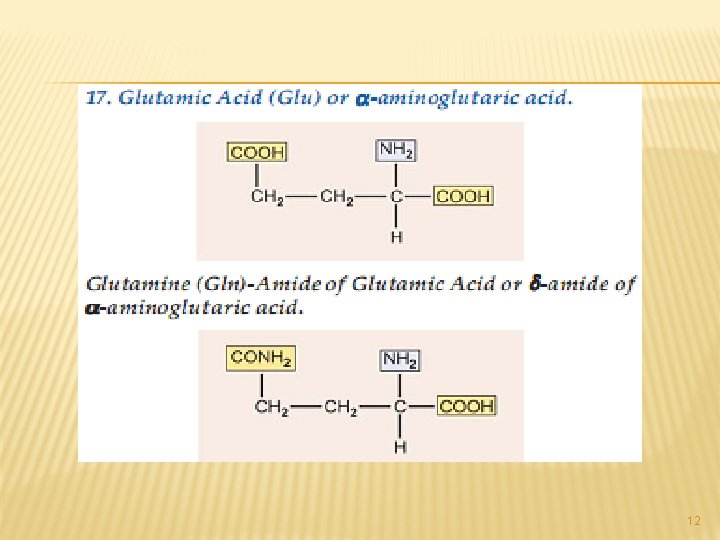
A. NEUTRAL AMINO ACIDS: This is the largest group of amino acids and can be further subdivided into aliphatic, aromatic, heterocyclic and Scontaining amino acids. 3
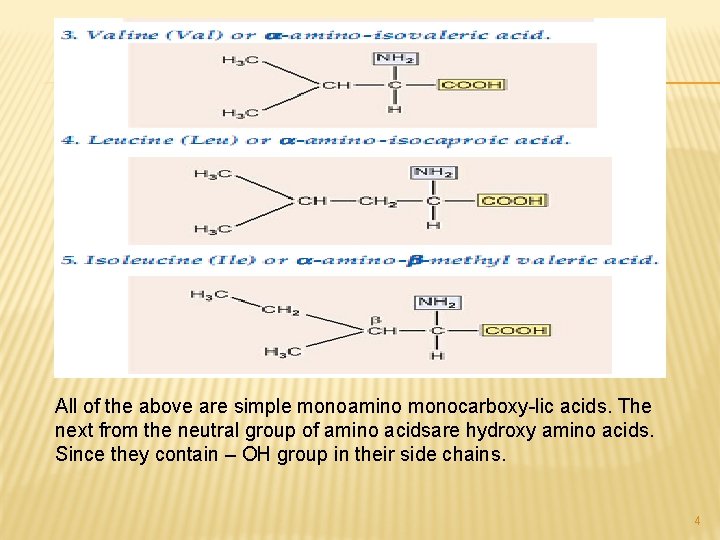
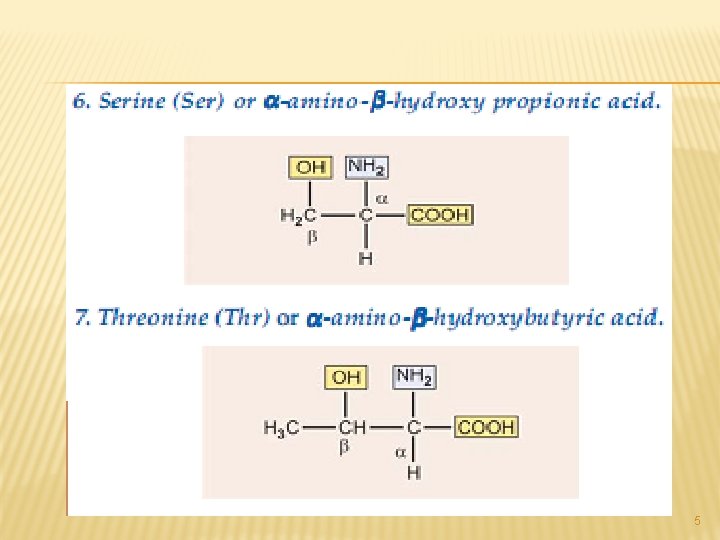
All of the above are simple monoamino monocarboxy-lic acids. The next from the neutral group of amino acidsare hydroxy amino acids. Since they contain – OH group in their side chains. 4
5
6
7

8

9
10
11
12
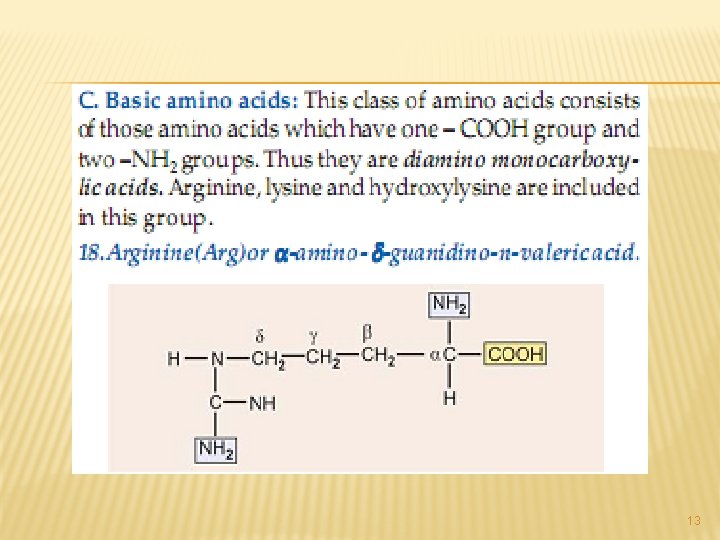
13
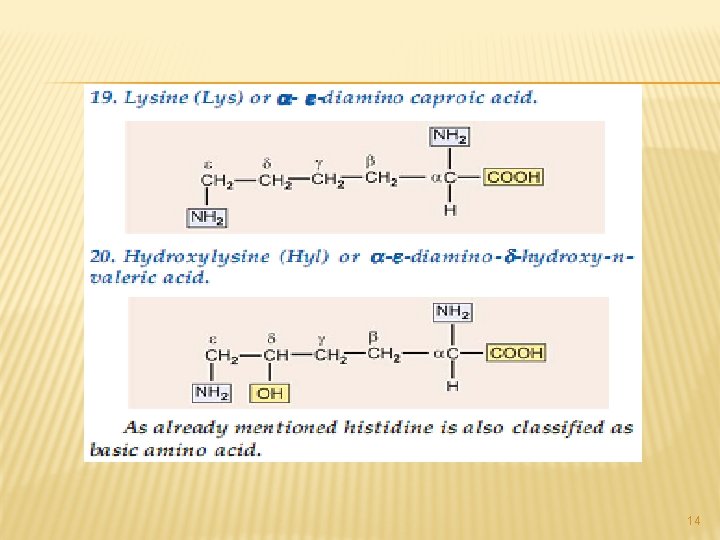
14

NON-STANDARD AMINO ACIDS A. The compounds similar to basic structure of amino acids but do not occur in proteins. Examples of some of those are: � • β-alanine: They found in coenzyme A. � • Taurine: They found in bile acids � • Ornithine and citrulline: They are intermediates in urea cycle � • Thyroxine (T 4) and Tri-iodo Thyronine (T 3): Thyroid hormones synthesised from tyrosine. � • γ-aminobutyric acid (GABA): A neurotransmitter produced from glutamic acid. � • β-amino isobutyric acid: These are end product of pyrimidine metabolism. � • δ-aminolaevulinic acid (δ-ALA): These are inter-mediate in haem synthesis. � • S-adenosyl methionine (SAM): These are methyl donor formed from L-methionine � • 3, 4 -dihydroxy phenyl alanine (DOPA): A precursor of 15 mela nine pigment.

B. D-AMINO ACIDS: These are non-standard amino acids—Amino acids normally isolated from animal and plants are L-amino acids. But certain D-amino acids are found in bacteria and antibiotics and in brain tissues of animals. • D-glutamic acid and D-Alanine are constituents of bacterial cell walls. � • D-amino acids are found in certain antibiotics, e. g. gramicidin-S, Actinomycin-D. � • Animal tissues contain L-amino acids which are deaminated by L-amino acid oxidase. � But there is also present D-amino acid oxidase the function of which was not known. � Now D-amino acids like D-aspartate and D-serine have been found in brain tissue. � This explains the existence of D-amino acid oxidase. � 16

FUNCTIONS OF AMINO ACIDS � � � Apart from being the monomeric constituents of proteins and peptides, amino acids serve variety of functions. (a) Some amino acids are converted to carbohydrates and are called as glucogenic amino acids. (b) Specific amino acids give rise to specialised products, e. g. • Tyrosine forms hormones such as thyroid hormones, (T 3, T 4), epinephrine and norepinephrine and a pigment called melanin. • Tryptophan can synthesise a vitamin called niacin. • Glycine, arginine and methionine synthesise creatine. • Glycine and cysteine help in synthesis of Bile salts. • Glutamate, cysteine and glycine synthesise glutathione. • Histidine changes to histamine on decarboxylation. • Serotonin is formed from tryptophan. 17

� � � Glycine is used for the synthesis of haem. Pyrimidines and purines use several amino acids for their synthesis such as aspartate and glutamine for pyrimidines and glycine, aspartic acid. (c) Some amino acids such as glycine and cysteine are used as detoxicants of specific substances. (d) Methionine acts as “active” methionine (S-adenosylmethionine) and transfers methyl group to various substances by transmethylation. (e) Cystine and methionine are sources of sulphur. , Glutamine and serine for purin synthesis. 18

� ESSENTIAL � � � AMINO ACIDS Nutritionally, amino acids are of two types: (a) Essential and (b) Non-essential. (c) There is also a third group of semiessential amino acids. (a) Essential amino acids: These are the ones which are not synthesised by the body and must be taken in diet. They include valine, leucine, isoleucine, phenylalanine, threonine, tryptophan, methionine and lysine. For remembering the following formula is used—MATT VIL PHLY. 19

(B) NON-ESSENTIAL AMINO ACIDS: � � � � They can be synthesised by the body and may not be the requisite components of the diet. (c) Semi-essential amino acids: These are growth promoting factors since they are not synthesised insufficient quantity during growth. They include arginine and histidine. They become essential in growing children, pregnancy and lactating women. Occurrence of amino acids: All the standard amino acids mentioned above occur in almost all proteins. Cereals are rich in acidic amino acids Asp and Glu while collagen is rich in basic amino acids and also proline and hydroxy-proline. 20

NEW AMINO ACIDS � � � In addition to 20 L-amino acids that take part in protein synthesis, recently two more new amino acids described. They are: A. Selenocysteine - 21 st amino acids B. Pyrrolysine - 22 nd amino acid A. Selenocysteine It is recently introduced as 21 st amino acid. Selenocysteine occurs at the “active site” of several enzymes. Examples include: • Thioredoxin reductase • Glutathione peroxidase which scavenges peroxides, • De-iodinase that converts thyroxine to tri-iodothyronine • Glycine reductase • Selenoprotein P, a glycoprotein containing 10 selenocysteine residues, found in mammalian blood. It has an antioxidant function and its concentration falls in selenium deficiency. 21

� Selenocysteine arises co-translationally during its incorporation into peptides. The UGA anticodon of the unusual t. RNA designated t. RNAsec, normally signals STOP. � The ability of the protein synthesising apparatus to identify a selenocysteine specific UGA codon involves the selenocysteine insertion element, a stem-loop structure in the untranslated region of the m-RNA. � Selenocysteine-t. RNAsec is first charged with serine by the Ligase that charges t. RNAsec. Subsequent replacement of the serine oxygen by selenium involves seleno-phosphate formed by Selenophosphate synthetase. 22
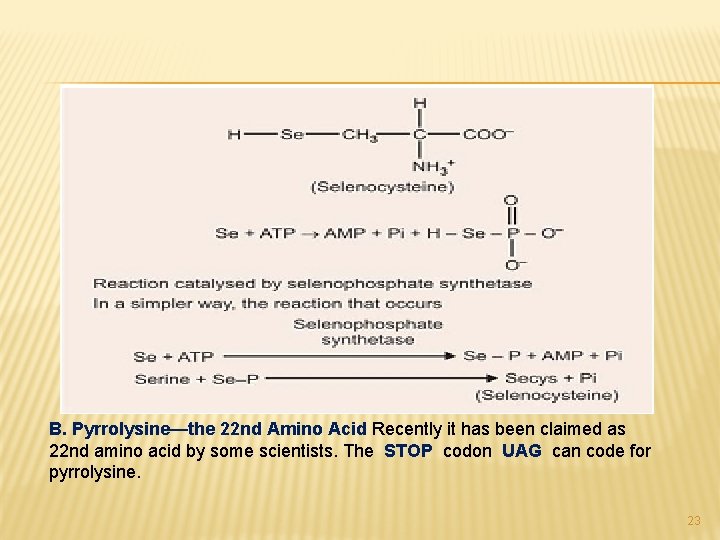
B. Pyrrolysine—the 22 nd Amino Acid Recently it has been claimed as 22 nd amino acid by some scientists. The STOP codon UAG can code for pyrrolysine. 23

� PROPERTIES � A. OF AMINO ACIDS Isomerism: Two types of isomerism are shown by amino acids basically due to the presence of asymmetric carbon atom. Glycine has no asymmetric carbon atom in its structure hence is optically inactive. � � (a) Stereoisomerism: All amino acids except glycine exist in D and L isomers. As described in the chapter on carbo-hydrates it is an absolute configuration. In D-amino acids– NH 2 group is on the right hand while in L-amino acids it is oriented to the left. It is the same orientation of – OH group of the central carbon of glyceraldehyde. 24

� Natural proteins of animals and plants generally contain Lamino acids. D-amino acids occur in bacteria. � (b) Optical Isomerism: All amino acids except glycine � have asymmetric carbon atom. Few amino acids like isoleucine and threonine have an additional asymmetric carbon in their structures. Consequently all but glycine exhibit ‘optical’ activities and rotate the plane of plane polarised light and exist as dextrorotatory (d) or laevorotatory (l) isomers. Optical activity depends on the p. H and side chain. 25

� B. AMPHOTERIC NATURE AND ISOELECTRIC p. H: � The -NH 2 and -COOH groups of amino acids are ionizable groups. Further, charged polar side chains of few amino acids also ionise. Depending on the p. H of the solution these groups act as proton donors (acids) or proton acceptors (bases). This property is called as amphoteric and therefore amino acids are called as ampholytes. At a specific p. H the ami 11 no acid carries both the charges in equal number and exists as dipolar ion or “Zwitterion”. At this point the net charge on it is zero, i. e. positive charges and negative charges on the protein/amino acid molecule equalizes. The p. H at which it occurs without any charge on it is called p. I or isoelectric p. H. On the acidic side of its p. I amino acids exist as a cation by accepting a proton and on alkaline as anion by donating a proton. � � � 26

� C. PHYSICAL PROPERTIES: � They are colourless, crystalline substances, more soluble in water than in polar solvents. � Tyrosine is soluble in hot water. � They have high melting point usually more than 200°C. � They have a high dielectric constant. � They possess a large dipole moment. 27

� D. CHEMICAL PROPERTIES � I. Due to Carboxylic (—COOH) Group 1. Formation of esters: They can form esters with � � alcohols. The COOH group can be esterified with alcohol. Treatment with Na 2 CO 3 solution in cold releases the free ester from ester hydrochloride. 2. Reduction to amino alcohol: This is achieved in presence of lithium aluminium hydride. 3. Formation of amines by decarboxylation: Action of specific amino acid decarboxylases, dry distillation or heating with Ba(OH)2 or with diphenylamine evolves CO 2 from the — COOH group and changes the amino acid into its amine. In vivo, the amino acids can be decarboxylated by the enzyme decarboxylase and forms the corresponding amines. 28
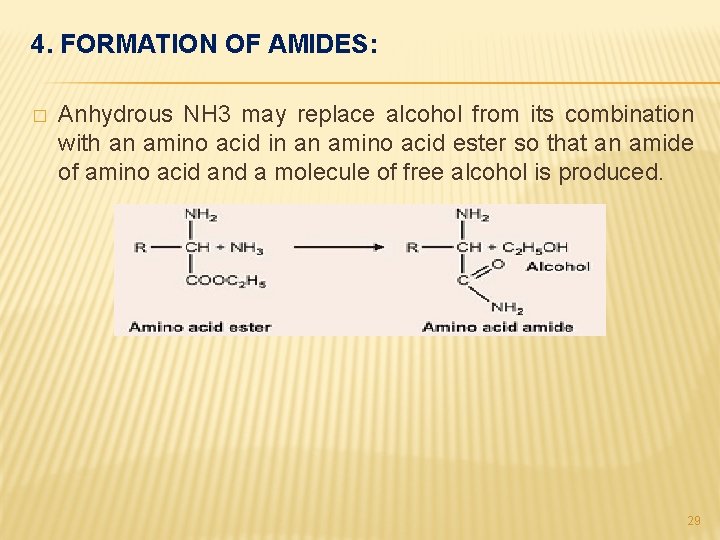
4. FORMATION OF AMIDES: � Anhydrous NH 3 may replace alcohol from its combination with an amino acid in an amino acid ester so that an amide of amino acid and a molecule of free alcohol is produced. 29
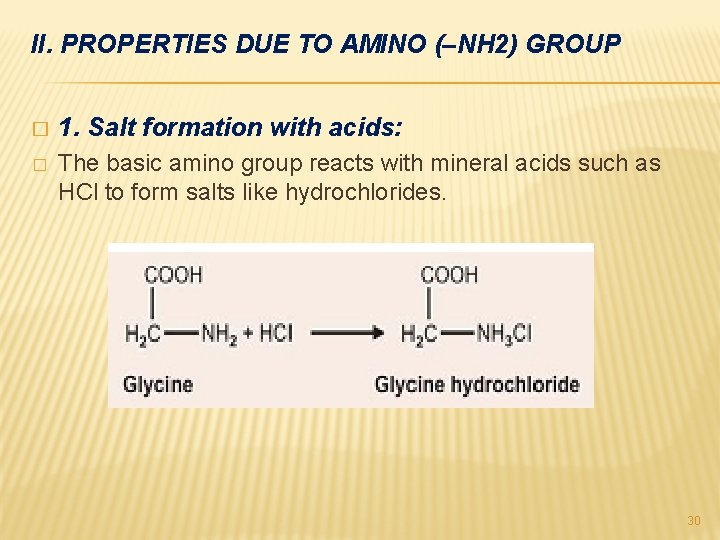
II. PROPERTIES DUE TO AMINO (–NH 2) GROUP � 1. Salt formation with acids: � The basic amino group reacts with mineral acids such as HCl to form salts like hydrochlorides. 30

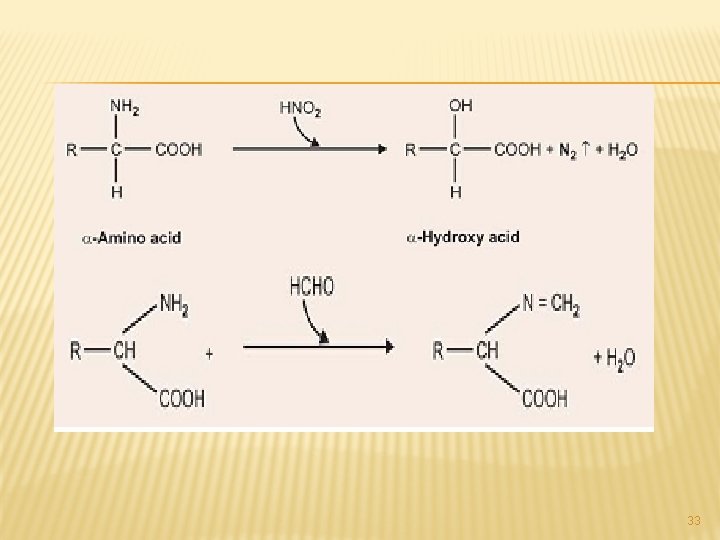
2. FORMATION OF ACYL DERIVATIVES: Amino group reacts with acyl anhydride or acyl halides such as benzoyl chloride and give acyl amino acids like benzoyl glycine (hippuric acid). � Incidentally, this is one of the mechanisms of detoxication in which glycine is used and this also forms the basis of one of the liver function tests. 3. Oxidation: Potassium permanganate or H 2 O 2 oxidises the NH 2 group and converts the amino acid into imino acid which reacts with water to form NH 3 and α-keto- acid. 4. Reaction with HNO 2: Like other primary amines, the amino acids except proline and hydroxyproline react with HNO 2 (nitrous acid) libering N 2 from NH 2 group. � This forms the basis of Van Slyke’s method for determining -NH 2 group (Nitrogen). � 31

5. REACTION WITH CO 2: The amino acid anion present in an alkaline solution may react with CO 2 through NH 2 group to form a carbaminoacid anion. 6. Reaction with formaldehyde: Formaldehyde reacts with the-NH 2 group to form a methylene compound. � Application: Because of the presence of free basic amino group in the amino acid molecule its amount cannot be estimated directly by titration with a standard alkali. � On addition of neutral formaldehyde it combines with the amino group to form either methylene amino acid or dimethylol amino acid. � Both these products are strong acids and may be estimatedby titration with a standard alkali. � This is known as “Sorensen’s” formol titration method. 32 �
33
- Slides: 33