Production of Metabolic Energy Mitochondrial Diseases Oxidation Reduction

Production of Metabolic Energy • Mitochondrial Diseases • Oxidation Reduction Reactions • Electron Transport • Oxidative Phosphorylation W. X. B. 11/26/2020 1

Mitochondrial Diseases • • • W. X. B. Rare genetic diseases with maternal inheritance patterns Etiology is inability to produce ATP Diseases vary in severity with a broad range of clinical symptoms. Tissues most prominently affected are those that require the most energy: – brain – heart – liver – skeletal muscle Presently no cures, but understanding of these disease has progressed to the point where we can manage the pathologies associated with these diseases There are physicians who specialize in treating mitochondria based diseases. • • • Mitochondrial DNA (mt. DNA) codes for 13 mitochondrial proteins, most mt. DNA proteins are functionally involved in aerobic mitochondrial ATP production. mt. DNA is easily mutated and poorly repaired causing rapid accumulation of mutations Because mitochondria are essential to aerobic production of ATP mutations in mt. DNA can be catastrophic to a cell. High oxygen consuming tissues do so because of their large numbers of mitochondria Mitochondria produce high levels of ATP needed to support their physiological function. The most prominent of the oxygen using tissues are brain, heart liver kidney and skeletal muscle. 11/26/2020 2

Selected Mitochondrial Diseases • Mytochondrial Myopathies – Postviral Fatigue Syndrome – MERRF- Myoclonic epilepsy with ragged red fibers • Neurodegenerative Diseases – – – W. X. B. Glutamate Toxicity: Leber Hereditary Optic Neuropathy (LHON) Parkinsons Syndrome 11/26/2020 3
Hypothyroid Function and Mitochondrial Insufficiency W. X. B. 11/26/2020 4
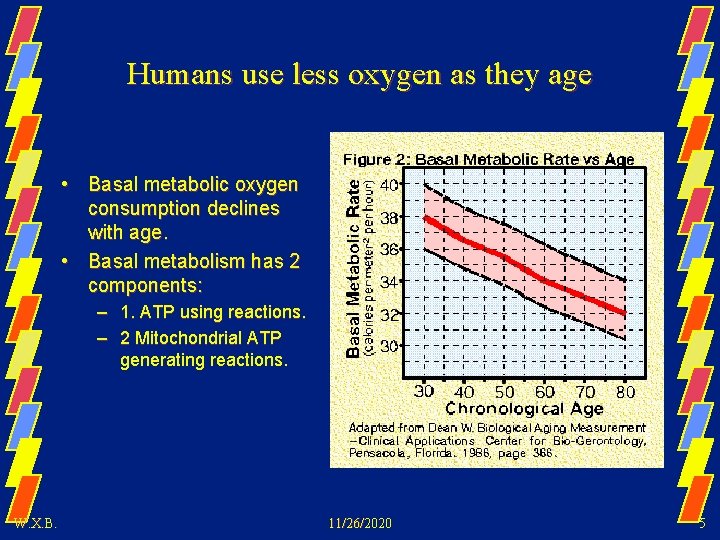
Humans use less oxygen as they age • Basal metabolic oxygen consumption declines with age. • Basal metabolism has 2 components: – 1. ATP using reactions. – 2 Mitochondrial ATP generating reactions. W. X. B. 11/26/2020 5

Lactic Acidosis in Mitochondrial Insufficiency • Inhibited e- transport causes high mito NADH and an lack of mito ATP. • High mito NADH inhibits shuttle activity, but low ATP stimulates glycolysis. • Result is production of glycolytic ATP and high cytosolic NADH. • Continuation of glycolysis with ATP and NADH production leads to conversion of pyruvate to lactate and ultimately lactic acidosis W. X. B. 11/26/2020 6
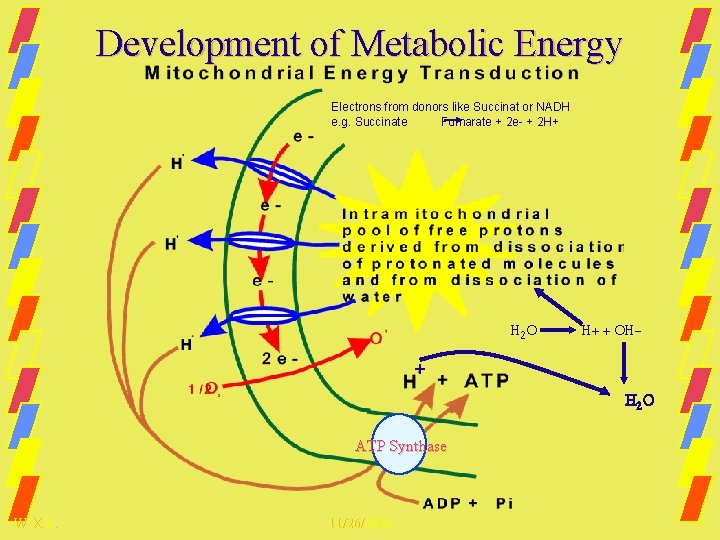
Development of Metabolic Energy Electrons from donors like Succinat or NADH e. g. Succinate Fumarate + 2 e- + 2 H+ H 2 O H+ + OH- + H 2 O ATP Synthase W. X. B. 11/26/2020 7
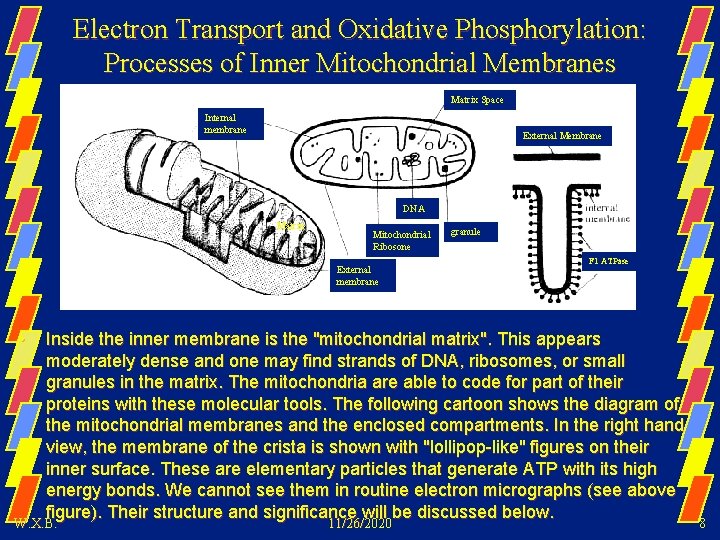
Electron Transport and Oxidative Phosphorylation: Processes of Inner Mitochondrial Membranes Matrix Space Internal membrane External Membrane DNA Matrix Mitochondrial Ribosone External membrane • granule F 1 ATPase Inside the inner membrane is the "mitochondrial matrix". This appears moderately dense and one may find strands of DNA, ribosomes, or small granules in the matrix. The mitochondria are able to code for part of their proteins with these molecular tools. The following cartoon shows the diagram of the mitochondrial membranes and the enclosed compartments. In the right hand view, the membrane of the crista is shown with "lollipop-like" figures on their inner surface. These are elementary particles that generate ATP with its high energy bonds. We cannot see them in routine electron micrographs (see above figure). Their structure and significance will be discussed below. W. X. B. 11/26/2020 8

Mitochondria: Structural Characteristics • W. X. B. 11/26/2020 Mitochondria are the cells' power sources. They are distinct organelles with two membranes. Usually they are rod-shaped, however they can be round. The outer membrane limits the organelle. The inner membrane is thrown into folds or shelves that project inward. These are called "cristae mitochondriales". from Fawcett, A Textbook of Histology, Chapman and Hall, 12 th edition, 1994. 9

Oxidation Reduction Half Cells W. X. B. 11/26/2020 10

The Standard H 2 1/2 Cell is a Reference Standard W. X. B. 11/26/2020 11

Free Energy of NADH Oxidation • Conventions: – – electric potential is in volts n = # of electrons F = Faraday 23 kcal/volt/mol algebraic differences are: • (e- acceptor) - (e- donor) • Calculations – – – W. X. B. Standard Free Energy ( DGo’) is about -52. 6 kcal/mol. Std. Free Energy of ATP hydrolysis is about -7. 3 kcal/mol 3 ATPs are made per NADH oxidized Efficiency of energy conversion is about 42%! Actual available Free Energy depends on molar concentrations of reactants and products. 11/26/2020 12
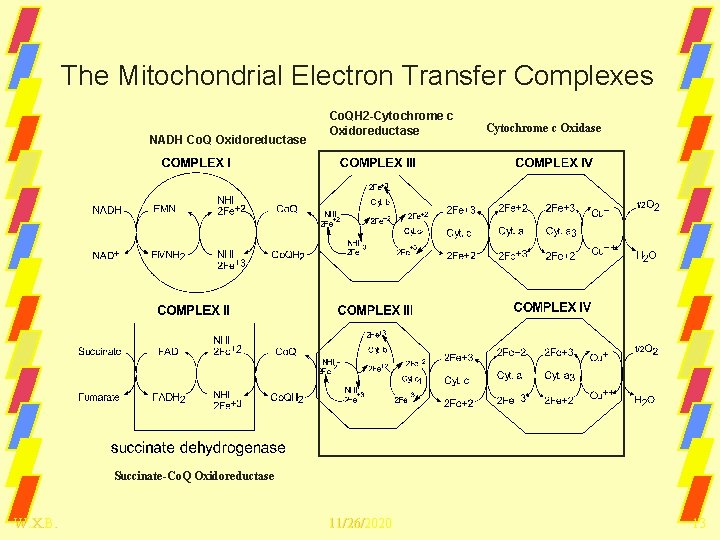
The Mitochondrial Electron Transfer Complexes NADH Co. Q Oxidoreductase Co. QH 2 -Cytochrome c Oxidoreductase Cytochrome c Oxidase Succinate-Co. Q Oxidoreductase W. X. B. 11/26/2020 13
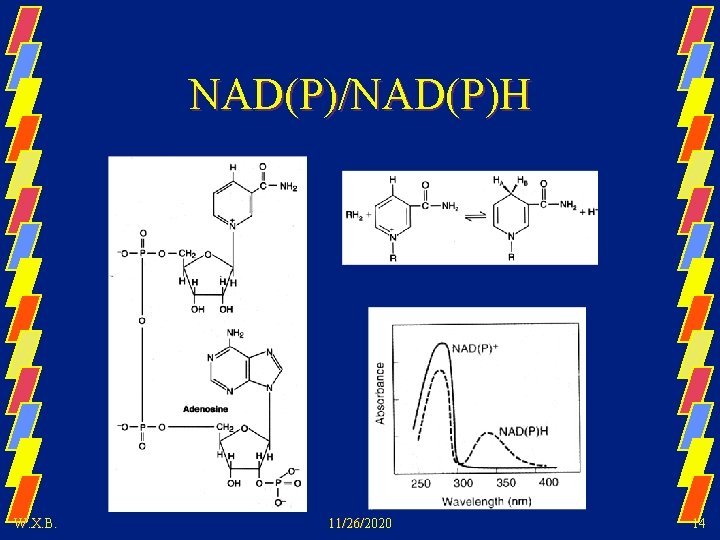
NAD(P)/NAD(P)H W. X. B. 11/26/2020 14
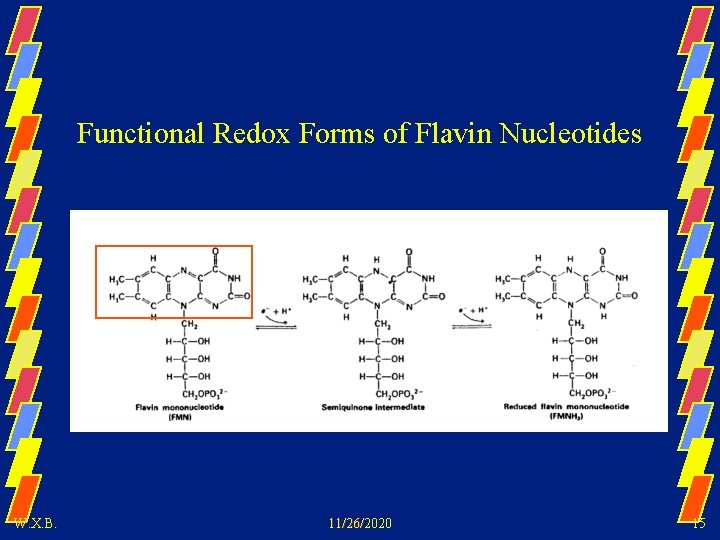
Functional Redox Forms of Flavin Nucleotides W. X. B. 11/26/2020 15
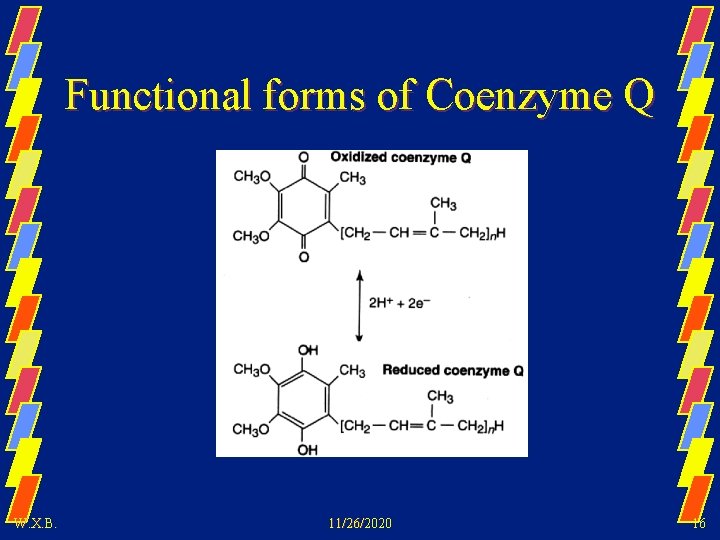
Functional forms of Coenzyme Q W. X. B. 11/26/2020 16
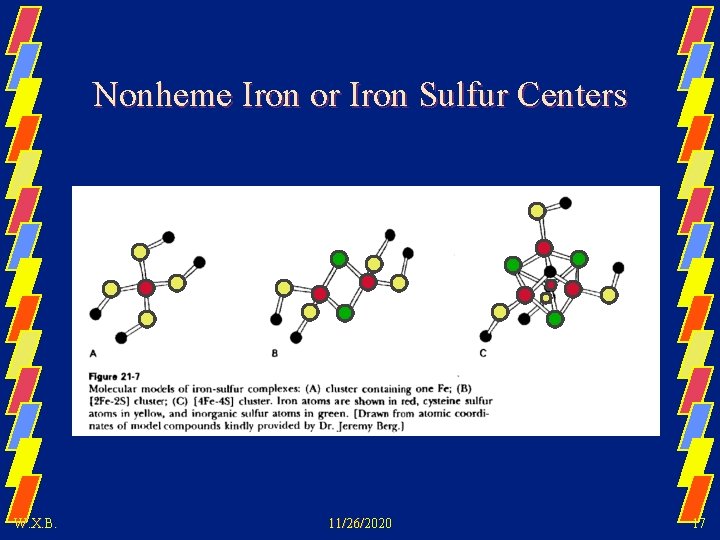
Nonheme Iron or Iron Sulfur Centers W. X. B. 11/26/2020 17
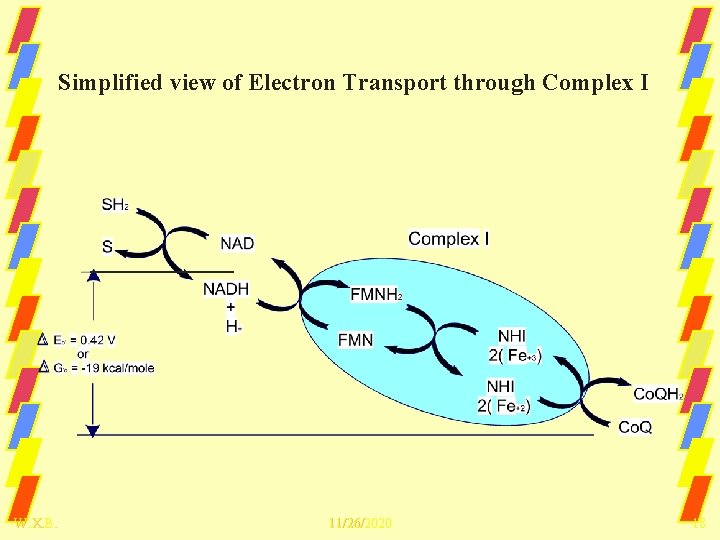
Simplified view of Electron Transport through Complex I W. X. B. 11/26/2020 18
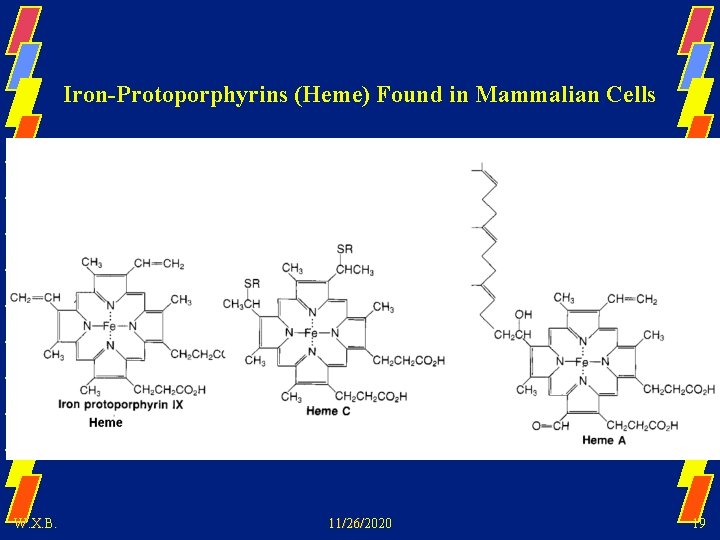
Iron-Protoporphyrins (Heme) Found in Mammalian Cells W. X. B. 11/26/2020 19
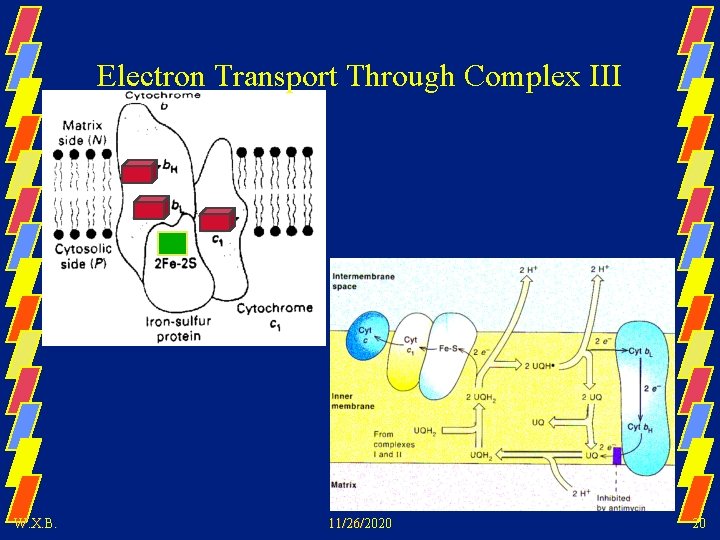
Electron Transport Through Complex III W. X. B. 11/26/2020 20
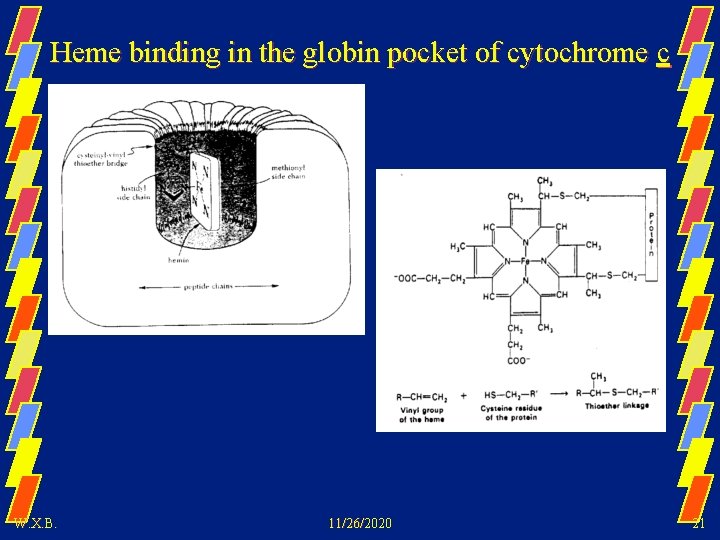
Heme binding in the globin pocket of cytochrome c W. X. B. 11/26/2020 21
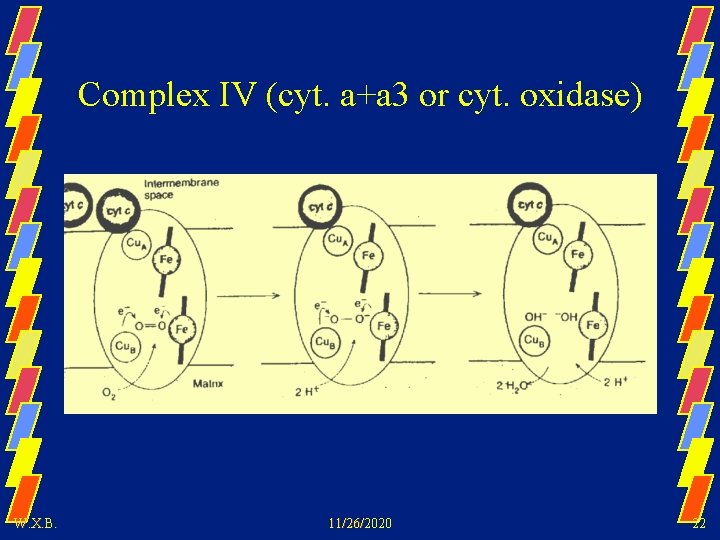
Complex IV (cyt. a+a 3 or cyt. oxidase) W. X. B. 11/26/2020 22
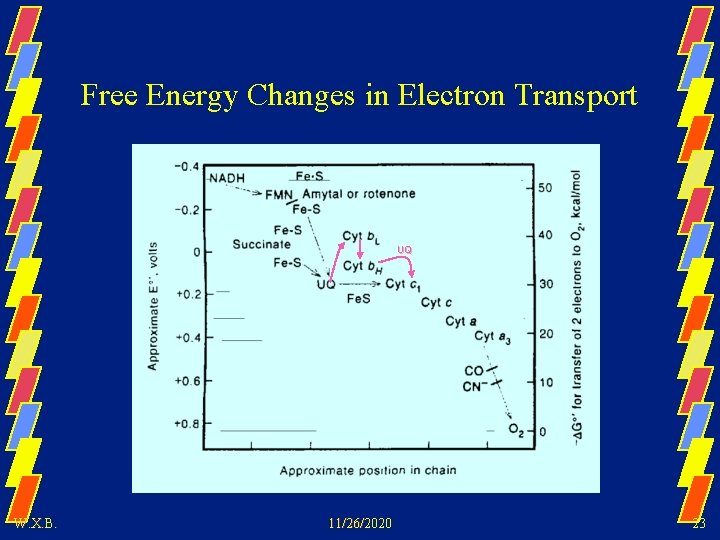
Free Energy Changes in Electron Transport UQ W. X. B. 11/26/2020 23
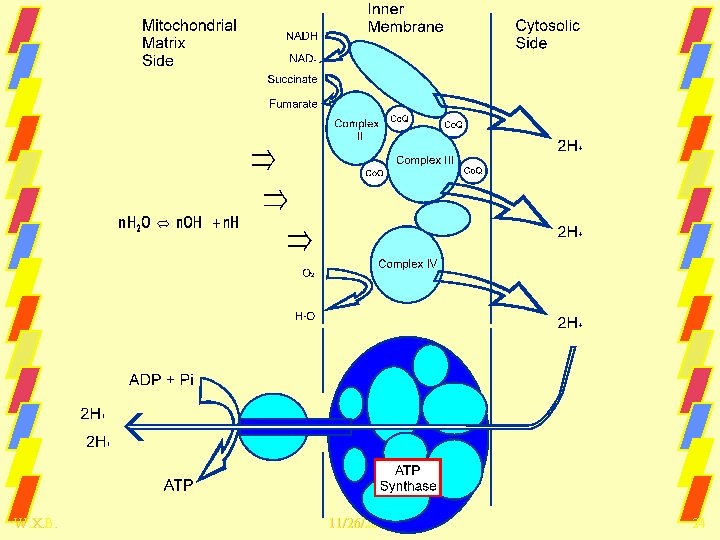
W. X. B. 11/26/2020 24
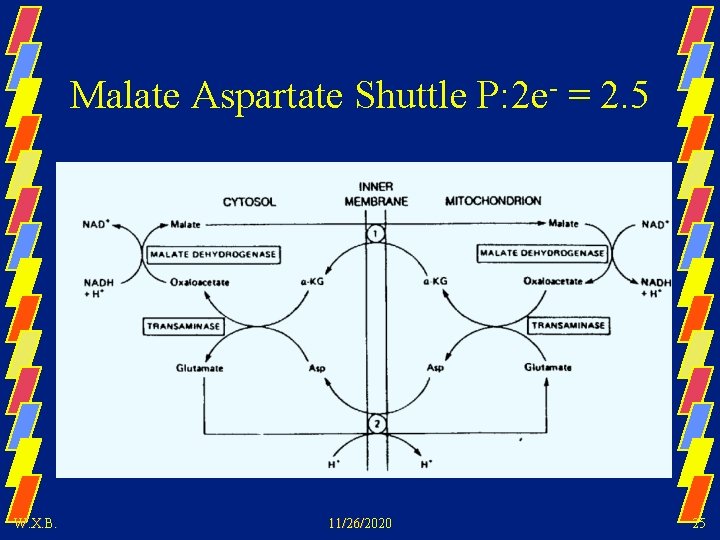
Malate Aspartate Shuttle P: 2 e- = 2. 5 W. X. B. 11/26/2020 25
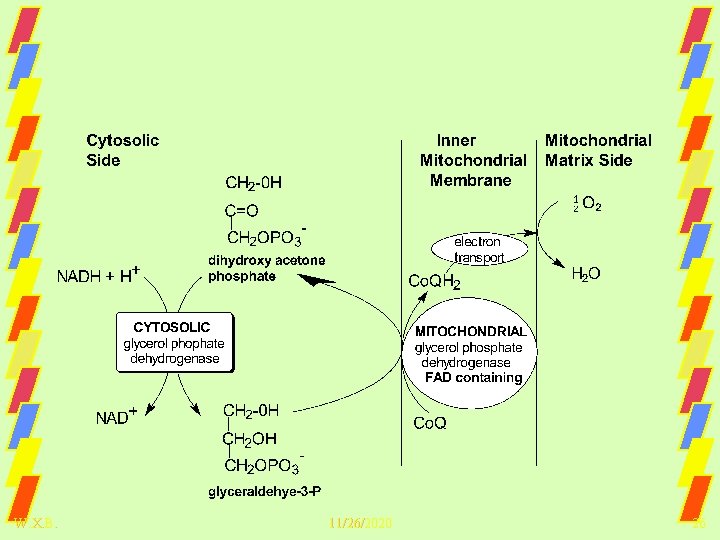
Glycerol Phosphate Shuttle, P: 2 e- = 1. 5 W. X. B. 11/26/2020 26
- Slides: 26