Production of Bauxite Residue with Low Soda and

Production of Bauxite Residue with Low Soda and High Iron Content: the ILTD Process Option György (George) Bánvölgyi Senior Process Consultant Budapest, Hungary 1

Outlines of the presentation • Principal uses of BR, drawbacks that impede • Issues with the Conventional Low Temp. Dig. • Comprehensive kinetic model for the reactions of gibbsite and kaolinite • Improved Low Temperature Digestion concept • Laboratory and bench scale tests • Composition results for BR of Trombetas bx • Recent achievements for the use or BR 2

The use of bauxite residue (BR, red mud) has been desired from the start • Karl Joseph Bayer claimed in his German Patent (1892): • “The red, iron-containing residue that occurs after digestion settles well and, with sufficient practice, can be filtered and washed. For its high iron and low alumina content it can be processed in an appropriate manner or smelted together with other iron ores for iron. ” 3

The use of bauxite residue, /cont. • Since the invention of the Bayer Process - over 1200 patents have been obtained, - hundreds of technically successful trials have been executed aiming at the use of BR. • However, some 3 % of the BR is being used out of the annually more than 150 million ton BR produced. • All over the world nearly 4 billion tons of BR (as dry matter) have been accumulated so far. • K. E. Evans, J. Sustain. Metall. (2016) 2, 316 -331 4

Principal uses of BR • Cement: 500, 000 – 1, 500, 000 tonnnes • Raw material/additive in iron and steel production: 200, 000 – 1, 500, 000 tonnnes • Roads, landfill capping, soil amelioration: • 200, 000 – 500, 000 tonnnes • Construction materials (bricks, tiles, ceramics): 100, 000 – 300, 000 tonnes • Other (refractory, adsorbent, acid mine drainage, catalyst): 300, 000 tonnes • Altogether: 2 -4. 5 million tonnes • K. E. Evans, J. Sustain. Metall (2016) 2, 316 -331 5

Drawbacks that impede the use of BR • “So far no process has been a commercial success, because of specific problems associated with the bauxite residue such as - low iron content, - high alkali content, - fineness of particles, - moisture. ” C. R. Borra et al. : J. Sustain. Metall. (2016) 2, 365 -386 6

Iron (oxide) content of BRs and iron ores • Iron oxide in different BRs is in the range of 5 to 60% Fe 2 O 3. This amounts to 3 to 42% Fe. The average is 38. 4% Fe 2 O 3 (26. 9% Fe). • In the iron metallurgy iron ores with 55 to 70% Fe content are preferred. In some countries low grade iron ore concentrates with 48 -50 % Fe content are being used. 7

Na 2 O content of BR • The chemically combined soda content of BR varies between 0. 7% and 12. 3% Na 2 O (average 5. 5%). In addition the soluble content is in the range of 0. 2% to 0. 6% Na 2 O. • The Na 2 O evaporates in the high temperature region of the blast furnaces, deposits in the colder region and attacks the refractory bricks. • It is deemed that an Na 2 O content of ≤ 3% is acceptable for most potential users of BR. 8

Moisture and soluble Na 2 O contents of dewatering options 9

Conventional BR red mud disposal in pond 10

Issues with the Conventional Low Temperature Digestion (CLTD) 1 of 2 • the digester reactor is designed for the crystallization of the dissolved reactive silica as DSP and not for the „complete” dissolution of gibbsite • a part of the digestion capacity of the Test Tank Liquor is not utilized in order to prevent excessive formation of boehmite • all the reactive silica is converted to DSP • the dissolved silica content in the digestion effluent can not be maintained close to the equilibrium, therefore

Issues with the Conventional Low Temperature Digestion (CLTD) 2 of 2 • usually excessive silica scalings occur in the liquor preheaters (double stream digestion) • the hydrate product is contaminated with silica • the A/C ratio maintained in the liquor to precipitation is restricted, thereby the liquor productivity is less than it could be • significant alumina losses (can be about 5 -7% if both pre-desilication and LT digestion are applied) due to hydrothermal formation of boehmite and/or pseudoboehmite out of gibbsite
 Basic reactions, reactive OH- concentr. § Dissolution reaction of gibbsite Al(OH)3 + OH- [Al(OH)4](1)](http://slidetodoc.com/presentation_image/7863652cec7060cb534db8dff42f7319/image-13.jpg)
Basic reactions, reactive OH- concentr. § Dissolution reaction of gibbsite Al(OH)3 + OH- [Al(OH)4](1) § Dissolution reaction of kaolinite Al 2(OH)4(Si 2 O 5) + 6 OH- + H 2 O 2[Si. O 2(OH)2]2 - + 2[Al(OH)4](2) § Reactive OH- concentration AH=2 x 17 [(c. Al 2 O 3 eq -c. Al 2 O 3)/102 – c. Si. O 2/60] § DSP formation: 2[Si. O 2(OH)2]2 - + 2[Al(OH)4]- + 2 Na+ Na 2 O. Al 2 O 3. 2 Si. O 2(2+k)H 2 O + 4 OH- + (2 -k)H 2 O (3) k = 0… 2, formula valid at about 100 o. C temperature
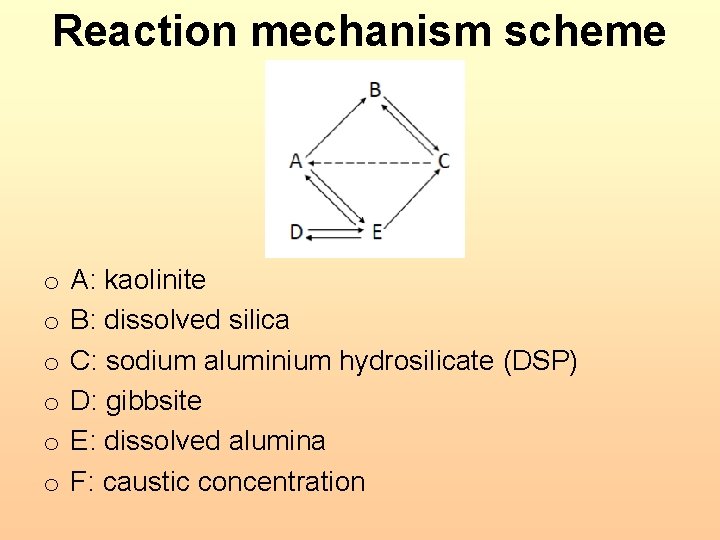
Reaction mechanism scheme o o o A: kaolinite B: dissolved silica C: sodium aluminium hydrosilicate (DSP) D: gibbsite E: dissolved alumina F: caustic concentration

Kinetic rate equations • r 1 = AH*(k 1*c. A – k 2*c. C/c 2 F) dissolution of kaolinite (Si. O 2) • r 2 = k 3*(c. B-ceq. B) formation of sodium aluminium hydrosilicate as DSP (Si. O 2) • r 3 = k 4*AH*c. D dissolution of gibbsite (Al 2 O 3) Reactive OH- concentration AH=2 x 17 [(c. Al 2 O 3 eq -c. Al 2 O 3)/102 – c. Si. O 2/60

Rate equations for the components
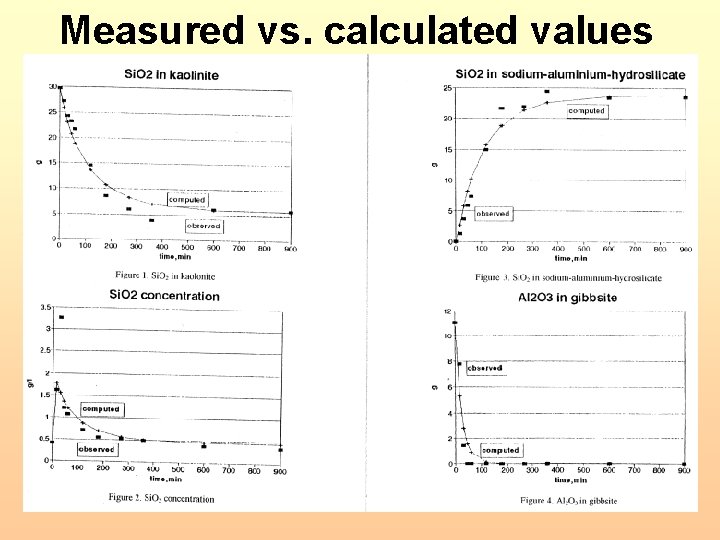
Measured vs. calculated values
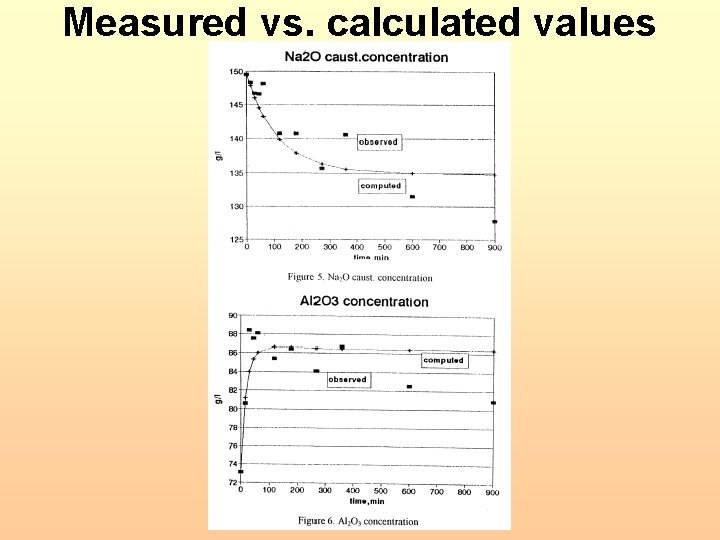
Measured vs. calculated values
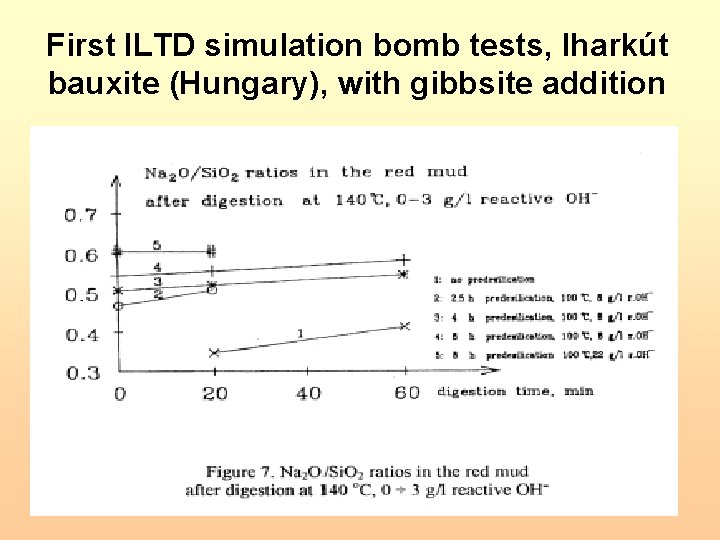
First ILTD simulation bomb tests, Iharkút bauxite (Hungary), with gibbsite addition
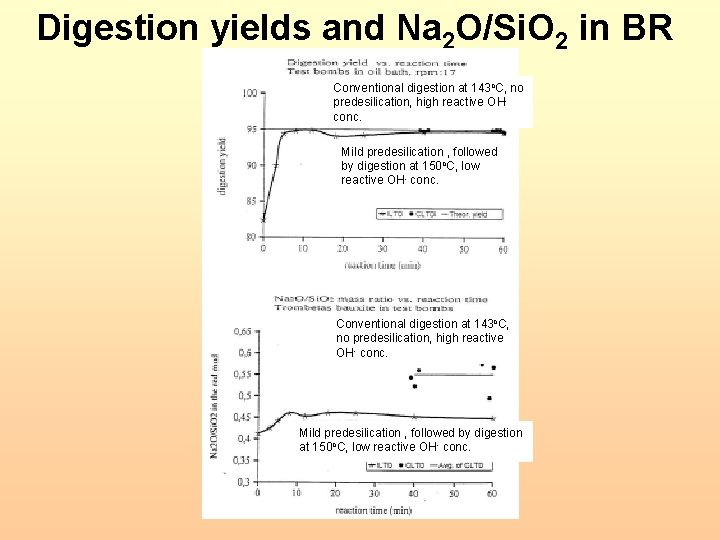
Digestion yields and Na 2 O/Si. O 2 in BR Conventional digestion at 143 o. C, no predesilication, high reactive OH- conc. Mild predesilication , followed by digestion at 150 o. C, low reactive OH- conc. Conventional digestion at 143 o. C, no predesilication, high reactive OH- conc. Mild predesilication , followed by digestion at 150 o. C, low reactive OH- conc.
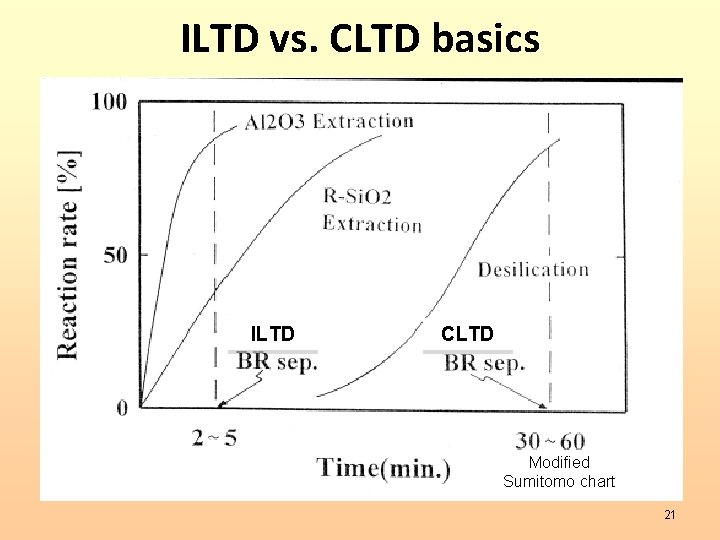
ILTD vs. CLTD basics ILTD CLTD Modified Sumitomo chart 21

ILTD bench scale tests Trombetas bauxite Two-stream digestion 150 o. C digestion temperature (130 -150 o. C) Achieved A/C ratio >. 83 (higher than equilibrium A/C ratios of Rosenberg and Healy) • Repeated digestion tests • Fairly vigorous agitation (rpm 200/min) • • 22
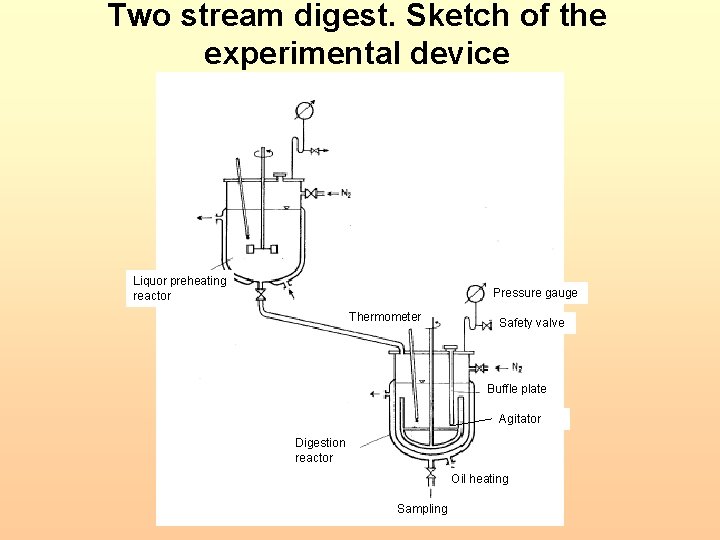
Two stream digest. Sketch of the experimental device Liquor preheating reactor Pressure gauge Thermometer Safety valve Buffle plate Agitator Digestion reactor Oil heating Sampling

A/C ratio vs. reaction time 24
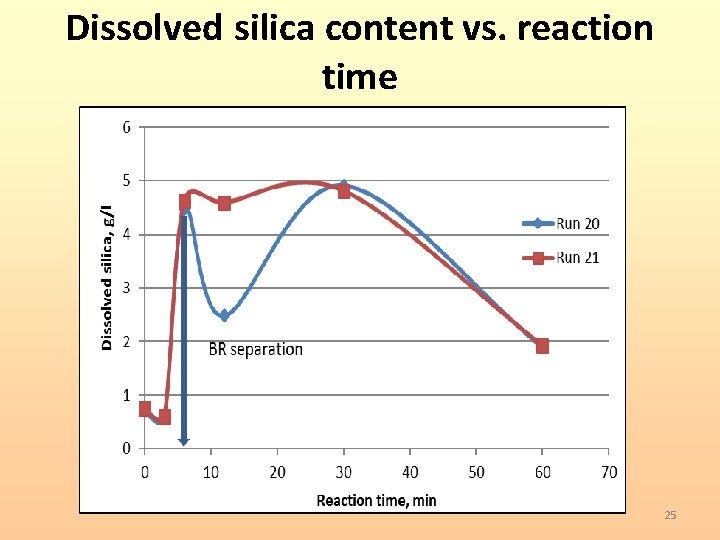
Dissolved silica content vs. reaction time 25
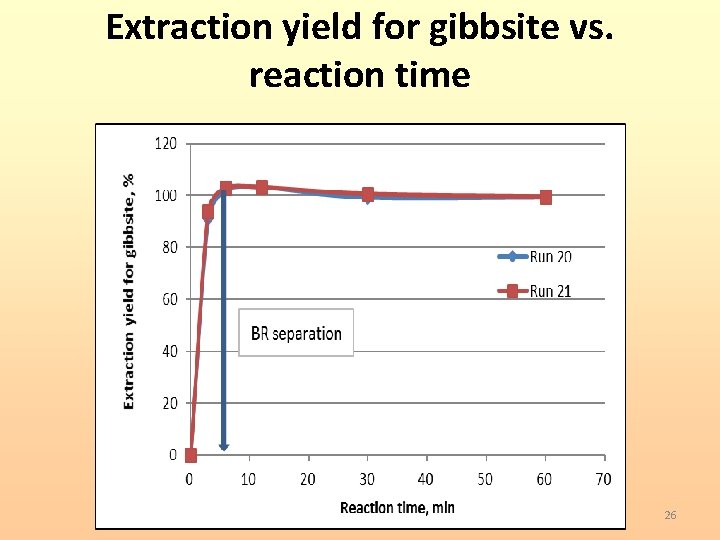
Extraction yield for gibbsite vs. reaction time 26
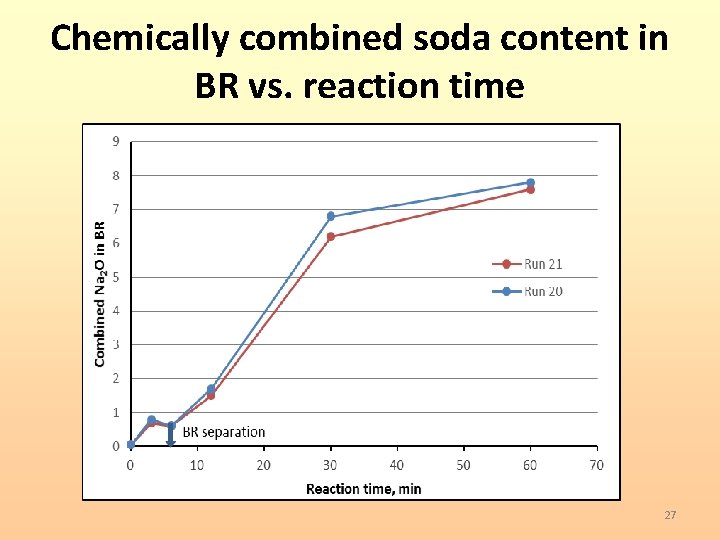
Chemically combined soda content in BR vs. reaction time 27
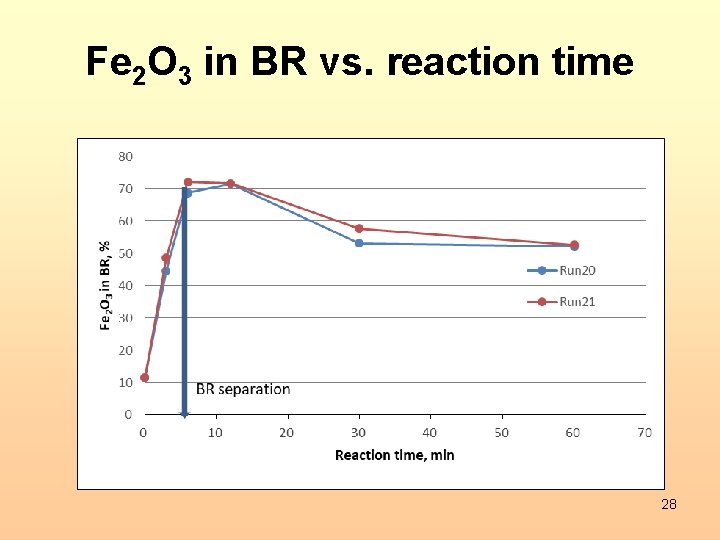
Fe 2 O 3 in BR vs. reaction time 28
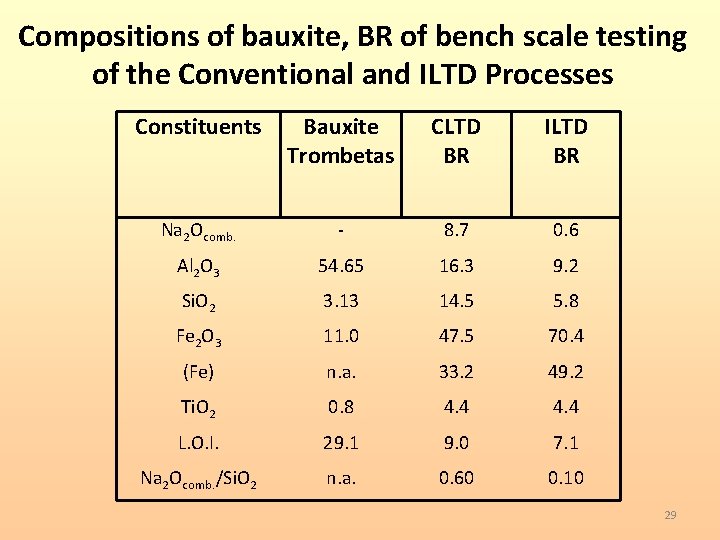
Compositions of bauxite, BR of bench scale testing of the Conventional and ILTD Processes Constituents Bauxite Trombetas CLTD BR ILTD BR Na 2 Ocomb. - 8. 7 0. 6 Al 2 O 3 54. 65 16. 3 9. 2 Si. O 2 3. 13 14. 5 5. 8 Fe 2 O 3 11. 0 47. 5 70. 4 (Fe) n. a. 33. 2 49. 2 Ti. O 2 0. 8 4. 4 L. O. I. 29. 1 9. 0 7. 1 Na 2 Ocomb. /Si. O 2 n. a. 0. 60 0. 10 29

ILTD Process, block flow diagram 30

ILTD Process, block flow diagram 31

Improved Low Temperature Digestion (ILTD) Process Principal features: ü no predesilication is needed ü high A/C ratio, short digestion time (3 -5 min) ü separation of the low soda high iron bauxite residue just after the digestion preferably with Hi-Bar Filtration and/or pressure decanter ü seeded pressure crystallization of the Desilication Product (DSP) out of the liquid phase. ü separation of the digestion reactor from the crystallization of DSP ü material and energy costs savings: 15 -18% 32

Potential of the ILTD Process • The global bauxite reserves and resources, including the potential resources, was estimated to be as much as 70 billion metric tons. • 90% of the total bauxite reserves/resources are estimated to be of gibbsitic ore. The gibbsitic bauxites are suitable feedstock for the low temperature digestion variant of the Bayer process. • G. Komlóssy, ICSOBA conference, 2010, and personal communication

Business potential of the ILTD Process • Refineries, where Low Temperature (or atmospheric) Digestion for processing gibbsitic bauxites is applied or anticipated. • Market potential: moderately estimated over 30 MTPY in about 20 alumina refineries • The savings potential is USD 35 -40/talumina • Business Potential, altogether: 30 MTPY x USD 35/ta = USD 1050 million/y • A Concept Study can justify each of the individual cases, capacity increase can possibly be considered.
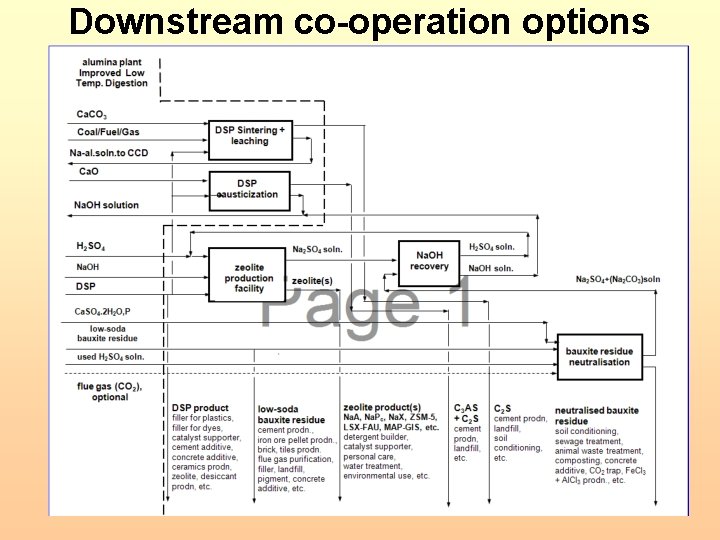
Downstream co-operation options

Present status of the ILTD Process ü Economic viability – proven ü All major steps on a commercial scale – proven ü Process concept – matured for commercialization, widely accepted ü Pilot scale demonstration – outstanding

BR from plate and frame filter presses (mainly diasporic bauxite, Ao. G) 37

BR of a gibbsitic bauxite from a Hi-Bar continuous pressure steam filter Courtesy of Bokela

Issues, state of the art, achievements Issue State of the art Required Demonstrated Low iron content, Fe 5 -42% (avg. 26. 9%) >48 -50% (pref. >55%) 49% (ILTD bench scale tests) High alkali content, Na 2 O 0. 7 -12. 3% (avg. 5. 5%) < 3% Na 2 O 0. 6% Na 2 O (ILTD bench scale tests) Fineness of particles lumpy Moisture content ~ 28 -32% < 30 -32% 25 -30% Hi-Bar Filter and pressure leaf filter

Thank you for your attention! Questions, comments are welcome. 40

György (George) Bánvölgyi’s Bio • Name: György (George) Bánvölgyi • Degrees and date earned: – MSc, Chemical Engineering, Veszprém Univ. , Hungary 1972 – Patent Attorney, Budapest Univ. of Economy, Hungary, 1992 • Affiliation: Bán-Völgy Ltd, Budapest, Hungary • Present position: – Technical Director, Senior Process Consultant • Work experience: – Due diligences, Feasibility Studies, design projects – Research and development projects – Principal inventor and driver of the Improved Low Temperature Digestion (ILTD) Process
- Slides: 41