Product Isolation Concentration Part II Ultrafiltration Membrane based

Product Isolation & Concentration: Part II Ultrafiltration

Membrane based Bioseparation • A membrane is a thin semi permeable and selective barrier that permits the separation of certain species in a fluid by combination of sieving and diffusion mechanisms such as: 1. 2. 3. 4. Particle-liquid separation Particle-solute separation Solute-solvent separation Solute-solute separation Figure 1: Diffusion driven separation. 2

Membrane based Bioseparation • Among many applications are; product concentration, product sterilization (i. e. removal of bacteria and virus particles), solute fractionation, solute removal from solutions (e. g. , desalination, demineralization), purification, and clarification. • Factors, which are considered in in membrane based separation, are: i. Solute size ii. Electrostatic charge iii. Diffusivity iv. Solute shape 3

Membrane Processes Four common types of membranes: 1. 2. 3. 4. Reverse Osmosis Nanofiltration Ultrafiltration Microfiltration 4
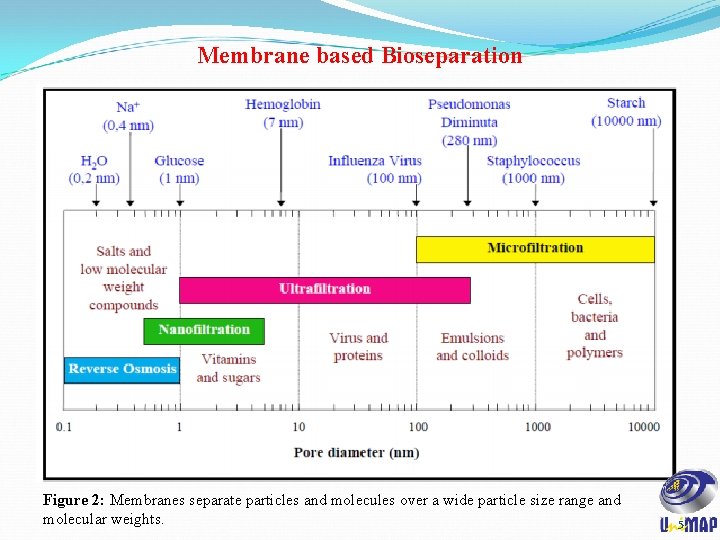
Membrane based Bioseparation Figure 2: Membranes separate particles and molecules over a wide particle size range and molecular weights. 5
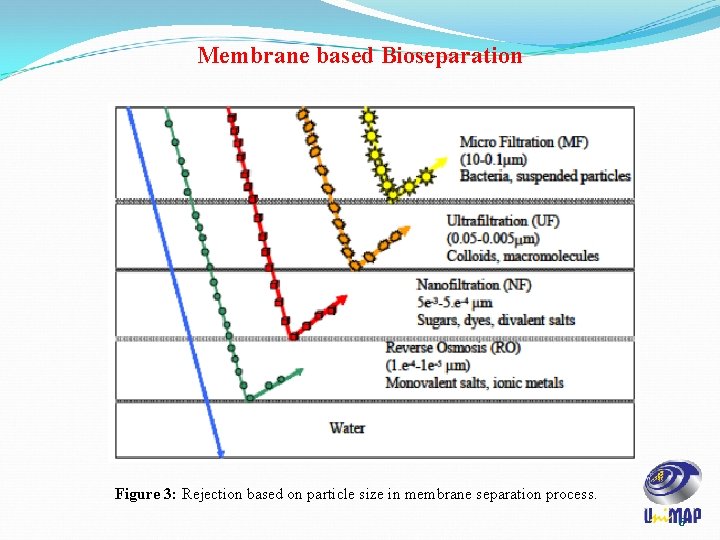
Membrane based Bioseparation Figure 3: Rejection based on particle size in membrane separation process. 6
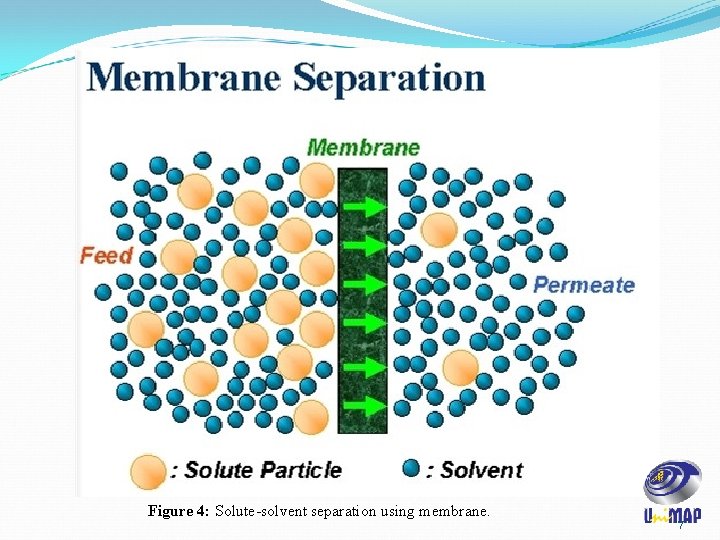
Figure 4: Solute-solvent separation using membrane. 7

Membrane based Bioseparation Membranes are broadly divided into two groups: I. Symmetric (or isotropic) II. Asymmetric (or anisotropic) Figure 5: Principal types of membranes 8
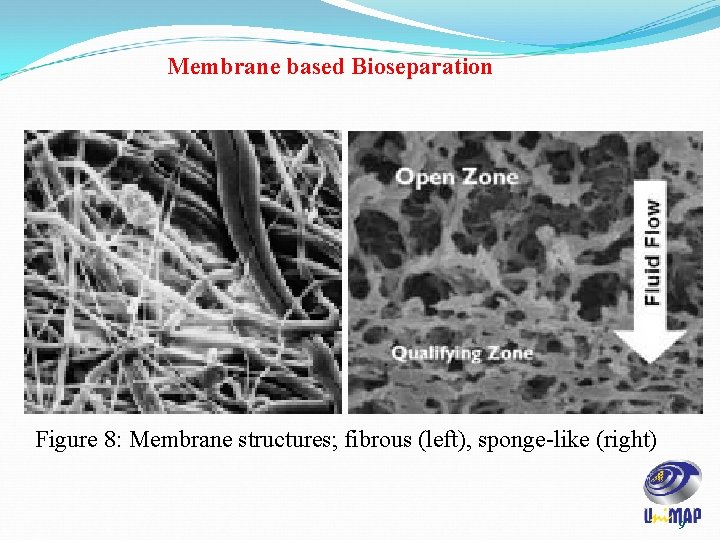
Membrane based Bioseparation Figure 8: Membrane structures; fibrous (left), sponge-like (right) 9

Membrane based Bioseparation Figure 6: Illustration of conventional membrane filtration. 10

Membrane based Bioseparation Figure 7: Illustration of cross-flow membrane filtration. 11
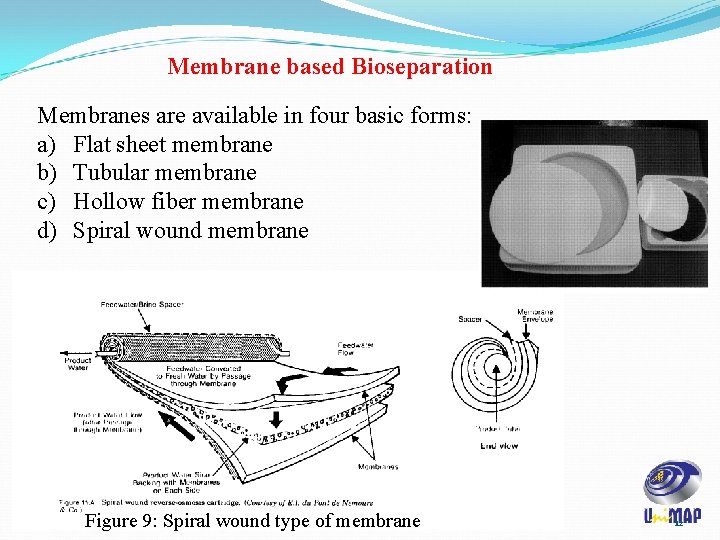
Membrane based Bioseparation Membranes are available in four basic forms: a) Flat sheet membrane b) Tubular membrane c) Hollow fiber membrane d) Spiral wound membrane Figure 9: Spiral wound type of membrane 12
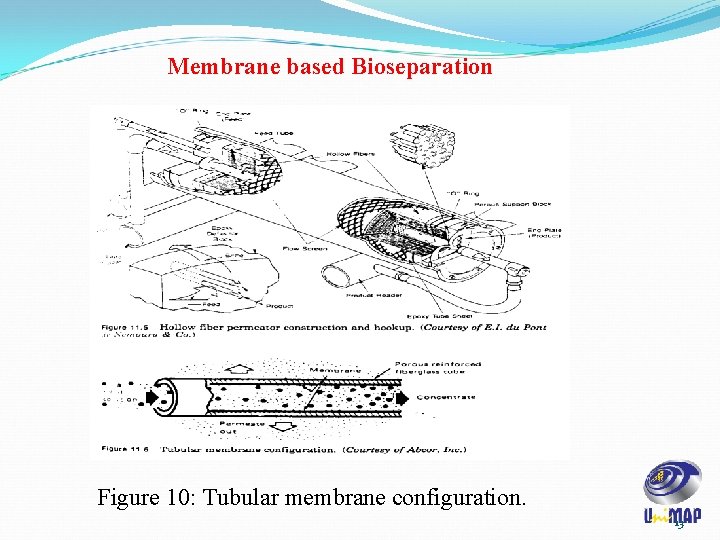
Membrane based Bioseparation Figure 10: Tubular membrane configuration. 13
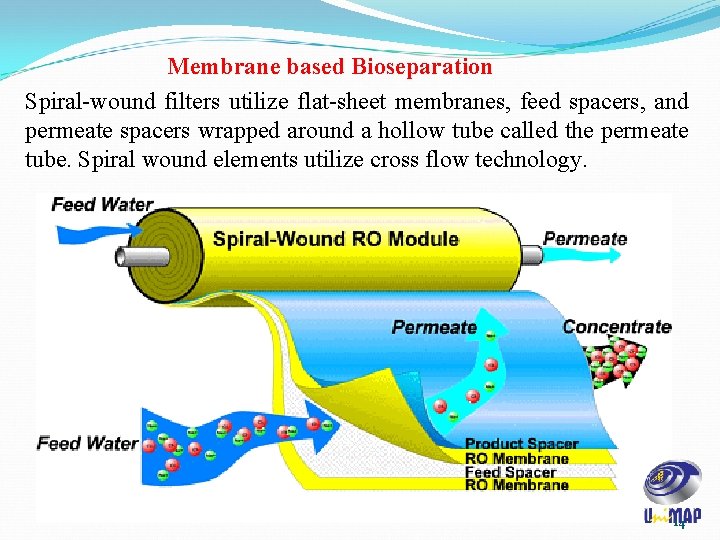
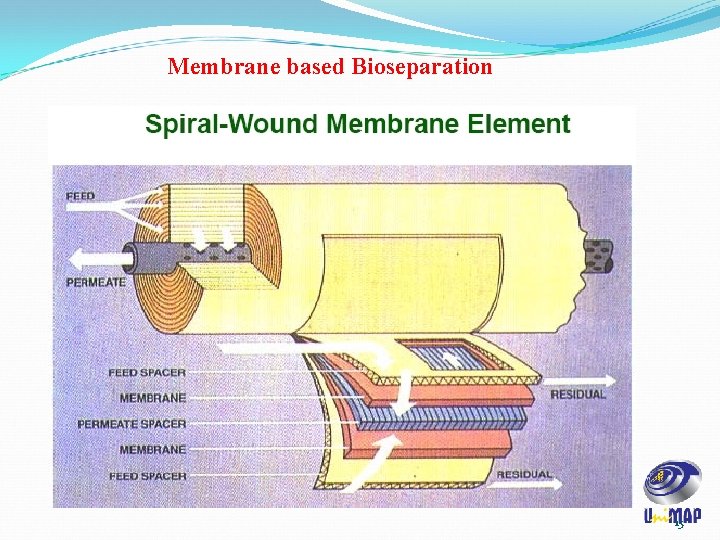
Membrane based Bioseparation Spiral-wound filters utilize flat-sheet membranes, feed spacers, and permeate spacers wrapped around a hollow tube called the permeate tube. Spiral wound elements utilize cross flow technology. 14
Membrane based Bioseparation 15

Membrane based Bioseparation 16

Membrane based Bioseparation Ceramic Membrane Elements 17

Membrane based Bioseparation 18

Membrane based Bioseparation 19

Membrane based Bioseparation 20

Membrane based Bioseparation Spiral UF system 21

Membrane based Bioseparation • The performance of a membrane depends to a large extent on the properties of the membrane such as: 1. Mechanical strength e. g. , tensile strength, bursting pressure 2. Chemical resistance e. g. , p. H range, solvent compatibility 3. Permeability to different species e. g. , pure water permeability, sieving coefficient 4. Average porosity and pore size distribution • Pressure requirements are based on osmotic pressure for R. O. , osmotic pressure and fluid mechanical frictional headloss (straining) for nanofiltration, and purely fluid mechanical frictional headloss (straining) for ultra- and microfiltration. 22

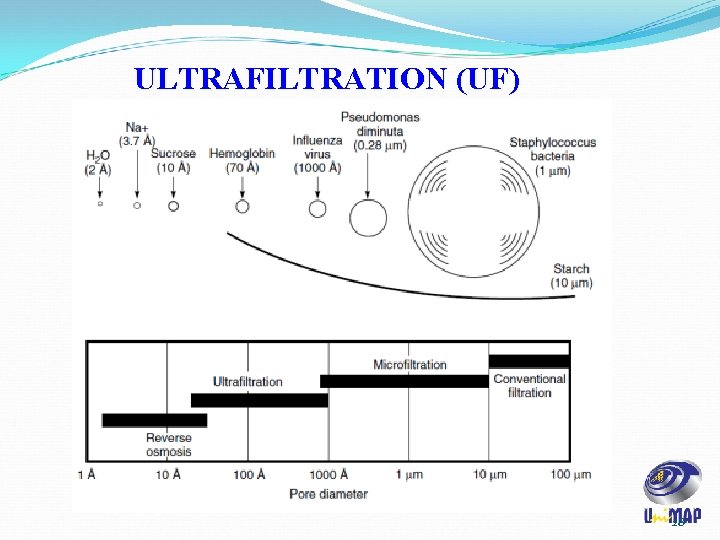
ULTRAFILTRATION (UF) ü Ultrafiltration (UF) membranes retain macromolecules such as proteins while allowing smaller molecules to pass through. ü UF is used to: a) Separate large molecules from solvents b) Separate large molecules from smaller molecules c) Separate large molecules from one another ü The primary separation mechanism in UF is size exclusion. ü However, physicochemical interactions between the solutes and the membrane, and operating conditions can influence the process significantly. 23

ULTRAFILTRATION (UF) Applications of Ultrafiltration: ü Conventional water treatment (replace all processes except disinfection). ü Pretreat water for R. O and nanofiltration. ü Iron/Manganese removal (after oxidation). ü Concentration of solutes ü Purification of solvents ü Fractionation of solutes ü Clarification UF is attractive because of the high throughput of product, low cost and ease of scale-up 24

ULTRAFILTRATION (UF) Applications of Ultrafiltration in Bioprocess: ü Purification of proteins and nucleic acids ü Concentration of macromolecules ü Desalting i. e. removal of salt and other low molecular weight compounds from solution of macromolecules ü Virus removal from therapeutic products 25

ULTRAFILTRATION (UF) ü UF membranes: i. separates solute in a molecular weight range of 5 k. Da to 500 k. Da. ii. Pores ranging from 1 to 20 nm in diameter. iii. Membranes are often anisotropic, with a thin ‘skin layer’ fused on top of a microporous backing layer. iv. Skin confers selectivity to membrane while the microporous backing layer provides mechanical support. v. The ability of an UF membrane to retain macromolecules is traditionally specified in terms of molecular weight cut-off (MWCO). 26
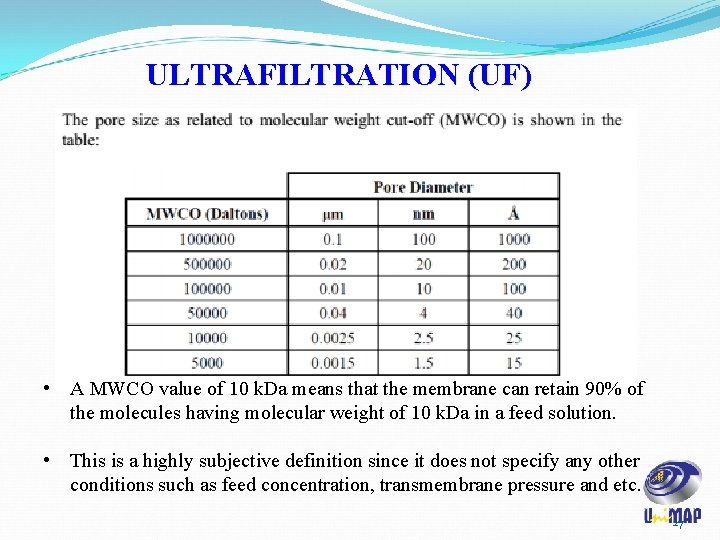
ULTRAFILTRATION (UF) • A MWCO value of 10 k. Da means that the membrane can retain 90% of the molecules having molecular weight of 10 k. Da in a feed solution. • This is a highly subjective definition since it does not specify any other conditions such as feed concentration, transmembrane pressure and etc. 27
ULTRAFILTRATION (UF) 28

Operating pressure ranges: R. O. /NF: 80 – 600 psig MF/UF: 5 – 60 psig 29
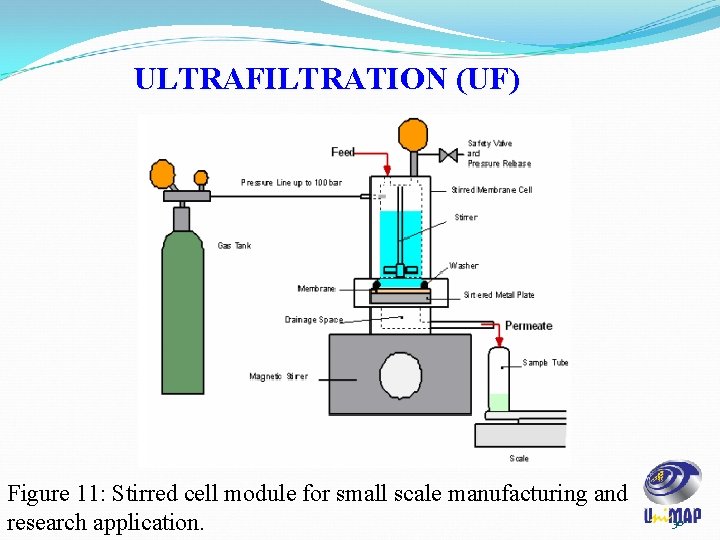
ULTRAFILTRATION (UF) Figure 11: Stirred cell module for small scale manufacturing and research application. 30
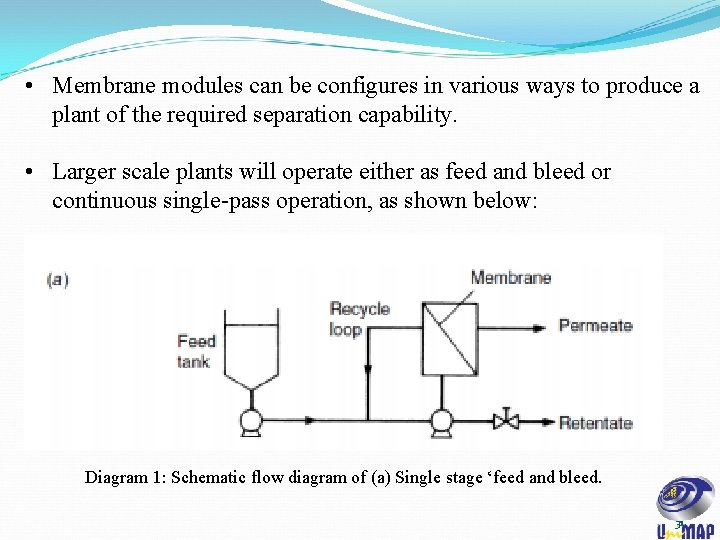
• Membrane modules can be configures in various ways to produce a plant of the required separation capability. • Larger scale plants will operate either as feed and bleed or continuous single-pass operation, as shown below: Diagram 1: Schematic flow diagram of (a) Single stage ‘feed and bleed. 31
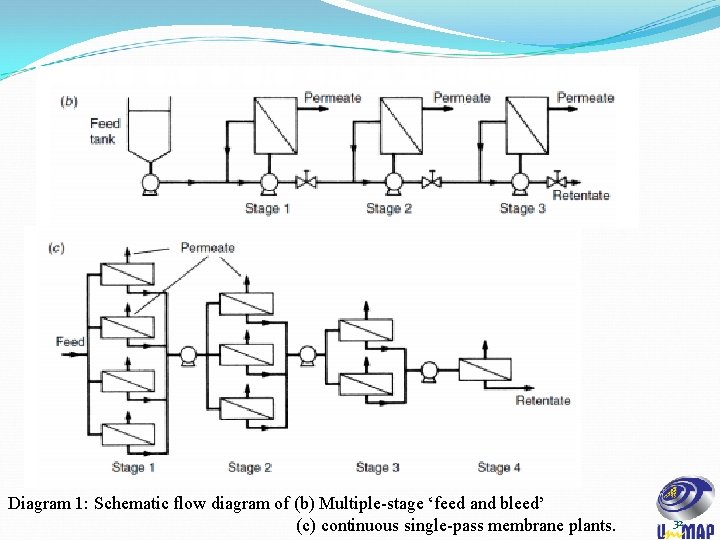
Diagram 1: Schematic flow diagram of (b) Multiple-stage ‘feed and bleed’ (c) continuous single-pass membrane plants. 32

ULTRAFILTRATION (UF) The liquid flow through a pore (q): The flux, or flow per unit membrane area, is the sum of all the flows through the individual pores (N pores): For membranes of equal pore area and porosity ( ), the number of pores per square centimetre is proportional to the inverse square of the pore diameter: 33

ULTRAFILTRATION (UF) The flow of a solvent through UF membranes can be described in terms of a pore flow model which assumes ideal cylindrical pores aligned normal to the membrane surface: Jv dp lp = volumetric permeate flux (m 3/m 2 s or m/s) = membrane porosity (-) = average pore diameter (m) = transmembrane pressure (Pa) = viscosity (kg/m. s) = average pore length (m) 34

ULTRAFILTRATION (UF) The transmembrane pressure (TMP) in cross-flow UF is given by: where: Pi = inlet pressures on the feed side (Pa) Po = outlet pressures on the feed side (Pa) Pp = pressure on the permeate side (Pa) *the TMP in UF is usually generated by pressurizing the feed side with a valve on the retentate line. 35

ULTRAFILTRATION (UF) • Permeate flux in UF determines its productivity. • Permeate flux depends on: i) the properties of the membrane and the feed solution ii) transmembrane pressure iii) the solute mass transfer coefficient (which affects the concentration polarization) iv) membrane fouling (permeate flux decreases with time due to fouling) • High permeate flux in UF is important. • This can be done by controlling the extent of concentration polarization and membrane fouling. 36
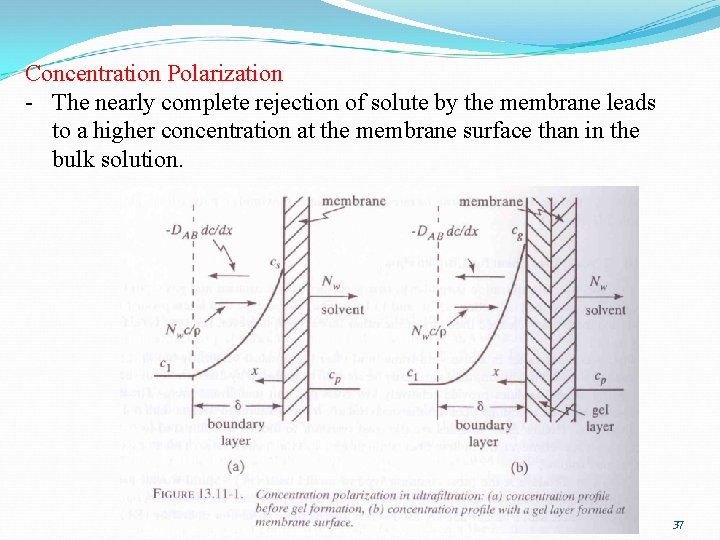
Concentration Polarization - The nearly complete rejection of solute by the membrane leads to a higher concentration at the membrane surface than in the bulk solution. 37

Membrane Fouling of membranes due to accumulation of solute/particulates at the membrane interface has to be addressed for economic reasons. The membranes are too expensive to be replaced for reasons of fouling. 38
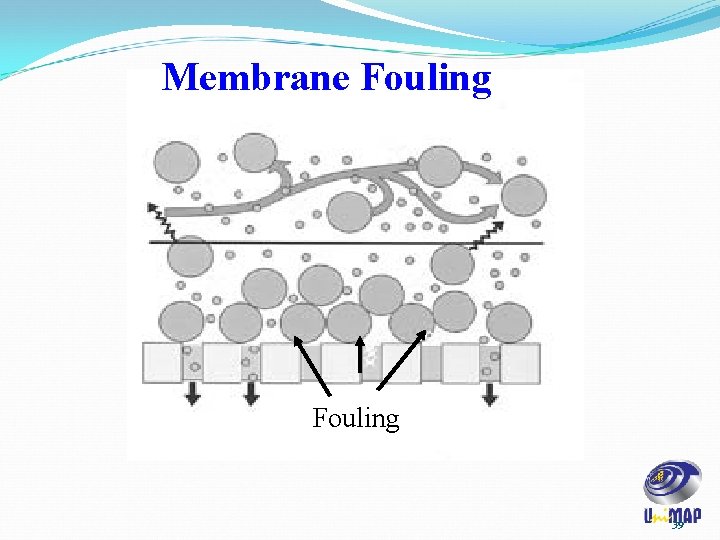
Membrane Fouling 39

Fouling issue • Traditional membrane technology is generally affected by fouling. This long-term loss in throughput capacity is due primarily to the formation of a boundary layer that builds up naturally on the membrane surface during the filtration process. • In addition to cutting down on the flux performance of the membrane, this boundary or gel layer acts as a secondary membrane reducing the native design selectivity of the membrane in use. This inability to handle the buildup of solids has also limited the use of membranes to low-solids feed streams. 40

Membrane Fouling There are various ways to reduce this fouling such as: • Periodic pulsing of feed • Periodic pulsing filtrate (backwashing) • Increasing shear at by rotating membrane • Vibrating membrane (VSEP technology, next slide) 41
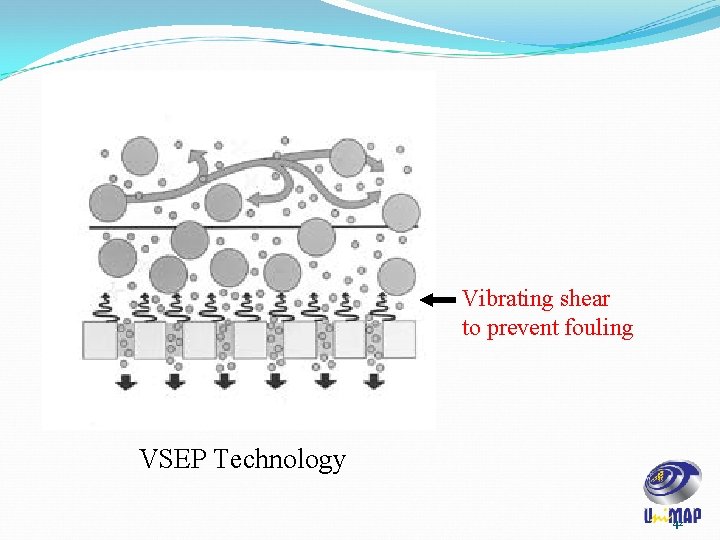
Vibrating shear to prevent fouling VSEP Technology 42
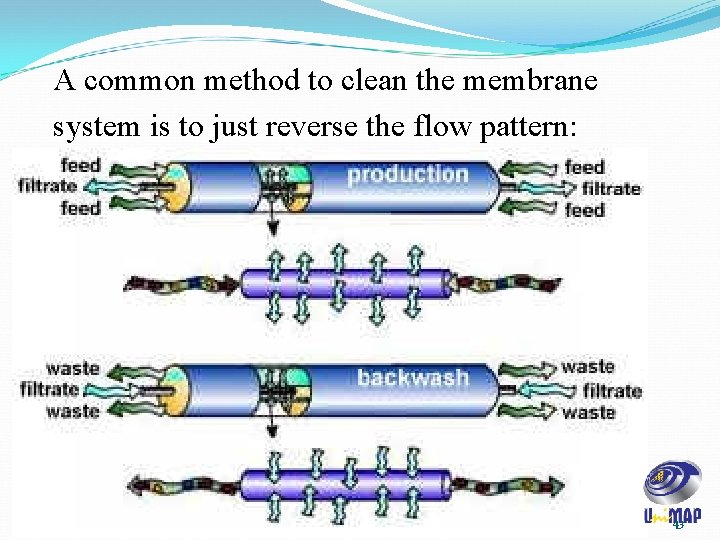
A common method to clean the membrane system is to just reverse the flow pattern: 43

GREEN TECHNOLOGY Membrane Processes are becoming popular because they are considered “Green” technology - no chemicals are used in the process. 44
- Slides: 44