Processes and Process Variables Processes and Process Variables

Processes and Process Variables

Processes and Process Variables

Processes and Process Variables

Processes and Process Variables average molecular weight = Component Mol Fraction, yi Mi N 2 0. 579 28 O 2 0. 263 32 CO 2 0. 158 44 Total 1. 0 Mav=31. 6 Average molecular weight = 31. 6 ECB 3013: Material and Energy Balances Ir. Abdul Aziz Omar 4


Density v The density of a substance is its mass per unit volume. The symbol most often used for density is ρ. v Mathematically, density is defined as mass divided by volume: where ρ is the density, m is the mass, and v is the volume.

Examples 1: 1) Calculate the density of an object with a mass of 7. 2 g and a volume of 24 cm 3. 2) Calculate the density of an object with a mass of 45. 4 lb and a volume of 0. 125 cm 3. 1. 0 lb = 454 g WE KNOW HOW TO SOLVE IT

Examples 2: 1) Calculate the density of an object with a mass of 0. 072 kg and a volume of 24 cm 3. 2) Calculate the density (in kg/m 3) of an object with a mass of 2. 5 lb and a volume of 0. 125 m 3. 1. 0 lb = 454 g

Example 3: Determine the volume of wood has a density of 0. 6 g/cm 3 and a mass of 1. 2 g.

Class Work 3. 1 1) Calculate the density of an object with a mass of 0. 1 lb and a volume of 24 cm 3. 2) Determine the volume of wood has a density of 0. 6 g/cm 3 and a mass of 120 g.

Specific volume Example 1: Calculate the density and specific volume of an object with a mass of 60 g and a volume of 2. 5 cm 3.

Specific volume Example 2: Calculate the volume and specific volume for 20 g of n-propyl alcohol. The density of n-propyl alcohol is 0. 8 g/cm 3.


Specific gravity Example 1: If the density of iron is 7. 85 g/cm 3, calculate the specific gravity of iron. (Water density (@ 4 °C) is 1000 kg/m 3 )

Example 2: If dibromopentane (DBP) has a specific gravity of 1. 57, what is the density in: (a) g/cm 3 (b) lbm/ft 3 and (c) kg/m 3






Moles & Concentrations v Mole is a unit of measurement used in chemistry to express amounts of a chemical substance, defined as the amount of any substance that contains as many elementary entities, as there atoms in 12 grams of pure carbon-12 (12 C) the isotope of carbon with relative atomic mass 12. v A mole is simply a unit of measurement. Units are invented when existing units are inadequate. Chemical reactions often take place at levels where using grams wouldn't make sense, yet using absolute numbers of atoms/molecules/ions would be confusing, too.



Mole and Mass Fraction mole fraction = (moles of a species)/(total moles) mass fraction = (mass of a species)/(total mass) = weight fraction ECB 3013: Material and Energy Balances Ir. Abdul Aziz Omar 24
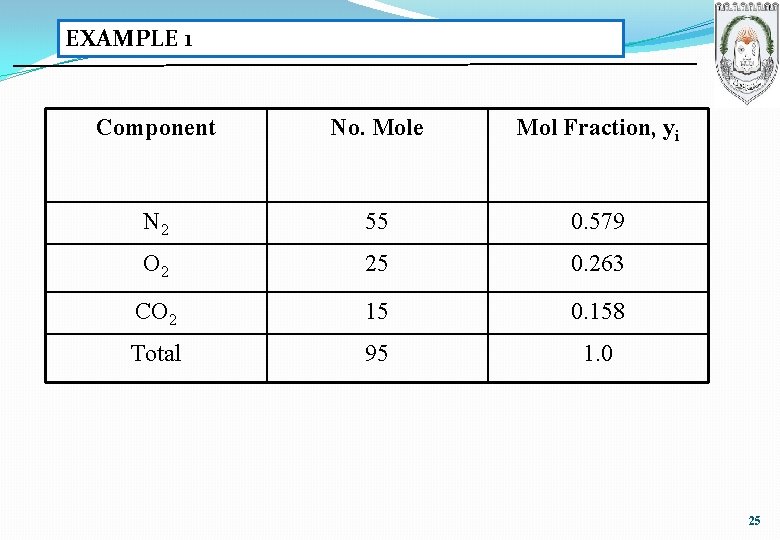
EXAMPLE 1 Component No. Mole Mol Fraction, yi N 2 55 0. 579 O 2 25 0. 263 CO 2 15 0. 158 Total 95 1. 0 25

EXAMPLE 2 A mixture of gases has the following composition by mass: O 2 CO CO 2 N 2 16% 4. 0% 17% 63% (x. O 2=0. 16 g O 2/g total) What is the molar composition? Solution Basis: 100 g of the mixture
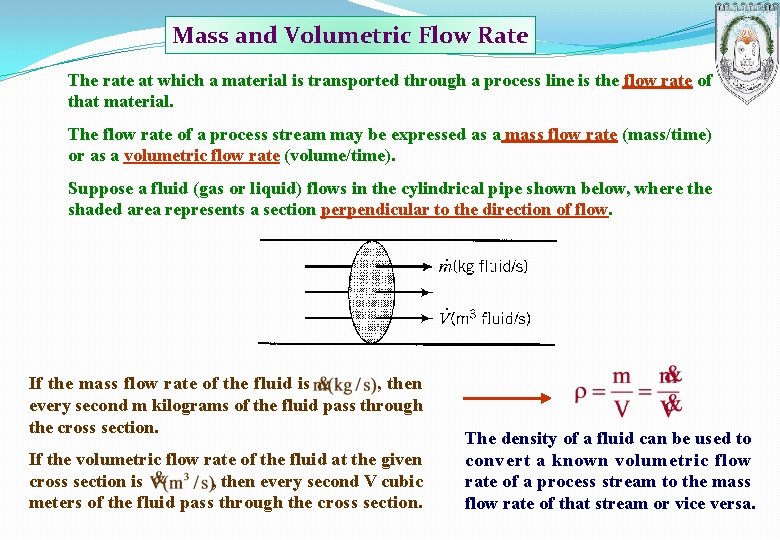
Mass and Volumetric Flow Rate The rate at which a material is transported through a process line is the flow rate of that material. The flow rate of a process stream may be expressed as a mass flow rate (mass/time) or as a volumetric flow rate (volume/time). Suppose a fluid (gas or liquid) flows in the cylindrical pipe shown below, where the shaded area represents a section perpendicular to the direction of flow. If the mass flow rate of the fluid is , then every second m kilograms of the fluid pass through the cross section. If the volumetric flow rate of the fluid at the given cross section is , then every second V cubic meters of the fluid pass through the cross section. The density of a fluid can be used to convert a known volumetric flow rate of a process stream to the mass flow rate of that stream or vice versa.
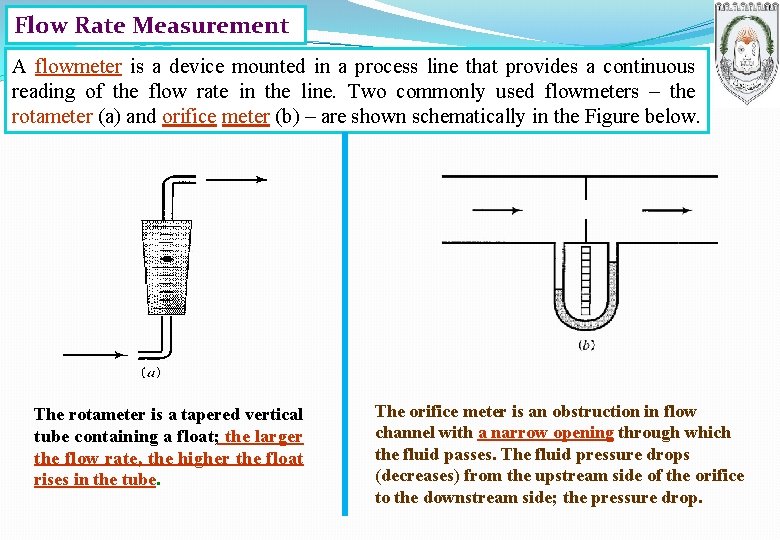
Flow Rate Measurement A flowmeter is a device mounted in a process line that provides a continuous reading of the flow rate in the line. Two commonly used flowmeters – the rotameter (a) and orifice meter (b) – are shown schematically in the Figure below. The rotameter is a tapered vertical tube containing a float; the larger the flow rate, the higher the float rises in the tube. The orifice meter is an obstruction in flow channel with a narrow opening through which the fluid passes. The fluid pressure drops (decreases) from the upstream side of the orifice to the downstream side; the pressure drop.

Flow Rate �Mass flow rate �Mass/time �Volumetric flow rate �Volume/time

Example 1 A solution contains 15% A by mass (x. A= 0. 15) and 20 mole% B (y. B = 0. 20). 1. Calculate the mass of A in 175 kg of the solution. 175 kg solution 0. 15 kg A kg solution = 26 kg A 2. Calculate the mass flow rate of A in a stream of solution flowing at a rate of 53 lbm/h. 53 lbm h 0. 15 lbm A lbm = 8. 0 lbm A/h If a mass or molar unit such as lbm in 53 lbm/h – is not followed by the name of a species, the unit should be understood to refer to the total mixture or solution rather than to a specific component. 3. Calculate the molar flow rate of B in a stream flowing at a rate of 1000 mol/min. 1000 mol min 0. 20 mol B mol = 200 mol B/min

4. Calculate the total solution flow rate that corresponds to a molar flow rate of 28 kmol B/s. 28 kmol B s 1 kmol solution 0. 20 kmol B = 140 kmol solution/s 5. Calculate the mass of the solution that contains 300 lbm of A. 300 lbm A 1 lbm solution 0. 15 lbm A 28 kmol B X = 2000 lbm solution 0. 2 kmol B 1. 0 kmol solution

Concentrations

Concentration There are quantities describe concentrations: 1) Mass concentration: 2) Molar concentration: 3) Number concentration:

Concentration 4) Part Per Million (ppm): Usually describes the concentration of very dilute substances like body fluid (blood):

Example 1: A solution contains Cu 2+ ions at a concentration of 3× 10 -4 M. What is the Cu 2+ concentration in ppm. Solution: Concentration of Cu 2+ ions = 3 × 10 -4 M = 3 × 10 -4 mol/L Mwt of Cu 2+ = 63. 55 g/mol Concentration × Mwt = 3 × 10 -4 mol/L × 63. 55 g/mol = 1. 9 × 10 -2 g/L 1 ppm = mg/L

Example 2: Calculate the moles of Cu. SO 4 in 250 m. L of 0. 02 M copper sulfate solution.

Example 3: Calculate the volume of 0. 08 M KBr solution containing 1. 6 moles of potassium bromide.

Example 4 A 0. 50 -molar aqueous solution of sulfuric acid flows into a process unit at a rate of 1. 25 m 3/min. The specific gravity of the solution is 1. 03. Calculate (1) the mass concentration of H 2 SO 4 in kg/m 3, (2) the mass flow rate of H 2 SO 4 in kg/s, and (3) the mass fraction of H 2 SO 4. Solution 1. 0. 50 mol H 2 SO 4 98 g L 2. 1. 25 m 3 min 1 kg 103 L mol 103 g 49 kg H 2 SO 4 m 3 1 min 60 s 1 m 3 = 49 kg H 2 SO 4/m 3 = 1. 0 kg H 2 SO 4/s 3. 1. 25 m 3 solution min 1030 kg m 3 solution 1 min 60 s = 21. 46 kg/s




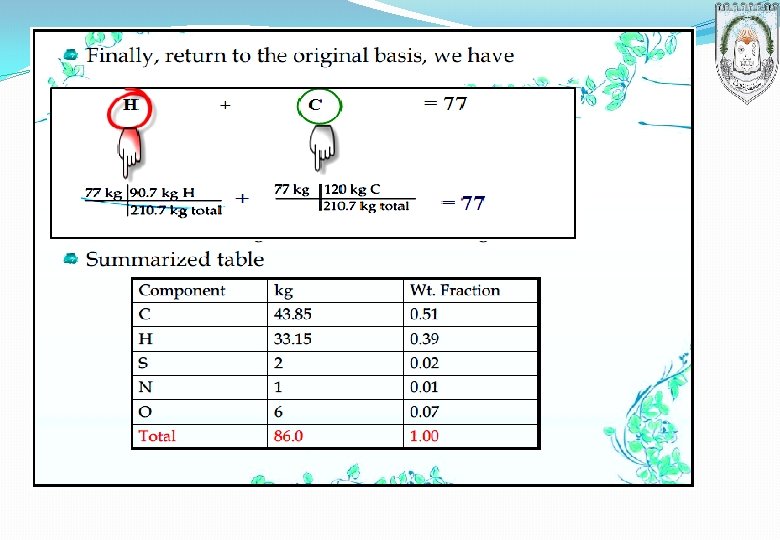




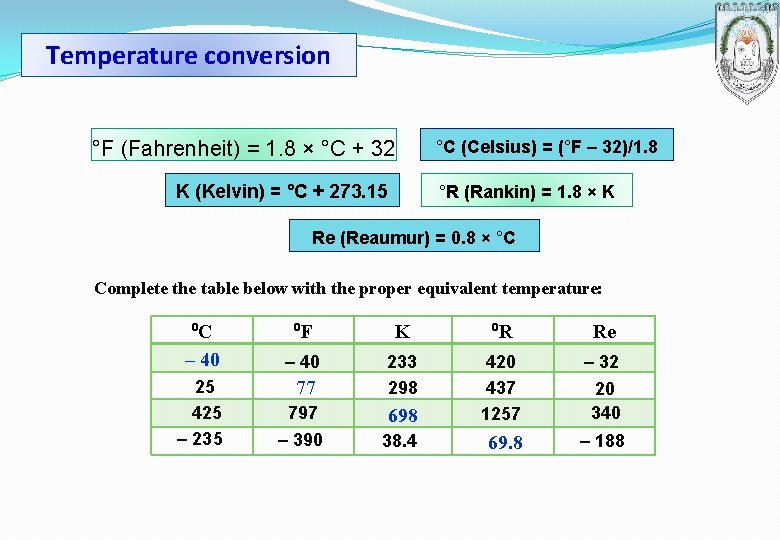
Temperature conversion °F (Fahrenheit) = 1. 8 × °C + 32 °C (Celsius) = (°F – 32)/1. 8 K (Kelvin) = °C + 273. 15 °R (Rankin) = 1. 8 × K Re (Reaumur) = 0. 8 × °C Complete the table below with the proper equivalent temperature: ⁰C – 40 25 425 – 235 ⁰F K ⁰R Re – 40 797 698 420 437 1257 – 32 77 233 298 – 390 38. 4 69. 8 – 188 20 340
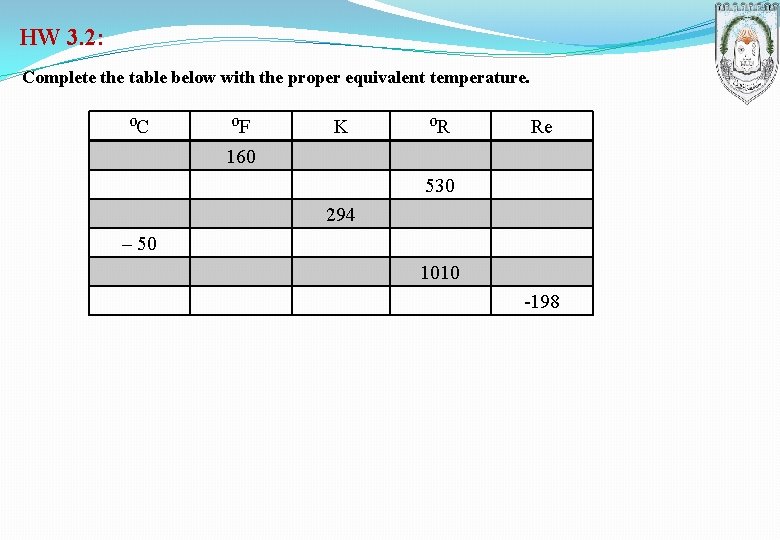
HW 3. 2: Complete the table below with the proper equivalent temperature. ⁰C ⁰F 160 K ⁰R Re 530 294 – 50 1010 -198
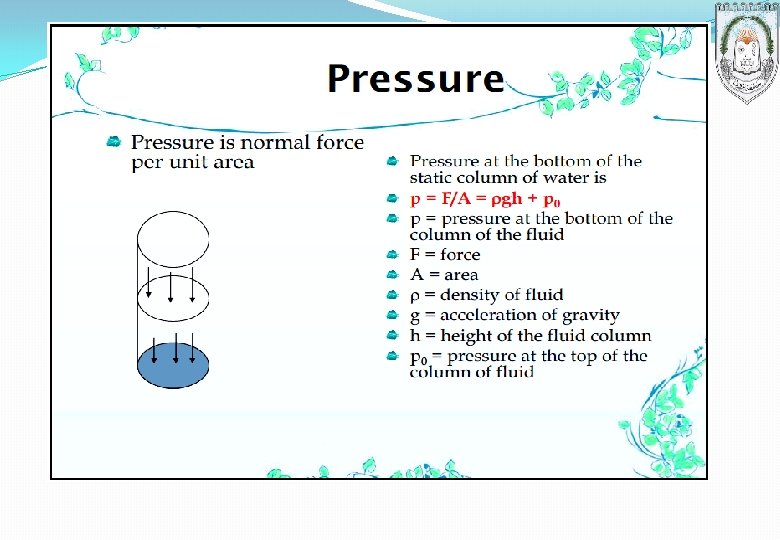










Example 2 What is the pressure 30. 0 m below the surface of a lake? Atmospheric pressure (the pressure at the surface) is 10. 4 m H 2 O, and the density of water is 1000. 0 kg/m 3. Assume that g is 9. 807 m/s 2. Solution 10. 4 m H 2 O 1. 013 105 N/m 2 10. 33 m H 2 O + 1000. 0 kg/m 3 9. 807 m s 2 = 3. 96 105 N/m 2 (Pa) = 396 k. Pa CLASS WORK Verify that the two calculated pressures are equivalent. 30. 0 m 1 N 1 kg. m/s 2
- Slides: 62