Process Skills in Science Science A body of

Process Skills in Science

Science- A body of knowledge and a process by which we learn the knowledge (scientific method). Scientific Method: Problem: question you are trying to answer Background Information: Information required to help answer the question Hypothesis: Materials: An educated guess (If, then and because statement) stuff we use Procedure: explains steps in detail (must be simplified enough for anyone to follow but precise enough to ensure that it can be replicated) Observation Analysis: and Results: what did you see/observe relating the results to the problem with background information Conclusion: hypothesis Answering the problem and supporting/disproving your

Variables: things that are changed in an experiment Manipulated: independent Responding: Dependent Controlled Manipulated: changed by the experimenter Responding: changes because of the manipulated variable Responds to your changes Controlled: Kept constant The results depend on what you’ve done here We use it to compare our results Control of an Experiment: A control group is used as a comparison to the experimental group.

Controlled Experiment: A controlled experiment is one where A) all of the variables are kept the same except for one B) There is only one manipulated variable and one responding variable Graphing Techniques: Don’t forget a title for the graph Don’t forget to label the axis X-axis: manipulated variable Y-axis: responding variable Lines of best fit used to describe a relationship that we see in a graph Straight Curved line: ONLY if you see a straight line Line: ONLY if you see a curved line

Cell Energy Why do we care about this? We need energy to keep cells alive Activities require energy Energy Requirements: Energy is released from foods The digestion process breaks down food into molecules Molecules are absorbed by cells

Two 1) uses of food by cells: Anabolism: synthesis of larger molecules required by the cell Example: Enzymes 2) Catabolism: broken down into smaller molecules to release energy for cellular activities Example: Metabolic contraction of muscle cells Activities: Sum of the chemical reactions that occur in cells Two type of chemical reactions: Exergonic Endrogenic

Exergonic Reactions: Example: Reactants Cellular Respiration Products + Energy Endrogenic Requires Reactions: energy Example: Reactants Photosynthesis + Energy Products

Energy Conversions All cells must have a constant supply of energy Energy is trapped and released by two processes: Photosynthesis Cellular The Respiration Basics: Photosynthesis: Light energy is changed into chemical energy and stored in glucose Respiration: to the cell Glucose is broken down to release energy

Photosynthesis Happens only in green plants in the presence of chlorophyll which is found in chloroplasts Chemical Energy Equation: stored in the form of glucose

Cellular Respiration Happens in ALL cells whereby energy is released to perform life processes Chemical Involves Energy Equation: burning of organic fuels by oxygen is released

Photosynthesis and Cellular Respiration Biology 20 - Unit 3

Let’s Review Plants and animals are made up of eukaryotic cells (have a nucleus) In spite of varied size, shape and appearance, cells have several things in common. All cells digest nutrients, excrete wastes, synthesize needed chemicals and reproduce. 12

Cellular Anatomy All cells are made up of parts known as organelles Although both plant & animal cells share many common types of organelles there also organelles which are unique to each type See pgs 154 - 155 13

The Cell Membrane Separates the internal environment of the cell from its external environment Made of a double layer of phospholipid molecules

Membranes also have other proteins and molecules that are embedded within them. Create passageways through the membrane

Membranes Two are semi-permeable ways molecules and ion move through the membrane are diffusion and osmosis (passive transport)

Diffusion Natural movement of molecules or ions from an area where they are more concentrated to an area of less concentration (moving down its concentration gradient) Does not require energy

Osmosis Diffusion of water across a membrane Movement of water depends of different things If water concentration of either side is equal than equal amounts of water move in and out (isotonic) If the water concentration is more outside the cell than in the water will move into the cell (hypotonic) If water concentration inside the cell is greater than outside water moves out (hypertonic). -Does not require energy


Facilitated Diffusion Substances that need help to move in and out of the cell (example: glucose) Particular transport proteins will recognize and help move a specific type of dissolved molecule or ion (carrier protein) Channel proteins also carry molecules across but they must be small enough to fit through the tunnel.

Active Transport Needs ATP- energy to move things in and out of the cell adenosine triphosphate

Endocytosis and Exocytosis Used for substances too large to move across a membrane Example: Can Cholesterol use endocytosis- folds in on itself to create a membrane enclosed sac (vesicle) to “eat” the substance

Types of Endocytosis 3 Type of Endocytosis: 1. Pinocytosis: intake of small amounts of liquids or small particles 2. Phagocytosis: intake of large amounts of liquids or larger particles 3. Receptor- assisted endocytosis: intake of specific molecules that attach to special proteins in the membrane

Exocytosis Removing Vesicles substances from the cell from inside the cell moves to the membrane and “bursts” releasing its contents

Cell Wall-Plant Cells A surrounding layer outside the cell membrane Composed of small fibres (microfibrils) of cellulose 25

Animal cells

Animal Cell Parts Mitochondria: provide the energy a cell needs to move, divide, reproduce Power Centre of the cell Cristae are folded to increase surface area

Parts of an Animal cell Cytoplasm: fluid that fills the cell Distributes materials such as oxygen and food to different parts of the cell Also helps support all the other parts of the cell

Parts of an Animal Cell Nucleus: large dark nucleus is often the most easily seen structure in the cell Controls the cell activities Contains the chromosomes Enclosed in a nuclear membrane which controls what enters and leaves the cell

Parts of an Animal Cell Nucleolus: Produces prominent structure in the nucleus ribosomes

Parts of an Animal Cell Vacuoles: Storage places for surplus food, wastes and other substances that the cell cannot use right away.

Parts of an Animal Cell Lysosomes: digestion

Parts of An Animal Cell Golgi Apparatus: important in packaging proteins for transport elsewhere in the cell
Parts of An Animal Cell Rough Endoplasmic Reticulum: Appears pebbled due to the presence of ribosomes. Synthesizes proteins

Parts of An Animal Cell Smooth Endoplasmic Reticulum: Appears smooth Functions: lipid and steroid synthesizes

Parts of an Animal Cell Ribosome: Site of protein synthesis

Animal cells

What You Need to have in an Animal Cell Nucleus Cell Membrane Cytoplasm Vacuoles Ribosomes Golgi Apparatus Rough Endoplasmic Reticulum Smooth Endoplasmic Reticulum Lysosomes Mitochondria Nucleolus

Plant cells

Parts of a Plant Cell Lucky for you plants have many of the same structures 1)Nucleus 2) Nucleolus 3) Cytoplasm 4) Golgi Apparatus 5) Lysosome 6) Mitochondria 7) Vacuole 8) Cell Wall 9) Smooth ER 10) Rough ER 11) Chloroplast

Parts of a Plant Cell Wall: are much thicker and more rigid than cell membranes and are made mostly of a tough material called cellulose. Provide support for the cell

Parts of a Plant Cell Chloroplasts: takes place Structures where photosynthesis

Photosynthesis & Cellular Respiration

ATP and Cellular Activity How does ATP supply energy for cellular activity?

ATP Supplies the energy for cellular activities Used rapidly so cells must be constantly creating Used for: it Active transport Movement of chromosomes Movement of muscles Cilia or flagella etc.

Photosynthesis Needed in order for life to survive on Earth Photosynthesizing organisms contain chloroplasts that trap the Sun’s energy Converted into chemical energy and stored as sugars and carbohydrates Other products produced by the Sun’s energy are oxygen. ATP and heat

Cellular Respiration Used by plants, animals and other multicellular organisms The break down of energy-rich compounds to release stored energy Broken This down inside the mitochondria makes ATP

Photosynthesis – Chemical reaction Energy from the sun is captured by green plants by the process called photosynthesis (P/S) 6 CO 2 + 6 H 2 O + E (light) C 6 H 12 O 6 + 6 O 2 48

Photosynthesis - Pigments For light energy to be used by living systems it must first be absorbed. A pigment is any substance that absorbs light. (Some pigments absorb all light and thus appear black. Others absorb light in the violet-blue and the orange-red spectrum and reflect green light. ) 49

Photosynthesis - Pigments Various pigments absorb energy of different wavelengths. (absorption spectrum) 50

Photosynthesis - Pigments All photosynthetic organisms contain chlorophyll Different types of plants use various pigments in P/S. 51

Photosynthesis - Pigments Chlorophyll a (blue-green) and chlorophyll b (yellowgreen) are the most common pigments but most plants also contain a pigment group called carotenes (Example: They beta-carotene) absorb photons with energies in the blue-violet and regions and reflect everything else 52

Chlorophyll a and b Chlorophyll a is the only pigment that can transfer the energy from sunlight to photosynthesis Chlorophyll b acts as an accessory pigment “helper” to catch the photons a misses and transfer the energy absorbed to a There are other compound, carotenoids , are also “helper” pigments.

Photosynthesis - Pigments In green leaves carotenes are masked by chlorophyll, thus when chlorophyll production slows in the fall the leaves change color to show the carotenes. 54

Photosynthesis - Chloroplast Chlorophyll pigments reflect green and absorb blue and red wavelengths. Carotenoids absorb violet and blue wavelengths reflecting yellow. Pigments absorb light of the correct wavelength to excite electrons to a higher energy level 55

Photosynthesis - Plastids are structures that contain pigment and give plants their colour. The most common plastid is the chloroplast in which the chemical reactions of P/S occur. 56

Cellular Respiration Produces ATP energy by the combustion reaction of glucose called cellular respiration. C 6 H 12 O 6 + 6 O 2 + 6 H 2 O 6 CO 2 + 12 H 2 O + E (ATP) 57
Mitochondrial structure Respiration occurs at the mitochondrian Mitochondrian is composed of 4 regions: 1. outer membrane - smooth and freely permeable; contains enzymes to catabolize fats 58

Mitochondrial structure 2. inner membrane – folded membrane in the mitochondria (cristae); made mostly of protein including the enzyme that makes ATP; impermeable to most small molecules and ions 59

Mitochondrial structure 3. Intermembrane space - contains enzymes which use ATP 60

Mitochondrial structure 4. matrix - control region; mixture of proteins including enzymes which oxidize major compounds 61

Metabolic Pathways In a metabolic pathway the product of one reaction is the starting reactant for another

The Role of Enzymes Metabolism refers to all the chemical reactions that occur within a cell to support and sustain life functions Can be broken into two distinct types of reactions: 1. Anabolic reactions & pathways create larger molecules from small subunits and require energy 2. Catabolic reactions & pathways break down large molecules into smaller pieces and release energy

Energy required to start a reaction is known as activation energy Catalysts Allows and enzymes reduce the activation energy the reactions to proceed more rapidly Enzymes are specialized proteins that lower the energy needed to activate biological reactions

Activation Energy Catalyzed vs. Uncatalyzed Reactions

Oxidation & Reduction Oxidation is a reaction where an atom or molecule loses electrons LEO – Loses Electrons = Oxidation Reduction is a reaction where an atom or molecule gains electrons GER – Gains Electrons = Reduction Free electrons from oxidation cannot exist on their own Electrons that are lost through oxidation of one substance cause the reduction of another compound Molecules energy in their reduced form contain large amounts of

Example: X + Y --> X+ + Yreducing oxidized reduced agent

Adenosine Triphosphate The cell obtains its energy requirements through cellular respiration which is an exothermic reaction manufacturing ATP 68

ATP produces energy by breaking a bond to a phosphate group This produces ADP (adenosine diphosphate) and a free phosphate group ATP This ADP + Pi process works in reverse to create more ATP

Adenosine Triphosphate Adenosine triphosphate (ATP) consists of: nitrogenous base adenine (1/5 types of nitrogenous bases) an attached ribose sugar. attached to the sugar are 3 phosphate groups. 70

Adenosine Triphosphate The terminal phosphate is bonded by a covalent bond of unusually high energy. 71

Adenosine Triphosphate During cellular respiration a free phosphate group is attached to a molecule of ADP to make ATP in a process called phosphorylation 72

Adenosine Triphosphate S Y A W L A t s u M u o F Y L E n S O R P U w T o O A Y N E K m H S o T A r S F I E R E H W ATP is used to provide the activation energy needed to power cell reactions (Energy is liberated by detaching the terminal phosphate group) 73

Review Cellular respiration is the process by which cells break down high-energy compounds and generate ATP.

Review Adenosine triphosphate, or ATP, is the direct source of energy for nearly all types of energy-requiring activities of living organisms.

Mitochondria Review have outer and inner membranes that surround a fluid-filled region called the matrix. The inner membrane has many deep infoldings called cristae.

Review The chemical reactions of photosynthesis and cellular respiration take place in a series of step-by-step reactions called metabolic pathways. Enzymes are biological catalysts that reduce the amount of startup energy needed for the reactions in the metabolic pathways. In the absence of enzymes, the reactions could not occur at temperatures at which living organisms thrive.

Review When a compound is oxidized in a chemical reaction, it loses electrons. When a compound is reduced in a chemical reaction, it gains electrons. Compounds contain more chemical energy in their reduced form than they do in their oxidized form.

Ph n y s o ot s i s e th

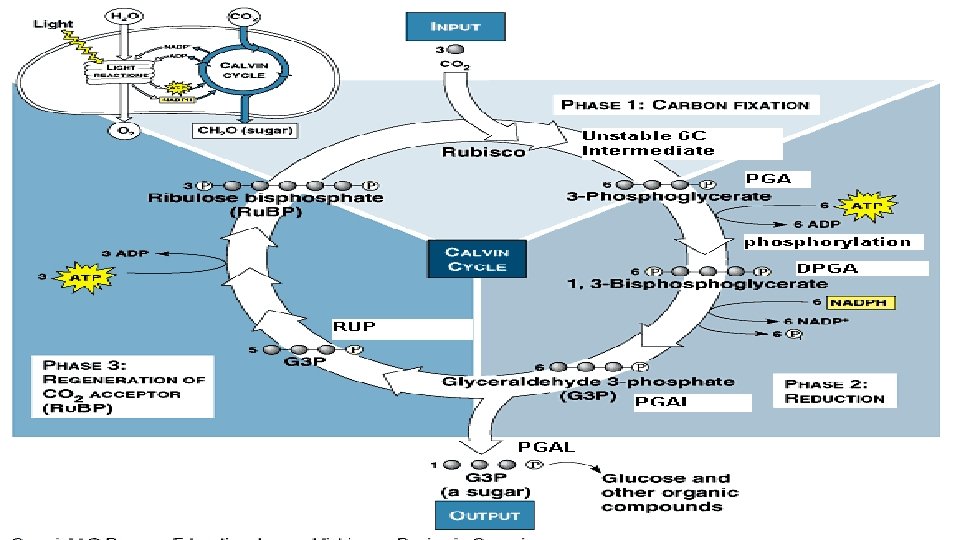
Photosynthesis Transforms the energy of the sun into chemical energy in glucose, ATP and NADPH Involves over 100 individual chemical reactions that work together These reactions can be summarized in two groups: 1. 2. Light-Dependent Reactions – generates high energy compounds ATP and NADPH Light-Independent Reactions – energy of ATP and reducing power NADPH are used to reduce carbon dioxide to make

Light Dependent Light Independent 81

Light-Dependent Reactions Requires sunlight in order to work During these reactions, the pigments contained inside thylakoid absorb light energy Although plants have a number of pigments, the most important for photosynthesis is chlorophyll

Photosystems Within the thylakoid membrane, chlorophyll and other pigments are organized into photosystems. Chloroplasts of plants have two photosystems: Photosystem I (PSI) Photosystem II (PSII) Each system is made of pigment molecules that include chlorophyll and carotenoid molecules All the pigment molecules in each photosystem can absorb various wavelengths of light energy

The various pigment molecules produce free electrons when light hits them These free electrons are passed along to the reaction center, a specialized chlorophyll a molecule When the electron in the reaction center is “excited” by the addition of energy, it passes to the electronacceptor molecule This reduces the electron acceptor and puts it at a high energy level

A summary of the Steps: The light reactions use the solar power of photons absorbed by both photosystem I and photosystem II to provide chemical energy in the form of ATP and reducing power in the form of the electrons carried by NADPH. Takes place in the thylakoid membranes of the chloroplast

86

Light Dependent Reaction – The Details: Photosystem II (PSII) Light enters PSII and is trapped by Pigment-680 (P 680) An electron from P 680 is boosted to a higher energy level where it is passed to an electron acceptor molecule This electron passes down an electron transport chain (cytochromes) to PSI forming ATP from ADP in a process called photophosphorylation 87

Light Dependent Reaction – The Details: Photosystem II (PSII) The lost electrons from P 680 are replaced by electrons produced by the lysis of water photolysis, which liberates O 2 as a waste product 88

Light Dependent Reaction – The Details: Photosystem I (PSI) The electron arriving from PSII is boosted to another electron acceptor molecule As it is passed along it releases energy This energy pulls hydrogen ions from the stroma into the thylakoid lumen 89

Light hits photosystem 1 An electron in this photosystem is “excited” and passed onto the smaller electron transport chain The excited electron from photosystem 1 passes down a chain of coenzymes (cytochromes) to make NADPH molecules from NADP

Light Dependent Reaction 91 http: //www. youtube. com/watch? v=BK_cjd 6 Evcw

ATP Production - Chemiosmosis The energy from the electrons in photosystem II is used to produce ATP indirectly The H+ ions in the thylakoid lumen are unable to escape except through special proteins called ATP synthase complexes As the H+ ion moves though this complex they release energy The complex uses some of this energy to combine ADP and Pi making ATP This ATP then moves onto the light-independent reaction to make glucose

Chemiosmosis Linking the movement of hydrogen ions to the production of ATP Occurs in a series of steps: 1. To return to the stroma, the H+ ions must move through a structure known as ATP synthase which provides the only pathway for H+ ions to move down their concentration gradient 2. ATP synthase uses the movement of the H+ ions to run a mechanism that bonds together ADP and free phosphates to form ATP

Your Task Case Study page 184 questions 1 and 2 optional Section Questions page 185 questions 1 -4 somewhat optional Practice Questions page 187 #1 -3 getting less optional Practice Questions page 188 #4 -6 not really a choice Practice Questions page 190 # 7 -10 I wouldn’t ignore these Practice Question page 191 # 11 -14 Do or suffer the consequences Expect a Quiz next class 94

Light Independent Reactions Does Also not require light known as the Calvin-Benson Cycle Occur in the stroma of the chloroplast Glucose is synthesized which requires: a. energy in the form of ATP and NADPH (there has to be enough) b. H since each glucose molecule has 12 H atoms

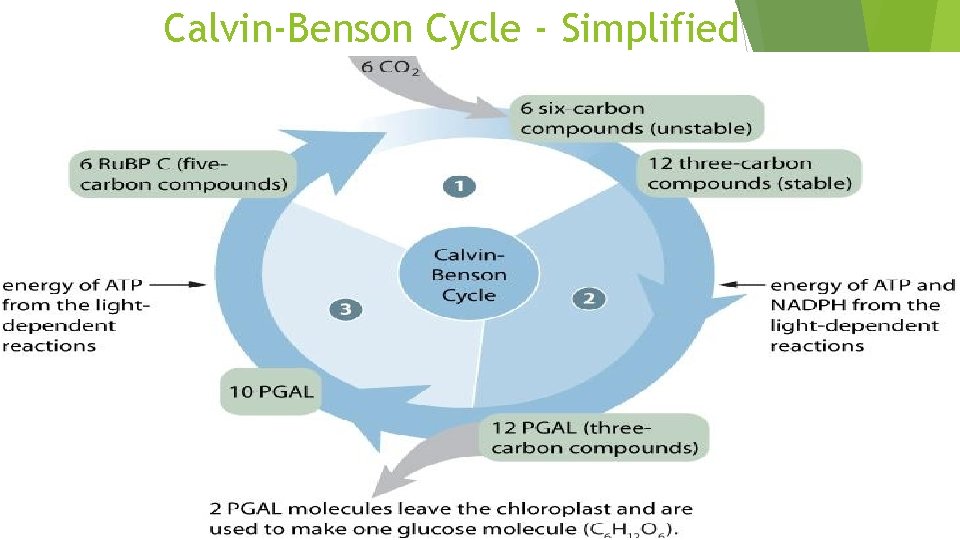
The Calvin-Benson Cycle The Calvin cycle regenerates its starting material after molecules enter and leave the cycle CO 2 enters the cycle and leaves as sugar The cycle spends the energy of ATP and the reducing power of electrons carried by NADPH to make the sugar The actual sugar product of the Calvin cycle is not glucose, but a three-carbon sugar, glyceraldehyde-3 -phosphate (PGAL)

Each turn of the Calvin cycle fixes one carbon. For the net synthesis of one PGAL molecule, the cycle must take place three times, fixing three molecules of CO 2. To make one glucose molecules would require six cycles and the fixation of six CO 2 molecules. The Calvin cycle has three phases. Calvin - Benson Cycle http: //www. youtube. com/watch? v=ixp. Nw 6 mx 3 lk
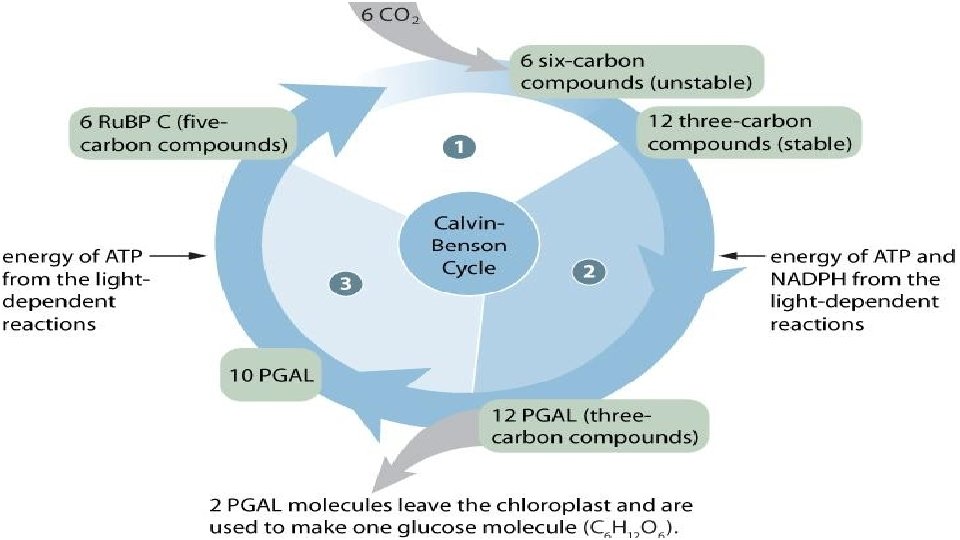
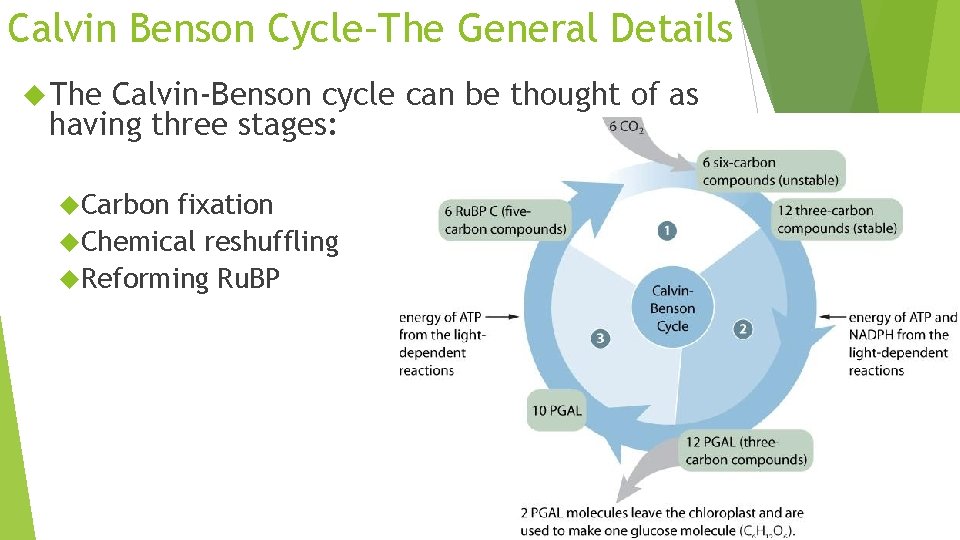
Calvin Benson Cycle–The General Details The Calvin-Benson cycle can be thought of as having three stages: Carbon fixation Chemical reshuffling Reforming Ru. BP 99

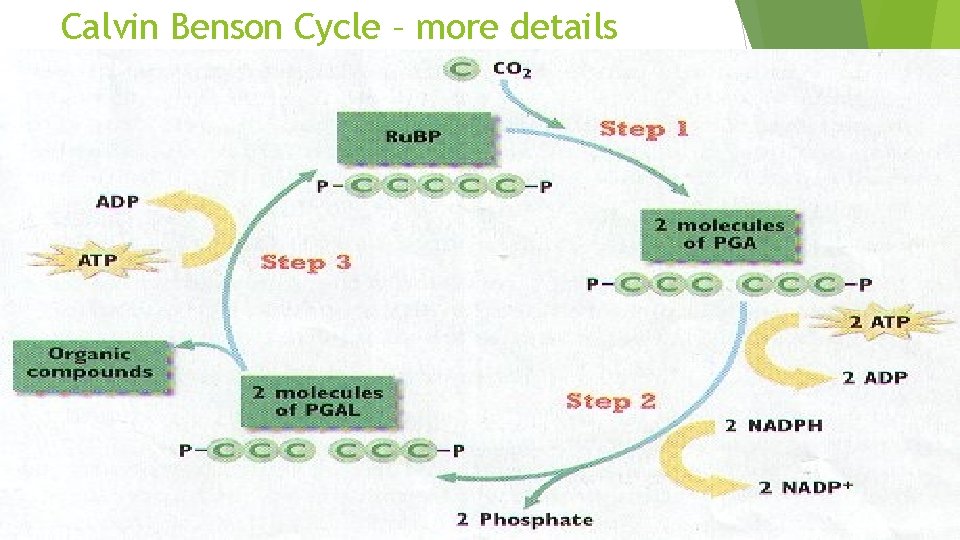
Light Independent Occurs Step in 3 stages: 1: Carbon Fixation Ru. BP (ribulose biphosphate) joins with CO 2 (catalyzed by Rubisco) to form an unstable 6 carbon molecule which splits to become PGA (phosphoglyceric acid)

Light Independent Step 2: Reduction The 3 carbon compounds are activated by ATP (given energy) and then reduced by NAPDH (given more energy) The molecule now become 12 molecules known as PGAL 2 PGAL molecules move on to make glucose, 10 go to step 3

Light Independent Step 3: Replacing Ru. BP Remaining ATP PGAL will be used to make more Ru. BP will help break and reform the chemical bonds to make the 5 -carbon Ru. BP
Calvin-Benson Cycle - Simplified 103
Calvin Benson Cycle – more details 104
105
Let’s Put It All Together… http: //www. youtube. com/watch? v=Fo. CS 3 Ep. Ud. V 4
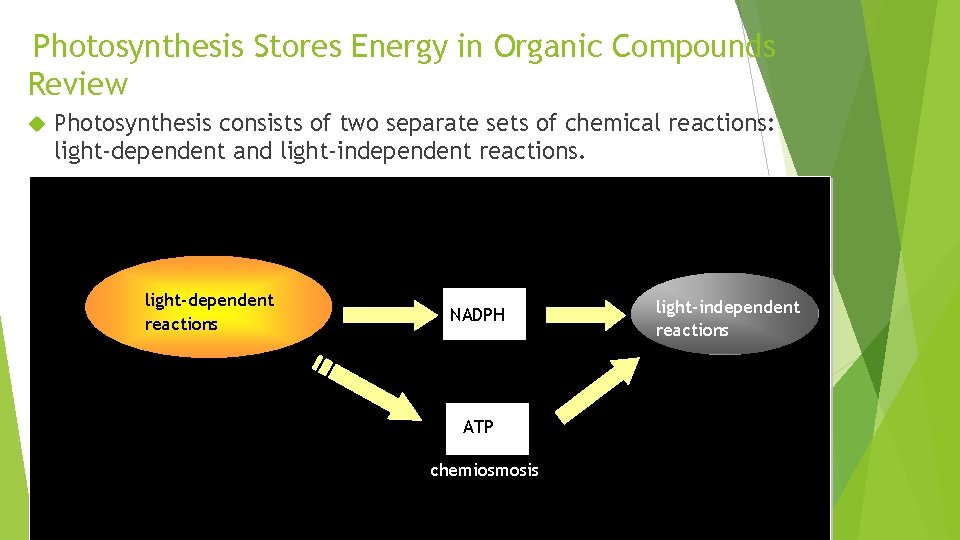
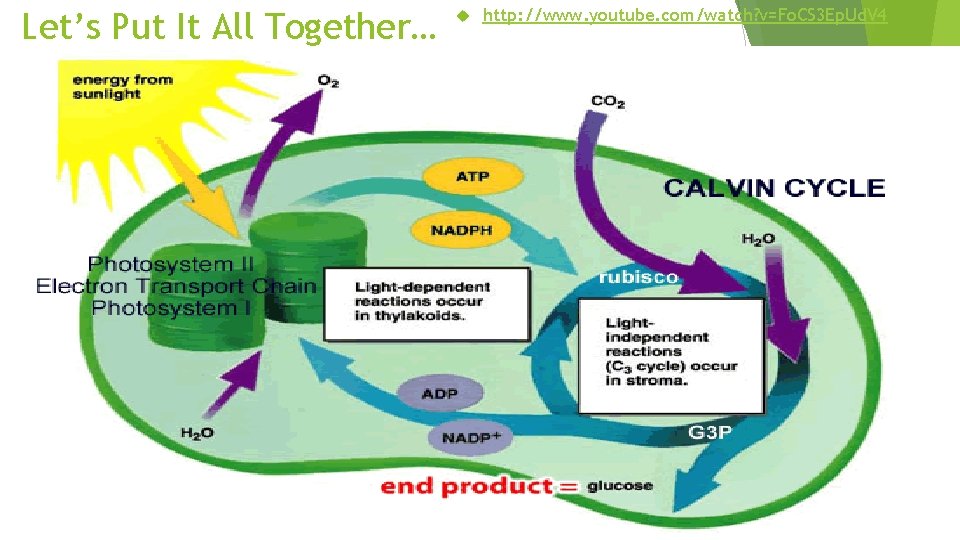
Photosynthesis Stores Energy in Organic Compounds Review Photosynthesis consists of two separate sets of chemical reactions: light-dependent and light-independent reactions. light-dependent reactions NADPH ATP chemiosmosis light-independent reactions
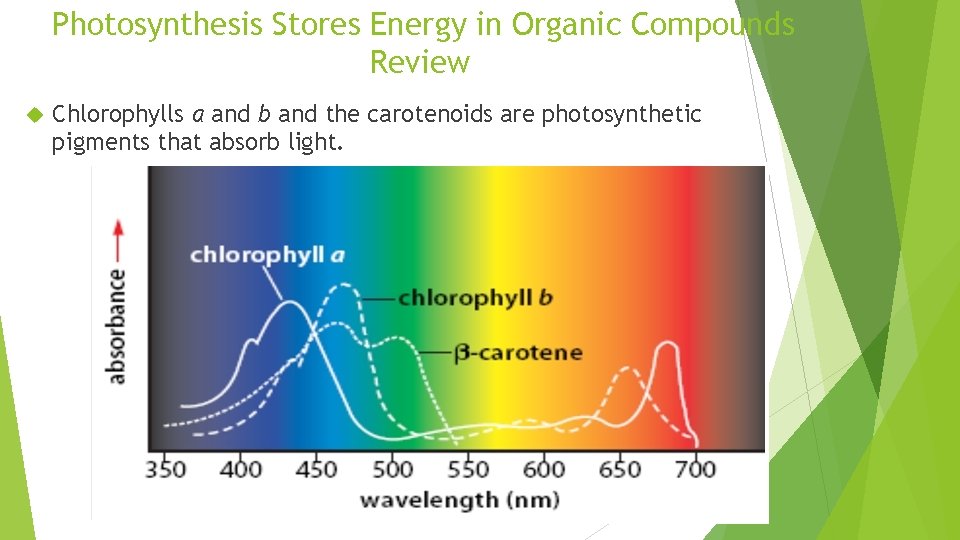
Photosynthesis Stores Energy in Organic Compounds Review Chlorophylls a and b and the carotenoids are photosynthetic pigments that absorb light.
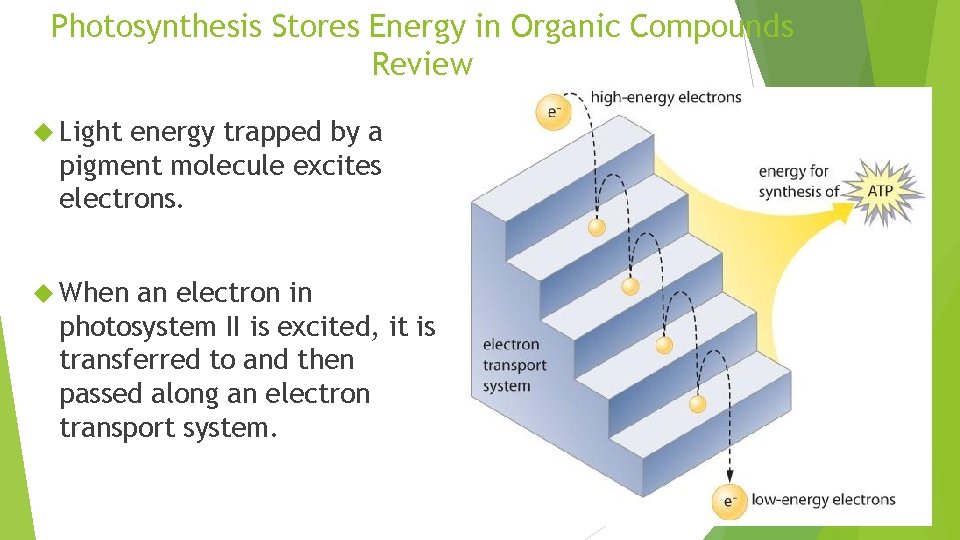
Photosynthesis Stores Energy in Organic Compounds Review Light energy trapped by a pigment molecule excites electrons. When an electron in photosystem II is excited, it is transferred to and then passed along an electron transport system.
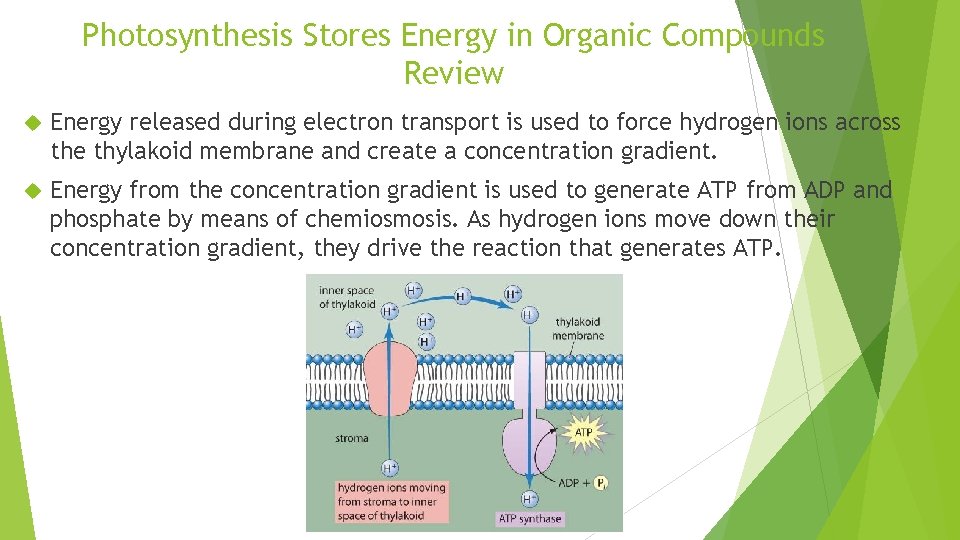
Photosynthesis Stores Energy in Organic Compounds Review Energy released during electron transport is used to force hydrogen ions across the thylakoid membrane and create a concentration gradient. Energy from the concentration gradient is used to generate ATP from ADP and phosphate by means of chemiosmosis. As hydrogen ions move down their concentration gradient, they drive the reaction that generates ATP.
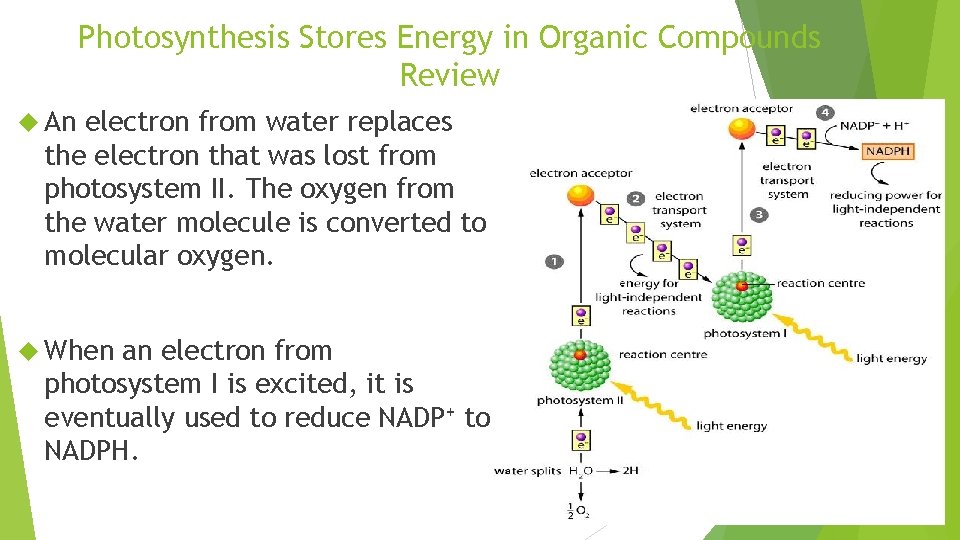
Photosynthesis Stores Energy in Organic Compounds Review An electron from water replaces the electron that was lost from photosystem II. The oxygen from the water molecule is converted to molecular oxygen. When an electron from photosystem I is excited, it is eventually used to reduce NADP+ to NADPH.

Photosynthesis Stores Energy in Organic Compounds Review • The series of reactions that synthesize carbohydrates is the Calvin-Benson cycle, which occurs in the stroma. • In this cycle, carbon dioxide combines with Ru. BP to form a six-carbon compound that immediately splits into two three-carbon compounds.

Photosynthesis Stores Energy in Organic Compounds Review ATP and NADPH from the light-dependent reactions provide energy and reducing power to form PGAL from the newly formed three-carbon compounds. Six cycles produce 12 PGAL molecules, 10 of which regenerate Ru. BP and 2 of which are used to make glucose.
- Slides: 113