Process and Reaction Engineering PRE Maria K Burka

Process and Reaction Engineering (PRE) Maria K. Burka --- Program Director 703 -292 -7030 mburka@nsf. gov National Science Foundation

Program Overview Process and Reaction Engineering Program supports research and educational projects related to: 1. Interactions between chemical reactions and transport phenomena in reactive systems, and the use of this information in the design of complex chemical and biochemical reactors (Reaction Engineering) a. Reactive processing of polymers , ceramics, and thin films b. Electrochemical and photochemical processes of engineering significance or with commercial potential 2. Design and optimization of complex chemical processes (Design) 3. Dynamic modeling and control of process systems and individual process units (Control) National Science Foundation

Reaction Engineering • Environmental issues – green chemistry – Developing a catalytic reactor to remove toxic components of landfill gas (LFG) so that LFG can be used as an alternate source of energy – Developing a fundamental, molecular level understanding of the hydrolysis reaction • Application of new CI paradigms – Dynamic complexity in reacting systems • Microreactors – Engineer active surfaces for the detection of nitroaromatic, nitramine, and nitrate ester compounds, which are the primary constituents of explosive devices • • Electro- and photo-chemical systems Reactors used in microelectronics manufacturing: CVD, plasma reactors Bioreactors – fermentation, biofuels, etc. Nano – Develop Cu/CNT nanowires as potential future interconnects to overcome current technology barriers including electromigration and increased electrical resistivity National Science Foundation
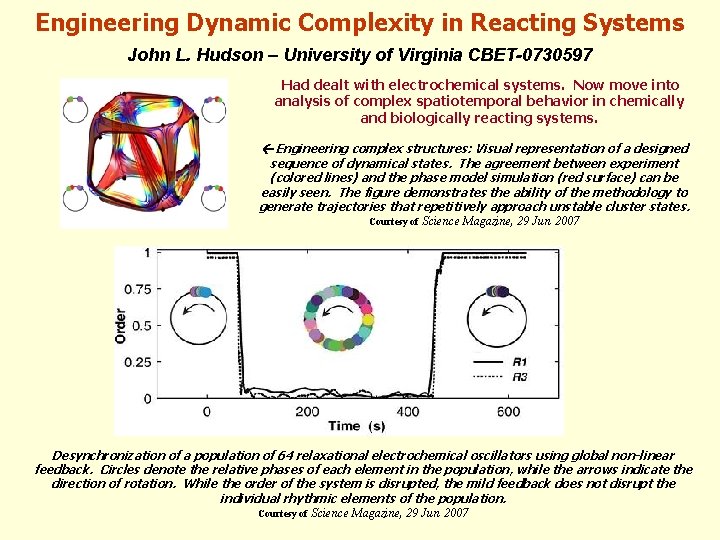
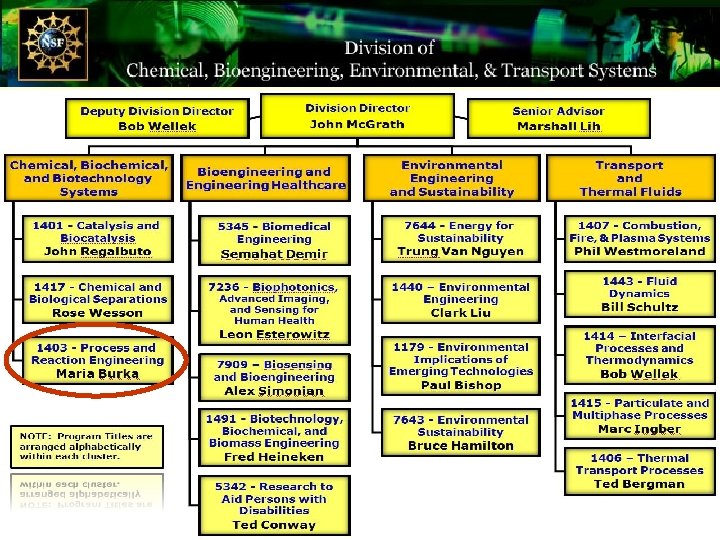
Engineering Dynamic Complexity in Reacting Systems John L. Hudson – University of Virginia CBET-0730597 Had dealt with electrochemical systems. Now move into analysis of complex spatiotemporal behavior in chemically and biologically reacting systems. Engineering complex structures: Visual representation of a designed sequence of dynamical states. The agreement between experiment (colored lines) and the phase model simulation (red surface) can be easily seen. The figure demonstrates the ability of the methodology to generate trajectories that repetitively approach unstable cluster states. Courtesy of Science Magazine, 29 Jun 2007 Desynchronization of a population of 64 relaxational electrochemical oscillators using global non-linear feedback. Circles denote the relative phases of each element in the population, while the arrows indicate the direction of rotation. While the order of the system is disrupted, the mild feedback does not disrupt the individual rhythmic elements of the population. Courtesy of Science Magazine, 29 Jun 2007

Chemical Process Control • Development of Fundamental Control Algorithms – Model predictive control – Robust, adaptive, etc. • Application areas – Using wireless sensors and actuators to control industrial chemical processes – Economic optimization of chemical processes using feedback control – Predict electrowetting dynamics first in two, then in three, spatial dimensions, to enable next-generation system analysis, design, and control – Dynamics and control of process networks with energy integration National Science Foundation

Chemical Process Design • Development of Fundamental Design Methodology – Developing global optimization methodologies – Hierarchical design methods for rapidly estimating economically optimal operating policies for plants with recycle – Using concept of shortest separation lines for design of energy efficient multiunit processes – Open cyberinfrastructure for mixed-integer nonlinear programming: collaboration and deployment via virtual environments • Application Areas – Modeling, parameter estimation and dynamic optimization of Lithium-ion batteries • Design of Tailor-made Molecules –> to Enterprises: – From nanoscale simulation to process engineering: building a network for understanding polymer dynamics National Science Foundation
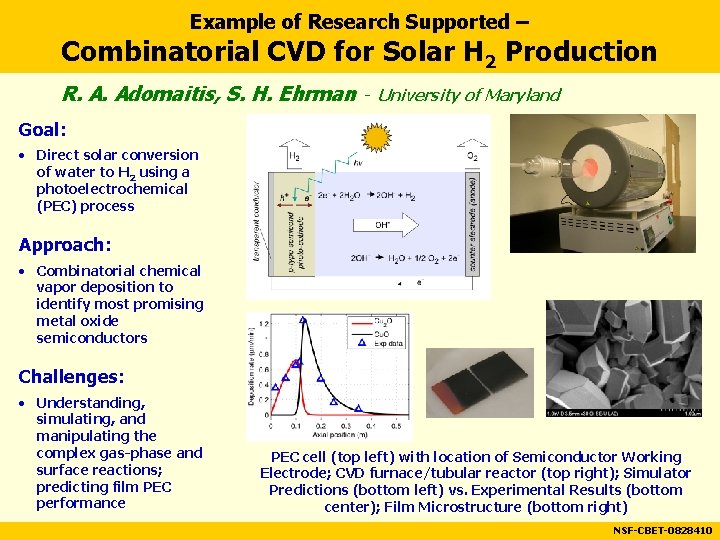
Example of Research Supported – Combinatorial CVD for Solar H 2 Production R. A. Adomaitis, S. H. Ehrman - University of Maryland Goal: • Direct solar conversion of water to H 2 using a photoelectrochemical (PEC) process Approach: • Combinatorial chemical vapor deposition to identify most promising metal oxide semiconductors Challenges: • Understanding, simulating, and manipulating the complex gas-phase and surface reactions; predicting film PEC performance PEC cell (top left) with location of Semiconductor Working Electrode; CVD furnace/tubular reactor (top right); Simulator Predictions (bottom left) vs. Experimental Results (bottom center); Film Microstructure (bottom right) NSF-CBET-0828410

Reactive Polymer Processing • Paints, coatings, thin films, etc. – Emulsion and miniemulsion polymerization • Microelectronics, environment, etc. : – Initiated chemical vapor deposition (i. CVD) to make semiconducting, conjugated polymers for use in solar cells with enhanced efficiency and performance – Depolymerization of hemicellulose for usability as biofuel feedstock – Vapor deposition polymerization of porous polymers – Design, synthesize and characterize molecule-based magnetic polymers with high Curie temperature National Science Foundation
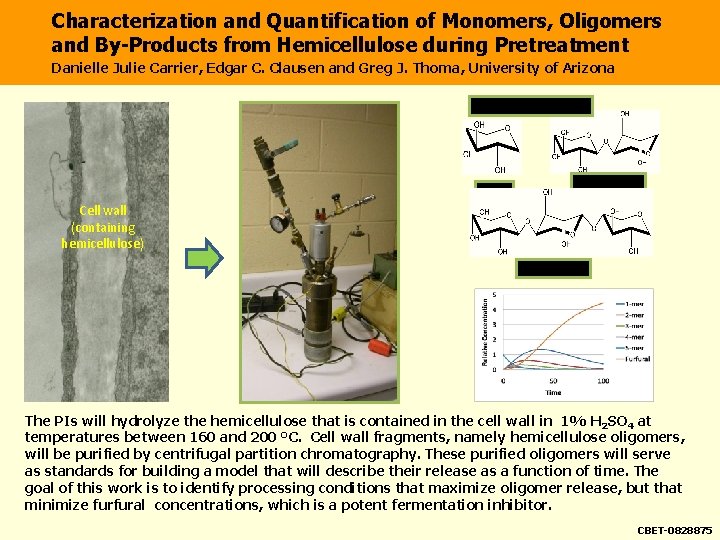
Characterization and Quantification of Monomers, Oligomers and By-Products from Hemicellulose during Pretreatment Danielle Julie Carrier, Edgar C. Clausen and Greg J. Thoma, University of Arizona Oligimers of Hemicellulose Xylose 2 Xylose, b 1 4 Cell wall (containing hemicellulose) 3 Xylose, b 1 4 The PIs will hydrolyze the hemicellulose that is contained in the cell wall in 1% H 2 SO 4 at temperatures between 160 and 200 °C. Cell wall fragments, namely hemicellulose oligomers, will be purified by centrifugal partition chromatography. These purified oligomers will serve as standards for building a model that will describe their release as a function of time. The goal of this work is to identify processing conditions that maximize oligomer release, but that minimize furfural concentrations, which is a potent fermentation inhibitor. CBET-0828875
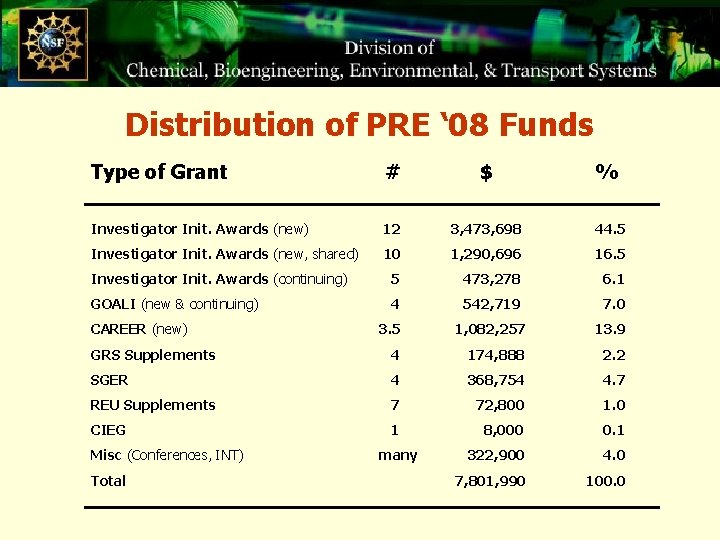
Distribution of PRE ‘ 08 Funds Type of Grant # $ % Investigator Init. Awards (new) 12 3, 473, 698 44. 5 Investigator Init. Awards (new, shared) 10 1, 290, 696 16. 5 Investigator Init. Awards (continuing) 5 473, 278 6. 1 GOALI (new & continuing) 4 542, 719 7. 0 3. 5 1, 082, 257 13. 9 GRS Supplements 4 174, 888 2. 2 SGER 4 368, 754 4. 7 REU Supplements 7 72, 800 1. 0 CIEG 1 8, 000 0. 1 322, 900 4. 0 7, 801, 990 100. 0 CAREER (new) Misc (Conferences, INT) Total many
- Slides: 11