PROCEDURE TO OBTAIN HALAL CERTIFICATION FOR COSMETIC PERSONAL

PROCEDURE TO OBTAIN HALAL CERTIFICATION FOR COSMETIC, PERSONAL CARE SECTOR AND PHARMACEAUTICAL GROUP NAME: CENG GROUP MEMBERS: NADIA AQILAH BINTI KHALID (161141022) FARAH DHIA BINTI ABDUL KARIM (161141007) SHARIFAH NAZURA BINTI SYED SABEER ALI (161141048) NOR AQILAH MUSFIRAH BINTI YUSRI (161141026)

INTRODUCTION HALAL CERTIFICATE OF FREE SALE COSMETIC NOTIFICATION GUIDELINES FOR CONTROL OF COSMETIC PRODUCTS IN MALAYSIA REGULATORY REQUIREMENT FOR COSMETICS POST MARKET SURVEILLIANCE PROGRAMME NOTIFICATION EXEMPTION NOTIFICATION WITHDRAWAL REGULATORY ACTION

n o i t c u d o r t n I q. Halal beauty isn’t something new. Muslim beauty lovers have been developing halal beauty products for decades. q. Recently though, it has surged in popularity, mostly because Muslim beauty-lovers are actively sourcing for halal beauty products, which have become more readily available in the market. q. Halal beauty products refer to those that are ingredient-conscious and cruelty-free.
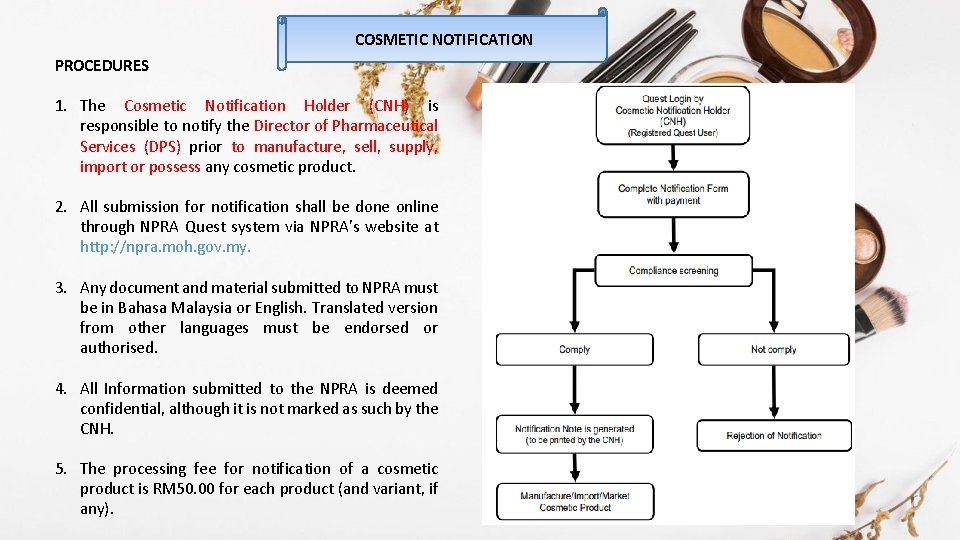
COSMETIC NOTIFICATION PROCEDURES 1. The Cosmetic Notification Holder (CNH) is responsible to notify the Director of Pharmaceutical Services (DPS) prior to manufacture, sell, supply, import or possess any cosmetic product. 2. All submission for notification shall be done online through NPRA Quest system via NPRA’s website at http: //npra. moh. gov. my. 3. Any document and material submitted to NPRA must be in Bahasa Malaysia or English. Translated version from other languages must be endorsed or authorised. 4. All Information submitted to the NPRA is deemed confidential, although it is not marked as such by the CNH. 5. The processing fee for notification of a cosmetic product is RM 50. 00 for each product (and variant, if any).

REGULATORY REQUIREMENTS FOR COSMETICS Products requirement Product name, type Name, address of manufacturer and assembler Name, address, valid contact of CNH Name, address of importer Product ingredients, label of product Safety requirement Must not cause damage CNH must ensure the safety assessment
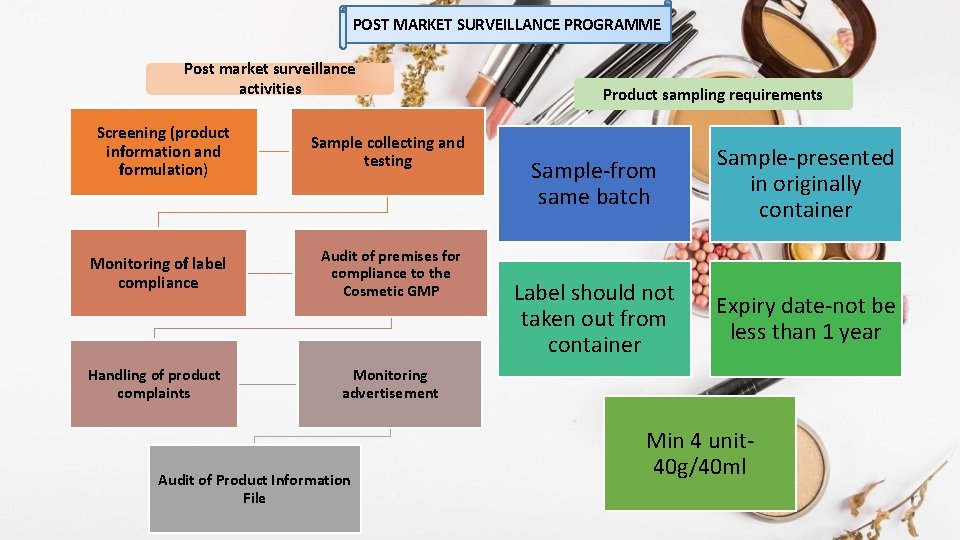
POST MARKET SURVEILLANCE PROGRAMME Post market surveillance activities Screening (product information and formulation) Sample collecting and testing Monitoring of label compliance Audit of premises for compliance to the Cosmetic GMP Handling of product complaints Monitoring advertisement Audit of Product Information File Product sampling requirements Sample-from same batch Sample-presented in originally container Label should not taken out from container Expiry date-not be less than 1 year Min 4 unit 40 g/40 ml
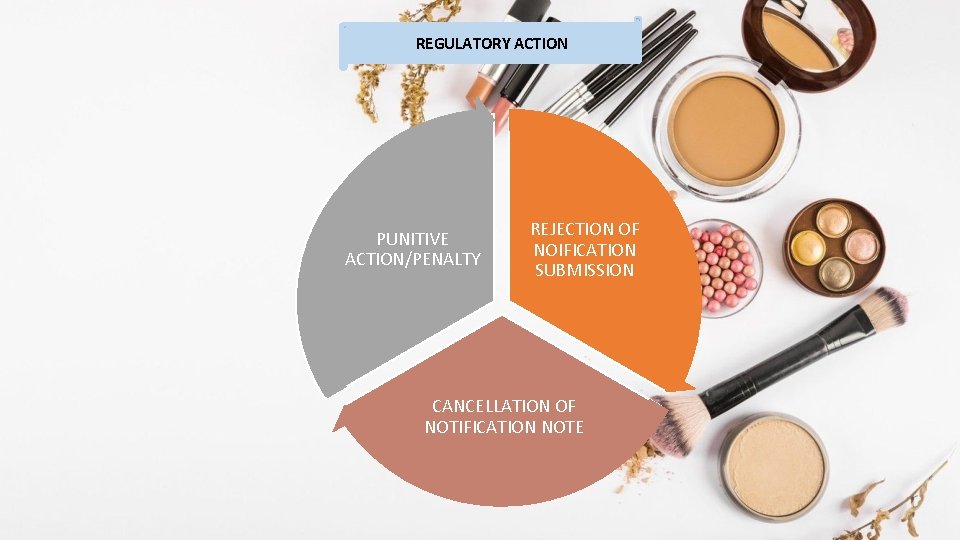
REGULATORY ACTION PUNITIVE ACTION/PENALTY REJECTION OF NOIFICATION SUBMISSION CANCELLATION OF NOTIFICATION NOTE

Notification withdrawal (Pemberitahuan pengeluaran) Shall not reinstated Invalid Product after withdrawn CNH Inform manufacturer/ contract manufacturer Inform NPRA

Notification exemption (Pengecualian pemberitahuan) Market sampling • One time entry of commercial products –Test market/aesthetic studies In-house validation • Process – product samples from R&D or production line • Demostration Products for Export (FEO) • Imported CPs – Packed, labelled etc. • Maintain records/documentation – NPRA In Transit • Notification not required – For products sent or brought from another country Product sold/supplied at Free Trade Zone/Leaving M’sia • Imported for sale/supply – through any sources • Company maintain proper documentation •
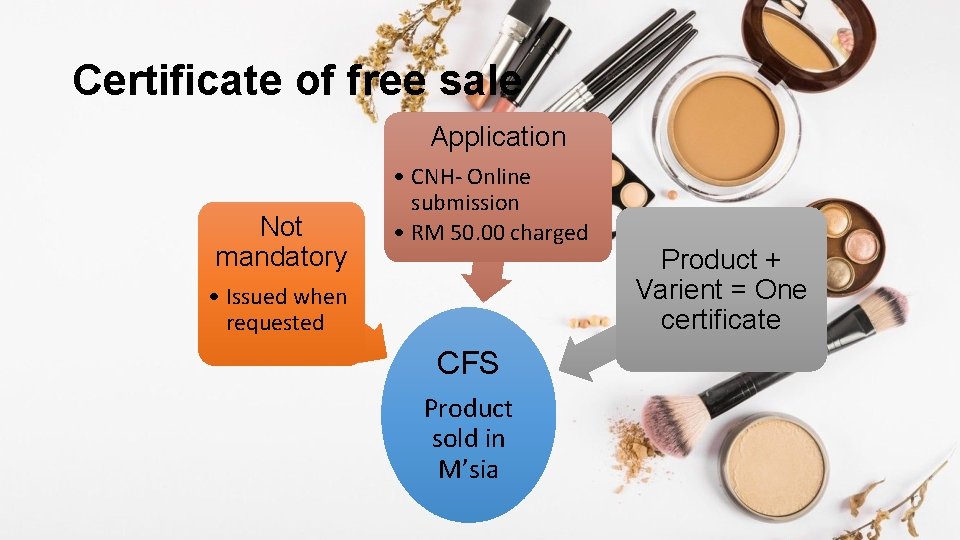
Certificate of free sale Application Not mandatory • CNH- Online submission • RM 50. 00 charged • Issued when requested CFS Product sold in M’sia Product + Varient = One certificate


Halal Logo Halal logo – used voluntarily, both local and export market v Procuct has been certified and approved by Department of Islamic Development (JAKIM)

PHARMACEUTICALS HALAL CERTIFICATE


Pharmaceutical refers to pharmaceutical products in the form of finished dosage including prescribed and non-prescribed medicinal products for human usage (biopharmaceutical, radiopharmaceutical, traditional medicine, dietary supplement and researched medicine) that has been registered with the Drug Control Authority under the Ministry of Health Malaysia.
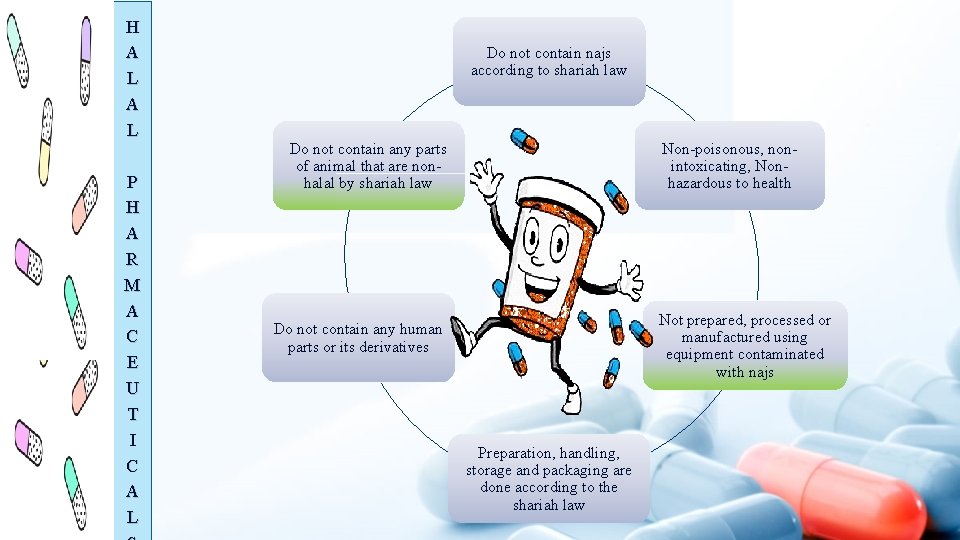
H A L P H A R M A C E U T I C A L Do not contain najs according to shariah law Do not contain any parts of animal that are nonhalal by shariah law Non-poisonous, nonintoxicating, Nonhazardous to health Not prepared, processed or manufactured using equipment contaminated with najs Do not contain any human parts or its derivatives Preparation, handling, storage and packaging are done according to the shariah law

PHARMACEUTICAL HALAL REQUIREMENTS QUALITY MANAGEMENT PACKAGING MANAGEMENT RESPONSIBILITY PERSONNEL HYGIENE HALAL ASSURANCE SYSTEM PREMISE AND EQUIPMENT GOOD MANUFACTURING PRACTICE PRODUCTION AND STORAGE PERSONNEL AND RESPONSIBILITY QUALITY CONTROL TRAINING MATERIALS AND PRODUCTION
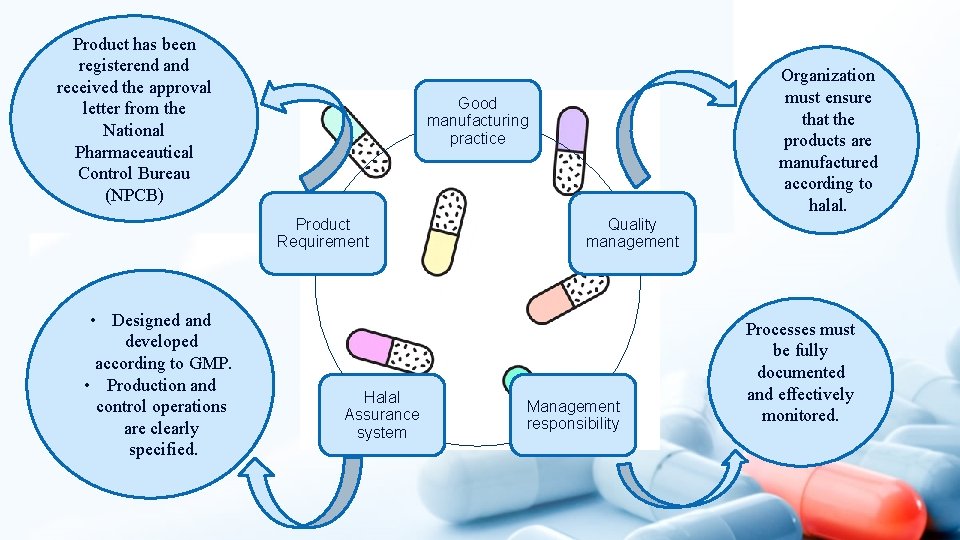
Product has been registerend and received the approval letter from the National Pharmaceautical Control Bureau (NPCB) Good manufacturing practice Product Requirement • Designed and developed according to GMP. • Production and control operations are clearly specified. Organization must ensure that the products are manufactured according to halal. Halal Assurance system Quality management Management responsibility Processes must be fully documented and effectively monitored.

Fundamentals for halal Pharmaceuticals In GMP Good manufacturing practice Product Requirement Halal Assurance system Quality management Management responsibility • Materials are clearly defined • Facilities and compliances are provided with the availability of : ü Qualified and trained personnel ü Adequate premises ü Dedicated equipment ü Correct materials ü Approved instructions and procedures ü Dedicated storage • Easily accessible history of the batch production • Distribution of products minimizes any risk to their halal integrity
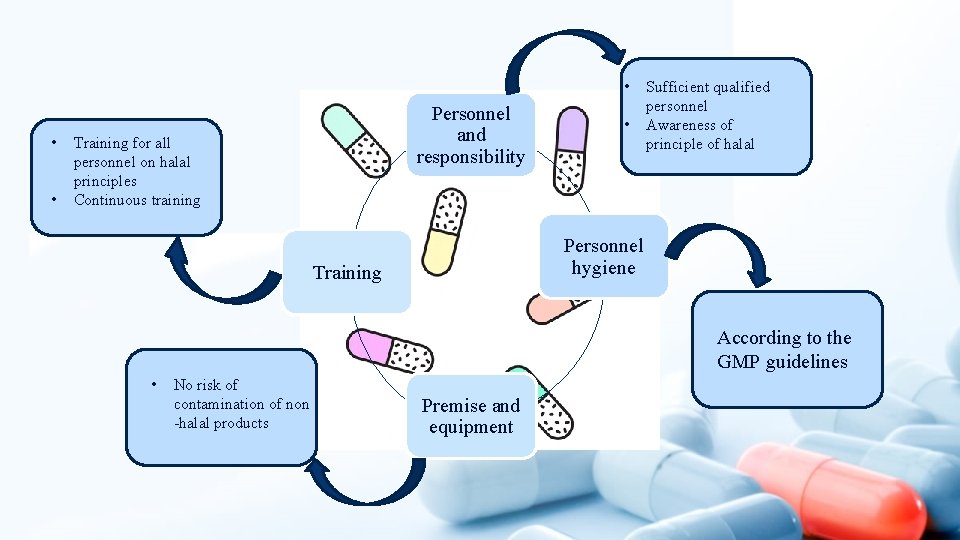
• • • Personnel and responsibility Training for all personnel on halal principles Continuous training • Sufficient qualified personnel Awareness of principle of halal Personnel hygiene Training According to the GMP guidelines • No risk of contamination of non -halal products Premise and equipment



THANK YOU!
- Slides: 23