Problems with the Bohr Model The Bohr model

Problems with the Bohr Model • The Bohr model applies only to one electron atoms. • The Bohr model doesn’t account for the observed spectra of multi-electron elements or ions. • The movement of electrons in atoms is much less clearly defined than Bohr allowed.

Quantum Mechanical Model • This is the current model of the atom • Proposed by Erwin Schrödinger (1887 -1961) – Schrödinger Equation

A Theory that Explains Electron Behavior • the quantum-mechanical model explains the manner electrons exist and behave in atoms • helps us understand predict the properties of atoms that are directly related to the behavior of the electrons – why some elements are metals while others are nonmetals – why some elements gain 1 electron when forming an anion, while others gain 2 – why some elements are very reactive while others are practically inert – and other Periodic patterns we see in the properties of 3 the elements
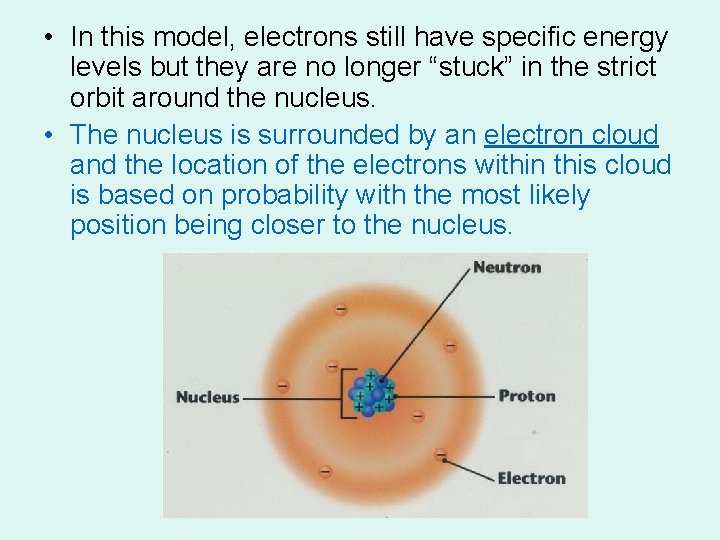
• In this model, electrons still have specific energy levels but they are no longer “stuck” in the strict orbit around the nucleus. • The nucleus is surrounded by an electron cloud and the location of the electrons within this cloud is based on probability with the most likely position being closer to the nucleus.

Atomic Orbitals- 3 -D sublevels within the electron cloud where the electrons are most likely to be found. • 4 types 1. 2. 3. 4. • *s orbitals* *p orbitals* *d orbitals* *f orbitals* Each individual orbital can only hold 2 e-s
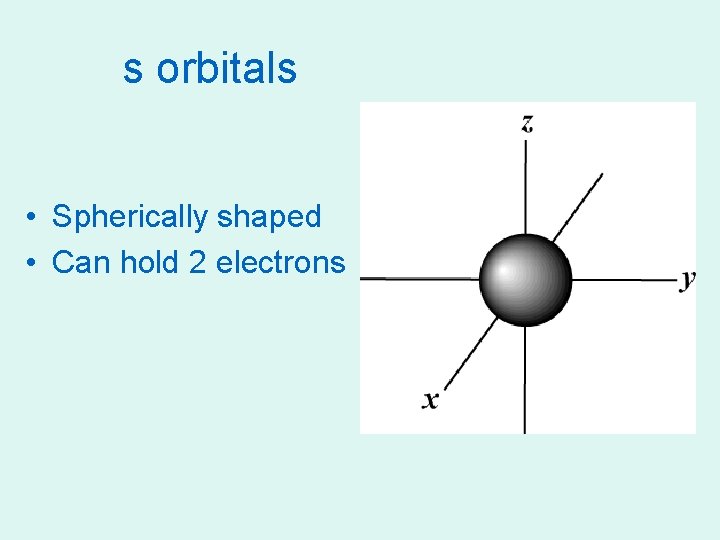
s orbitals • Spherically shaped • Can hold 2 electrons
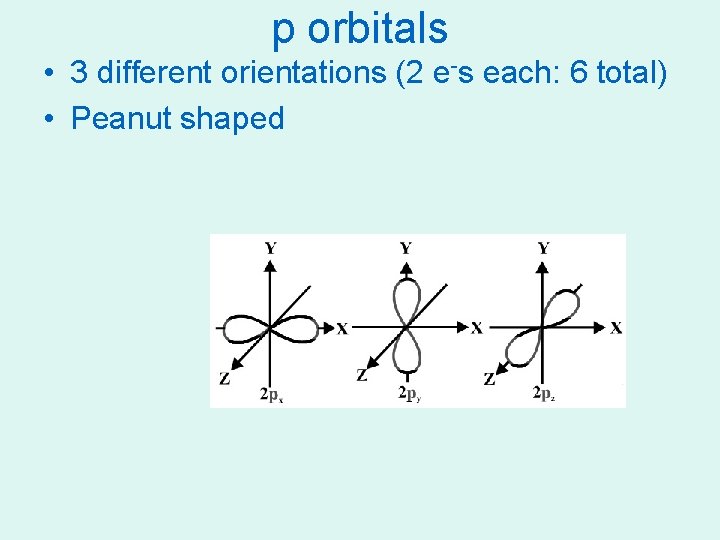
p orbitals • 3 different orientations (2 e-s each: 6 total) • Peanut shaped
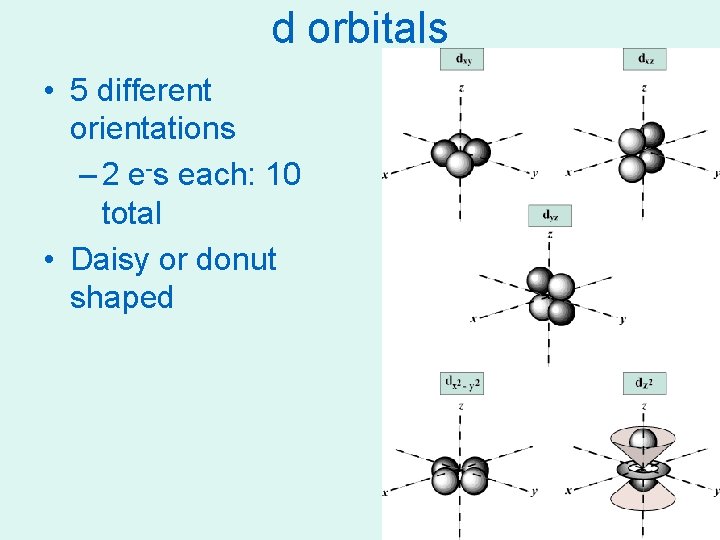
d orbitals • 5 different orientations – 2 e-s each: 10 total • Daisy or donut shaped
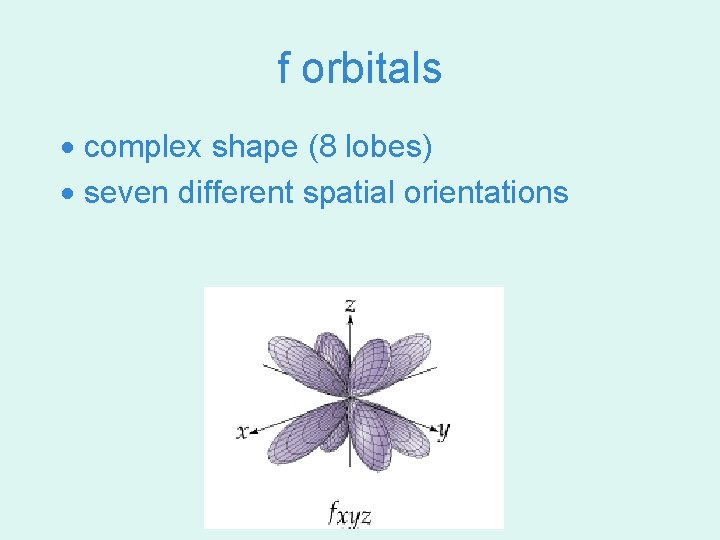
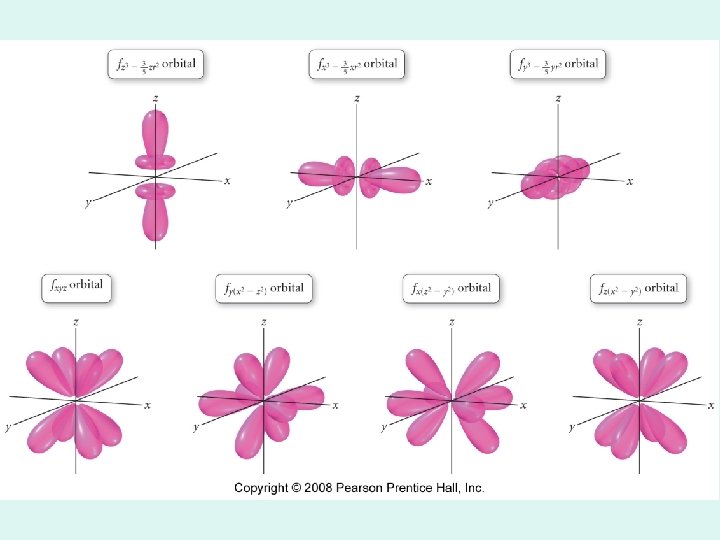
f orbitals · complex shape (8 lobes) · seven different spatial orientations


Identifying Orbitals • The orbitals are notated by the energy level and then the type of orbital – Ex. 1 s: is an s orbital in the first energy level • Each energy level has specific orbitals associated with it and therefore a specific number of electrons that it can hold

Principal Energy Level Number of Sublevels Type of Sublevel Number of ein the energy level n=1 1 1 s (1 orbital) 2 n=2 2 2 s (1 orbital) 2 p (3 orbitals) 8 n=3 n=4 3 4 3 s (1 orbital) 3 p (3 orbitals) 3 d (5 orbitals) 4 s, 4 p, 4 d, 4 f (7 orbitals) (2 in s and 2 x 3 in p) 18 (2 in s, 2 x 3 in p, and 2 x 5 in d) 32
- Slides: 13