Problems with the Bohr Model The Bohr model

Problems with the Bohr Model • The Bohr model could only predict the emission spectrum for hydrogen. Ø The Bohr model failed to predict the emission spectra for all of the other elements. Ø The Bohr model did not take into account the fact that electrons repel each other, thus causing shifts in the lines in the spectra. 1

Add to table of contents Orbital Diagram Pg. 26 QMM Model Pg. 27 2

QMM Model 3

The Quantum-Mechanical Model of the Atom • **created by Erwin Schrödinger (Write at the top of your paper!) He applied the mathematics of probability and the ideas of quantizing energy to the physics equations that describe waves, resulting in an equation that predicts the probability of finding an electron with a particular amount of energy at a particular location in the atom. Erwin Schrodinger 4

The Quantum-Mechanical Model of the Atom: the Schrodinger Equation 5
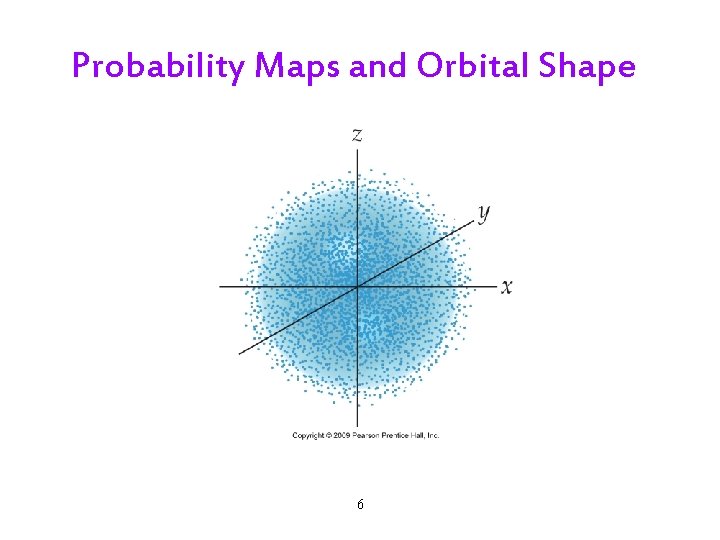
Probability Maps and Orbital Shape 6

The Quantum-Mechanical Model: Orbitals • The result is a map of regions in the atom that have a particular probability for finding the electron. • An orbital is a region where we have a very high probability of finding the electron when it has a particular amount of energy. üGenerally set at 90 or 95%. 7

Orbits vs. Orbitals Pathways vs. Probability 8

The Quantum-Mechanical Model: Quantum Numbers • The Principal Quantum Number, n, specifies the energy level for the orbital. • The number of electrons in each principal quantum number still follows the 2 n 2 rule. 9

The Quantum-Mechanical Model: Subshells • Each principal energy level (shell) has one or more sublevels (subshells). ü The number of subshells = the principal quantum number. • Each subshell is designated by a letter. ü s, p, d, f. • Each kind of subshell has orbitals with a particular shape. ü The shape represents the probability map. Ø 90% probability of finding electron in that region. 10
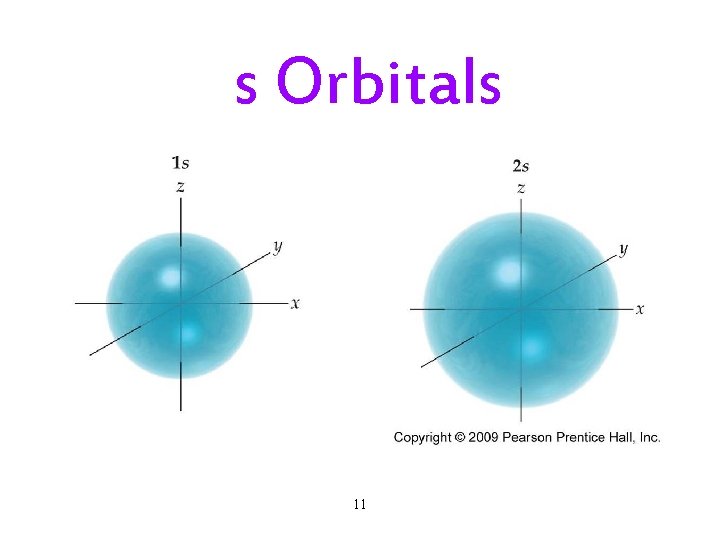
s Orbitals 11
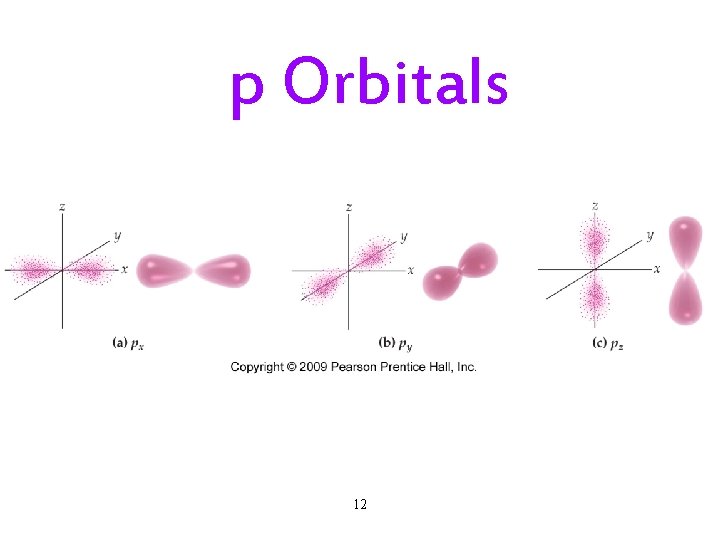
p Orbitals 12
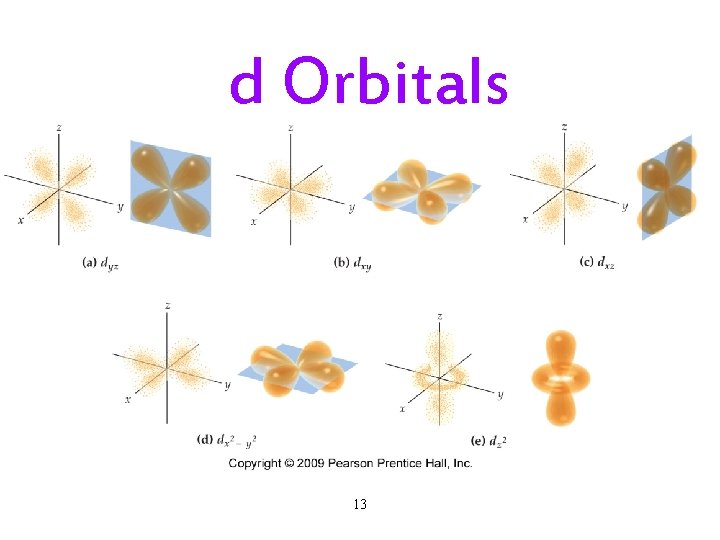
d Orbitals 13
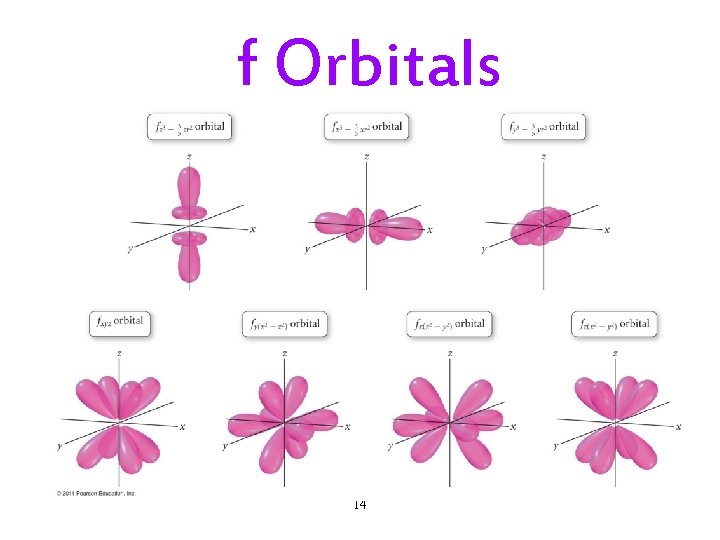
f Orbitals 14

Shells and Subshells 15

The Number of Sublevels on an Energy Level • The number of subshells on an energy level can be calculated by the n 2 rule. ün = the principal energy level number. ü 1 st energy level = (1)2 = 1 orbital (s only) ü 2 nd energy level = (2)2 = 4 orbitals (s + p) ü 3 rd energy level = (3)2 = 9 orbitals (s + p + d) 16

Subshells and Orbitals • The subshells of a principal shell have slightly different energies. ü s < p < d < f. • Each subshell contains one or more orbitals: ü s subshells have 1 orbital ü p subshells have 3 orbitals ü d subshells have 5 orbitals ü f subshells have 7 orbitals 17

Electron Configurations • The distribution of electrons into the various energy shells and subshells in an atom in its ground state is called its electron configuration. • Each energy shell and subshell has a maximum number of electrons it can hold (1 orbital = 2 e-). ü s = 2, p = 6, d = 10, f = 14. • Aufbau principle: place electrons in the shells and subshells in order of energy, from low to high 18
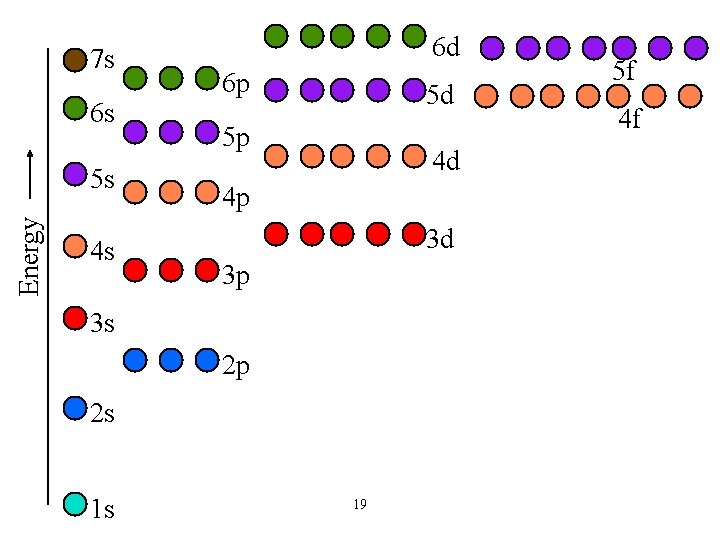
7 s 6 s Energy 5 s 4 s 6 d 6 p 5 d 5 p 4 d 4 p 3 d 3 p 3 s 2 p 2 s 1 s 19 5 f 4 f

Filling an Orbital with Electrons • Pauli Exclusion principle: Each orbital may have a maximum of 2 electrons with opposite spins. • Electrons spin on an axis. üGenerating their own magnetic field. • When two electrons are in the same orbital, they must have opposite spins. üSo their magnetic fields will cancel. 20
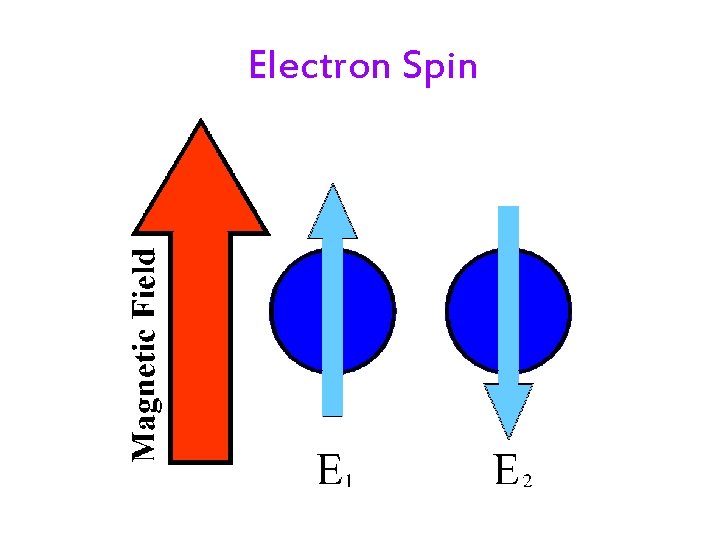
Electron Spin 21
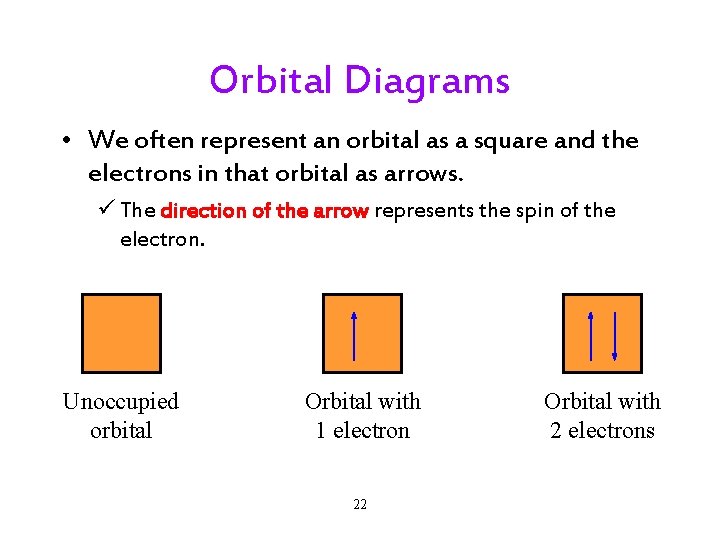
Orbital Diagrams • We often represent an orbital as a square and the electrons in that orbital as arrows. ü The direction of the arrow represents the spin of the electron. Unoccupied orbital Orbital with 1 electron 22 Orbital with 2 electrons

Filling an Orbital with Electrons • Hund’s rule: When filling orbitals that have the same energy, place one electron in each before completing pairs and they must have the same spin. 23
- Slides: 23