Problem Types Ka Kb p H p OH

Problem Types Ka, Kb, p. H, p. OH Percent Ionization neutralization 1
![Consider a 0. 035 M hydrochloric acid solution. + − Calculate [H ], [OH Consider a 0. 035 M hydrochloric acid solution. + − Calculate [H ], [OH](http://slidetodoc.com/presentation_image_h/1e6602331ac832f545174db369a173d4/image-2.jpg)
Consider a 0. 035 M hydrochloric acid solution. + − Calculate [H ], [OH ], p. H, p. OH Yes, calculator 2
![Consider a 0. 035 M hydrochloric acid solution. Calculate [H+], [OH−], p. H, p. Consider a 0. 035 M hydrochloric acid solution. Calculate [H+], [OH−], p. H, p.](http://slidetodoc.com/presentation_image_h/1e6602331ac832f545174db369a173d4/image-3.jpg)
Consider a 0. 035 M hydrochloric acid solution. Calculate [H+], [OH−], p. H, p. OH • Because HCl is a strong acid, you can assume all of the 0. 035 M of HCl has ionized into 0. 035 M H+ • Calculate p. H = −log[0. 035] = 1. 46 • It’s an easy calculation from p. H to p. OH ‣ Since p. H + p. OH = 14, thus p. OH = 14 - 1. 46 = 12. 54 ‣ Since [OH−] = 10−p. OH thus [OH−] = 2. 86 x 10− 13 • Alternatively you could calculate the [OH−] from the [H+] ‣ [H+] [OH−] = 1 x 10− 14 ‣ Thus ‣ And p. OH = −log[OH−] = −log(2. 86 x 10− 13) = 12. 54 3

Calculate the molarity of a nitric acid solution with a p. H of 2. 34 What is the [OH−]? 4

Calculate the molarity of a nitric acid solution with a p. H of 2. 34 What is the [OH−]? • Calculate [H+] from p. H 10− 2. 34 = [H+] = 0. 0046 M • Remember HNO 3 is a strong acid, you can assume the 0. 0046 M of H+ represents completely ionized HNO 3 • Thus the concentration of the HNO 3 is the same, 0. 0046 M • Calculate [OH-] from p. OH (or go the Kw route) ‣ 10− 11. 66 = [OH-] = 2. 2 x 10 -12 M 5
![− [OH ] Calculate the for a Na. OH solution with a p. H − [OH ] Calculate the for a Na. OH solution with a p. H](http://slidetodoc.com/presentation_image_h/1e6602331ac832f545174db369a173d4/image-6.jpg)
− [OH ] Calculate the for a Na. OH solution with a p. H of 11. 89 Calculate the mass of Na. OH that was dissolved to produce 250 ml of this solution. Na. OH = 40 g/mol Calculate the molarity of the hydronium ion of this solution. 6
![Calculate the [OH−] for a Na. OH solution with a p. H of 11. Calculate the [OH−] for a Na. OH solution with a p. H of 11.](http://slidetodoc.com/presentation_image_h/1e6602331ac832f545174db369a173d4/image-7.jpg)
Calculate the [OH−] for a Na. OH solution with a p. H of 11. 89 Calculate the mass of Na. OH that was dissolved to produce 250 ml of this solution. Calculate the hydronium ion of this solution. • Change the p. H to p. OH and “undo” it. ‣ p. OH = 14 -11. 89 = 2. 11 ‣ −log[OH−] = 2. 11 ‣ Solve 10− 2. 11 ‣ to get [OH−] = 7. 8 x 10− 3 M • Alternatively, you could undo the p. H, solve for [H+] and then use [OH−] [H+] = 1 x 10− 14 to solve for [OH−] ‣ −log[H+] = 11. 89 ‣ Solve 10− 11. 89 to get [H+] = 1. 3 x 10− 12 ‣ substitute into Kw = [OH−] [H+] ‣ 1 x 10− 14 = [OH−] [1. 3 x 10− 12] = ‣ [OH−] = 7. 8 x 10− 3 M 7
![Calculate the [OH−] for a Na. OH solution with a p. H of 11. Calculate the [OH−] for a Na. OH solution with a p. H of 11.](http://slidetodoc.com/presentation_image_h/1e6602331ac832f545174db369a173d4/image-8.jpg)
Calculate the [OH−] for a Na. OH solution with a p. H of 11. 89 Calculate the mass of Na. OH that was dissolved to produce 250 ml of this solution. Calculate the hydronium ion of this solution. • since [OH−] = 7. 8 x 10− 3 M • Thus [Na. OH] = 7. 8 x 10− 3 M x 0. 25 L = 0. 0019 mol • 0. 0019 mo x 40 g/mol = 0. 078 g Na. OH dissolved • Remember, hydronium is H 3 O+ or H+ • From the p. H, solve 10− 11. 89 ‣ to get [H+] = [H 3 O+] = 1. 3 x 10− 12 • Or substitute [OH−] into Kw = [OH−] [H+] ‣ 1 x 10− 14 = [7. 8 x 10− 3] [H+] ‣ [H+] = 1. 3 x 10− 12 to solve for [H+] 8
Calculate the p. H of a 0. 050 M barium hydroxide solution. • Want to avoid a mistake? • If so, it might be be helpful if you write the formula for barium hydroxide first 9

Calculate the p. H of a 0. 050 M barium hydroxide solution. • First write the formula for barium hydroxide ✓ Ba(OH)2 • Consider the stoichiometry of the dissociation Rx. ✓ Ba(OH)2 → Ba 2+ + 2 OH− • Since Ba(OH)2 and thus concentration of − OH 2+ [Ba ] is 0. 05 M the = 0. 10 M • Calculate p. OH = −log[0. 10 M] = 1 ✓ Since p. H = 14 − p. OH = 13. 00 • Alternatively, use Kw = [OH−] [H+] to solve for , [H+] then calculate p. H. 10

Calculate the p. H of a 0. 20 M solution of hypochlorous acid. Ka = 3. 0 x 10− 8 • Write a reaction for the ionization of hypochlorous acid. • Write the Ka expression.

Calculate the p. H of a 0. 20 M solution of hypochlorous acid. Ka = 3. 0 x 10− 8 • Write a reaction for the ionization of hypochlorous acid. ✓ HCl. O ⇋ H+ + Cl. O− • Write the Ka expression.

Calculate the p. H of a 0. 20 M solution of hypochlorous acid. Ka = 3. 0 x 10− 8 • Write a reaction for the ionization of hypochlorous acid. ✓ HCl. O ⇋ H+ + Cl. O− • Write the Ka expression. • Construct an ricebox put in the concentrations we already know. . . next slide please

Calculate the p. H of a 0. 20 M solution of hypochlorous acid. Ka = 3. 0 x 10− 8 • We can assume that the initial [H+] = 0, because the amount of H+ in pure water is too small compared to any H+ caused by the presence of the hypochlorous acid. • Since we don’t know what the changes are. . ✓ call them x • And thus the equilibrium concentrations? I C E HCl. O 0. 20 ⇋ H+ + ~0 Cl. O− 0

Calculate the p. H of a 0. 20 M solution of hypochlorous acid. Ka = 3. 0 x 10− 8 I C E HCl. O 0. 20 -x 0. 20 - x ⇋ H+ + ~0 +x x Cl. O− 0 +x x • How will we solve? ✓ UGH. . . this ✓ Solving is a quadratic for x = [H+] = 7. 747 x 10− 5 M • But wait. . can we avoid the use of the quadratic? ?

Calculate the p. H of a 0. 20 M solution of hypochlorous acid. I C E HCl. O ⇋ 0. 20 -x 0. 20 - In x AP we ~0. 20 H+ + Cl. O− ~0 0 +x +x x ALWAYS be x able to ignore the will -x when the alternative means using the quadratic • Let’s consider how significant figures allow us to avoid the quadratic formula. ✓ We can consider the −x to be insignificant compared to the 0. 20 M for HCl. O, thus the equilibrium [Cl. O−] = ~0. 20 M = [H+] = 7. 7 x 10− 5 M • Use the [H+] to calculate p. H ✓ p. H = 4. 11 ✓x

Calculate the p. H of a 0. 20 M solution of ammonia, p. Kb = 4. 74. • Write a reaction for the ionization of ammonia. • Write the Kb expression. • Construct a ricebox put in the concentrations we know, calculate the others, ignore insignificant x’s

Calculate the p. H of a 0. 20 M solution of ammonia, p. Kb = 4. 74. • Write a reaction for the ionization of ammonia. ✓ NH 3 + H 2 O ⇋ NH 4+ • Write the Kb expression. ✓What is the value for Kb? + OH−

Calculate the p. H of a 0. 20 M solution of ammonia. • Write a reaction for the ionization of ammonia. ✓ NH 3 + H 2 O ⇋ NH 4+ + OH− • Write the Kb expression, p. Kb = 4. 74 ✓ Kb = -4. 74 10 = 1. 8 x − 5 10

Calculate the p. H of a 0. 20 M solution of ammonia. • Write a reaction for the ionization of ammonia. ✓ NH 3 + H 2 O ⇋ NH 4+ + OH− • Write the Kb expression, p. Kb = 4. 74 ✓ Kb = -4. 74 10 = 1. 8 x − 5 10 if you did use the quadratic, x = 1. 8884 x 10− 3, p. OH = 2. 72 , p. H = 11. 28 This is an x 2 problem!

After preparing a 0. 10 M solution of nitrous acid, the p. H was 2. 17, calculate the Ka for this acid. • Write the reaction for the equilibrium dissociation of nitrous acid in water. • Write the Ka expression. 21

After preparing a 0. 10 M solution of nitrous acid, the p. H was 2. 17, calculate the Ka for this acid. • Write the reaction for the equilibrium dissociation+ of nitrous acid in water. − ‣ HNO 2 ⇋ H + NO 2 • Write the Ka expression. • “Do we know + [H ] or [NO 2 −]” 22

After preparing a 0. 10 M solution of nitrous acid, the p. H was 2. 17, calculate the Ka for this acid. • Write the reaction for the equilibrium dissociation+ of nitrous acid in water. − ‣ HNO 2 ⇋ H + NO 2 • Write the Ka expression. • “Un-log” the p. H to determine the equilibrium + [H ] ‣ 6. 8 x 10− 3 M • What is the [NO 2−] ? ‣ the same as [H+] 23

After preparing a 0. 10 M solution of nitrous acid, the p. H was 2. 17, calculate the Ka for this acid. • What’s the equilibrium concentration of NO 2−? as H+ • What do you know about the initial [H+] and [NO 2−]? ✓ Assume there is none in the container to start. ✓ Why do we assume the initial [H+] = 0? Isn’t there 1 x 10− 7 M in water? ‣ 1 x 10− 7 M is so much smaller than any amounts of that will form due to the presence of the acid so sig figs allow the 1 x 10− 7 M to be considered ~0 ✓ same I C E HNO 2 0. 1 ⇋ H+ + ~0 6. 8 x 10− 3 M NO 2− 0 24

After preparing a 0. 10 M solution of nitrous acid, the p. H was 2. 17, calculate the Ka for this acid. • Now you can calculate the change row. • Then the equilibrium concentration of HNO 2 = 0. 0932 M • Fill in the equilibrium expression and calculate Ka R HNO 2 ⇋ H+ + I 0. 1 0 C − 6. 8 x 10− 3 M +6. 8 x 10− 3 M E 0. 10 - 6. 8 x 10− 3 M = 0. 0932 M NO 2− 0 +6. 8 x 10− 3 M In this case, we know the “x” so we should put it in to calculate Calculating Ka without the subtraction = 4. 6 x 10 -4 25

After preparing a 0. 10 M solution of nitrous acid, the p. H was 2. 17, calculate the Ka for this acid. What percentage of the acid is ionized in this 0. 10 M solution? • Remember that % is part out of total. ‣ What part represents the part that is ionized? ‣ What part represents total acid? I C E HNO 2 0. 1 − 6. 8 x 10− 3 M 0. 0932 M ⇋ H+ + 0 +6. 8 x 10− 3 M NO 2− 0 +6. 8 x 10− 3 M

After preparing a 0. 10 M solution of nitrous acid, the p. H was 2. 17, calculate the Ka for this acid. What percentage of the acid is ionized in this 0. 10 M solution? • Remember that % is part out of total. ‣ What part represents the part that is ionized? ‣ What part represents total acid? • Calculate I C E HNO 2 0. 1 − 6. 8 x 10− 3 M 0. 0932 M ⇋ H+ + 0 +6. 8 x 10− 3 M NO 2− 0 +6. 8 x 10− 3 M

Calculate the Ka of lactic acid, HC 3 H 5 O 3. The acid is 3. 7% dissociated in a 0. 100 M solution 28
Calculate the Ka of lactic acid, HC 3 H 5 O 3. The acid is 3. 7% dissociated in a 0. 100 M solution • Write the equilibrium reaction and Ka expression. • Set up a RICE box • Since we are told in the problem that there is 3. 7% dissociation, we can calculate [H+] R HC 3 H 5 O 3 ⇋ H + + C 3 H 5 O 3− I C E 29

Calculate the Ka of lactic acid, HC 3 H 5 O 3. The acid is 3. 7% dissociated in a 0. 100 M solution • Write the equilibrium reaction and Ka expression. • HC 3 H 5 O 3 �H+ + C 3 H 5 O 3− • Set up a RICE box • Since we are told in the problem that there is 3. 7% dissociation, we know that the x is 3. 7% of 0. 100 M ‣x = 0. 0037 • This allows you to fill in the remainder of the RICE box. I • Using the E line of the RICE box C allows you to calculate Ka E R HC 3 H 5 O 3 ⇋ H + + C 3 H 5 O 3− 0. 1 ~0 0 - 0. 0037 +0. 0037 0. 0963 0. 0037 30

The p. H of a 0. 060 M solution of pyridine, C 5 H 5 N is 9. 00. Calculate the Kb for this base. What is the percent dissociation of this solution? 31

The p. H of a 0. 060 M solution of pyridine, C 5 H 5 N is 9. 00. Calculate the Kb for this base. What is the percent dissociation of this solution? • Write the ionization reaction for pyridine in water. • Write the Kb expression, • Make a rice box. 32

The p. H of a 0. 060 M solution of pyridine, C 5 H 5 N is 9. 00. Calculate the Kb for this base. What is the percent dissociation of this solution? • C 5 H 5 N + H 2 O �C 5 H 5 NH+ + OH− • Set up an RICE box • p. H = 9, p. OH = 5, thus [OH−] = 1 x 10− 5 33

The p. H of a 0. 060 M solution of pyridine, C 5 H 5 N is 9. 00. Calculate the Kb for this base. What is the percent dissociation of this solution? • C 5 H 5 N + H 2 O �C 5 H 5 NH+ + OH− • Set up an RICE box • p. H = 9, p. OH = 5, thus [OH−] = 1 x 10− 5 R C 5 H 5 N → C 5 H 5 NH+ + OH− I 0. 06 0 ~0 C - 1 x 10− 5 + 1 x 10− 5 E 0. 06 - 1 x 10 ~ 0. 06 − 5 Why is [OH−] ~ 0 ? You could argue that since it is an aqueous solution, that there [OH−] = 1 x 10− 7 OH−, but since the base will contribute more than that, we can ignore the OH− contribution from the water. 34

Calculate the volume of 0. 034 M potassium hydroxide required to neutralize 25. 0 ml of 0. 050 M nitric acid. • Write the overall neutralization reaction. • Write the net ionic reaction 35

Calculate the volume of 0. 034 M potassium hydroxide required to neutralize 25. 0 ml of 0. 050 M nitric acid. • KOH + HNO 3 → H 2 O + KNO 3 • OH− + H+ → H 2 O 36

Calculate the volume of 0. 034 M potassium hydroxide required to neutralize 25. 0 ml of 0. 050 M nitric acid. • KOH + HNO 3 → H 2 O + KNO 3 • OH− + H+ → H 2 O 37

Calculate the volume of 0. 034 M potassium hydroxide required to neutralize 25. 0 ml of 0. 050 M nitrous acid. Ka = 4. 9 x 10 -4 • Write the overall neutralization reaction. • Write the net ionic reaction 38

Calculate the volume of 0. 034 M potassium hydroxide required to neutralize 25. 0 ml of 0. 050 M nitrous acid. Ka = 4. 9 x 10 -4 • KOH + HNO 2 → H 2 O + KNO 2 • OH− + HNO 2 → H 2 O + NO 2− • Though the net ionic equation (of WA + SB) is different (than SA + SB), you must represent the WA as the molecule not simply as H+, since most of the WA exists in the water at the start of the neutralization as HNO 2. • However, solving for the quantity of base required for the neutralization of a weak acid would be exactly the same procedure. 39

Calculate the volume of 0. 034 M potassium hydroxide required to neutralize 25. 0 ml of 0. 050 M nitrous acid. Ka = 4. 9 x 10 -4 • KOH + HNO 2 → H 2 O + KNO 2 • OH− + HNO 2 → H 2 O + NO 2− • Though the net ionic equation is different, • solving for the quantity of base required for the neutralization of a weak acid would be exactly the same procedure. Do NOT be distracted by the Ka. There is no reason to use it. In the end, the WA will give up ALL of it H+’s 40

Calculate the volume of 0. 0045 M perchloric acid required to neutralize 65. 0 ml of 0. 0072 M barium hydroxide. • Write the overall neutralization reaction. • Write the net ionic reaction 41

Calculate the volume of 0. 0045 M perchloric acid required to neutralize 65. 0 ml of 0. 0072 M barium hydroxide. • Ba(OH)2 + 2 HCl. O 4 → 2 H 2 O + Ba(Cl. O 4)2 • OH− + H+ → H 2 O • Beware of the “buy-one-get-two” base • Each mol (or molarity) of base provides double moles (or molarity) of OH- 42

Calculate the volume of 0. 0045 M perchloric acid required to neutralize 65. 0 ml of 0. 0072 M barium hydroxide. • Ba(OH)2 + 2 HCl. O 4 → 2 H 2 O + Ba. Cl. O 4 • OH− + H+ → H 2 O • Beware of the “buy-one-get-two” base • Each mol (or molarity) of base provides double moles (or molarity) of OH- 43

Calculate the p. H after mixing 35 ml of 0. 025 M hydrochloric acid with 40 ml of 0. 030 M Na. OH • Write the neutralization reaction. • Determine the mmol of the SA and the mmol of SB • What do you notice? What’s left over after neutralization? 44

Calculate the p. H after mixing 35 ml of 0. 025 M hydrochloric acid with 40 ml of 0. 030 M Na. OH • Write the neutralization reaction. • H+ + OH- → H 2 O • What do you notice? What’s left over after neutralization? 45

Calculate the p. H after mixing 55 ml of 0. 025 M hydrochloric acid with 40 ml of 0. 030 M Na. OH • Remember the SA + SB neutralization reaction. ✓H+ + OH- → H 2 O • Determine the mmol of the SA and the mmol of SB • What do you notice? What’s left over after neutralization? 46

Calculate the p. H after mixing 55 ml of 0. 025 M hydrochloric acid with 40 ml of 0. 030 M Na. OH • Remember the SA + SB neutralization reaction. ✓H+ + OH- → H 2 O • Determine the mmol of the SA and the mmol of SB • What do you notice? What’s left over after neutralization? 47

Calculate the p. H after mixing 48 ml of 0. 025 M hydrochloric acid with 40 ml of 0. 030 M Na. OH • Determine the mmol of the SA and the mmol of SB • What do you notice? What’s left over after neutralization? 48

Calculate the p. H after mixing 48 ml of 0. 025 M hydrochloric acid with 40 ml of 0. 030 M Na. OH • Let’s think about the overall SA + SB neutralization reaction. ✓HCl + Na. OH → H 2 O + Na. Cl ✓HCl + Na. OH → H 2 O + Na+ + Cl • What species are in the beaker after this pour together? ✓just water and two pathetics ✓and so the p. H? ? 49

Calculate the p. H after mixing 48 ml of 0. 025 M hydrochloric acid with 40 ml of 0. 030 M Na. OH • Let’s think about the overall SA + SB neutralization reaction. ✓HCl + Na. OH → H 2 O + Na. Cl ✓HCl + Na. OH → H 2 O + Na+ + Cl− • What species are in the beaker after this pour together? ✓just water and two pathetics ✓and so the p. H? ? ★p. H = 7 50

Let’s summarize what you can do so far • Write equations to represent the reaction of SA, SB, WA or WB in water. • Calc [H+], [OH-], p. H, p. OH of a SA or SB ‣ Given molarity, (no need for volume) • Calc [H+], [OH-], % ionized, p. H, p. OH of a WA or WB. . . x 2 problem ‣ Given molarity, given Ka or Kb, (no need for volume) • Calc Ka or Kb given p. H or % ionization of a WA or WB ‣ Given molarity, (no need for volume) • Write an net ionic equation to represent the neutralization of SA + SB, WB + SA • Calculate volume of SA or SB needed to neutralize B or A ‣ watch out for “buy-one-get-more” • Calculate [H+], [OH-], p. H, p. OH after any amount of SA mixed with SB ‣ think limiting reactant (or excess) and pour together total volume 51

Calculate the p. H of 100. ml of 0. 50 M hydrofluoric acid Ka of HF = 6. 8 x -4 10 • Write out the equilibrium reaction of the ionization of HF 52

Calculate the p. H of 100. ml of 0. 50 M hydrofluoric acid Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + F− • This is an x 2 problem 53

Calculate the p. H of 100. ml of 0. 50 M hydrofluoric acid Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + F− • This is an x 2 problem 54

Let’s think about what’s in the beaker after mixing 100. ml of 0. 50 M hydrofluoric acid with 50. 0 ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • Write out the equilibrium reaction of the ionization of HF, NOT the neutralization reaction 55

Calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 50. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + F− • Resist the temptation of writing the Neutralization Reaction, because we do not know the equilibrium constant of this neutralization reaction • HF + OH− → H 2 O + F− 56

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 50. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + F− 57

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 150. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + OH− 58

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 150. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + F− • Resist the temptation of writing the Neutralization Reaction, because we do not know the equilibrium constant of this neutralization reaction • HF + OH− → H 2 O + F− 59

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 150. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + F− • Resist the temptation of writing the Neutralization Reaction, because we do not know the equilibrium constant of this neutralization reaction • HF + OH− → H 2 O + F− 60

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 125. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + OH− 61

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 125. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + OH− • Resist the temptation of writing the Neutralization Reaction, because we do not know the equilibrium constant of this neutralization reaction • HF + OH− → H 2 O + F− 62

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 125. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + OH− • Resist the temptation of writing the Neutralization Reaction, because we do not know the equilibrium constant of this neutralization reaction • HF + OH− → H 2 O + F− 63

Let’s think about what’s in the beaker after mixing 100. ml of 0. 50 M hydrofluoric acid with 250. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • Write out any relevant reactions that you should be thinking about when doing this problem to this problem 64

Let’s think about what’s in the beaker after mixing 100. ml of 0. 50 M hydrofluoric acid with 250. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • KOH + HF → H 2 O + K+ + F− • Water, a pathetic, and F−. • How does F− react with water? 65

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 250. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • H 2 O + F− → HF + OH− • F− is a weak base • What is the concentration of this weak base? 66

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 250. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • H 2 O + F− → HF + OH− • F− is a weak base Wait. . . we need a Kb for this reaction. and this problem becomes an x 2 problem 67

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 250. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • H 2 O + F− → HF + OH− • F− is a weak base 68

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 300. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + OH− 69

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 300. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + OH− • yikes, we have gone beyond the neutralization 70

Let’s calculate the p. H after mixing 100. ml of 0. 50 M hydrofluoric acid with 300. ml of 0. 20 M KOH Ka of HF = 6. 8 x -4 10 • HF ⇄ H+ + OH− • yikes, we have gone beyond the neutralization • 10 mmol OH− 71

Let’s summarize what you can do so far • Calc p. H of a WA or WB. . . this is an x 2 problem ‣ Given molarity, given Ka or Kb, (no need for volume) • Calc p. H of a WA or WB. . . this is an x 2 problem ‣ Given molarity, (no need for volume) • Write an net ionic equation to represent the neutralization of SA + SB, WB + SA • Calculate volume of SA or SB needed to neutralize B or A ‣ watch out for “buy-one-get-more” • Calculate [H+], [OH-], p. H, p. OH after any amount of SA mixed with SB ‣ think limiting reactant (or excess) and pour together total volume 72
Clicker Questions Weak Acids 73

For a certain weak acid, Ka = 2. 0 x What is Kb for its conjugate base? 1. 2. 3. 4. 5. 6. 4. 5 x − 8 5. 0 x 10 − 9 1. 0 x 10 − 10 4. 0 x 10 − 10 5. 0 x 10 − 5. 0 x 10 19 − 10 3 − 5 10. No calculator 74
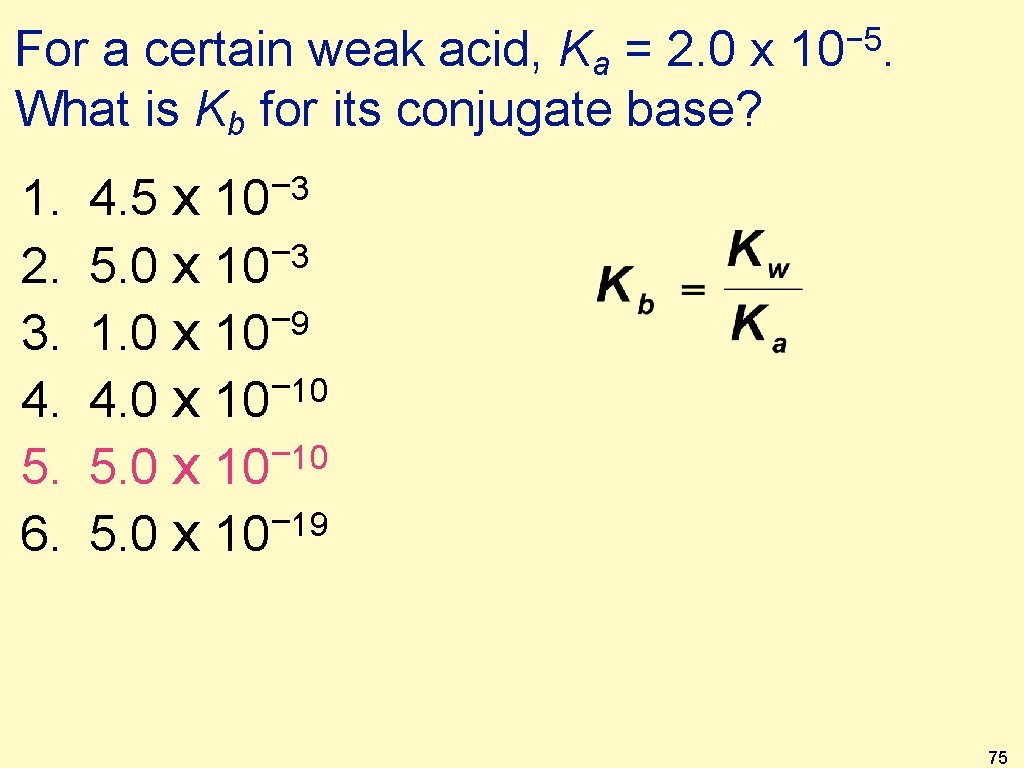
For a certain weak acid, Ka = 2. 0 x What is Kb for its conjugate base? 1. 2. 3. 4. 5. 6. − 5 10. 4. 5 x − 3 5. 0 x 10 − 9 1. 0 x 10 − 10 4. 0 x 10 − 10 5. 0 x 10 − 5. 0 x 10 19 − 10 3 75

Which acid is the weakest? 1. 2. 3. 4. 5. 6. chlorous nitrous acetic hypobromous they are all equally weak there is not enough information to distinguish acid chlorous nitrous acetic hypobromous p. Ka 1. 96 3. 35 4. 74 8. 6 76
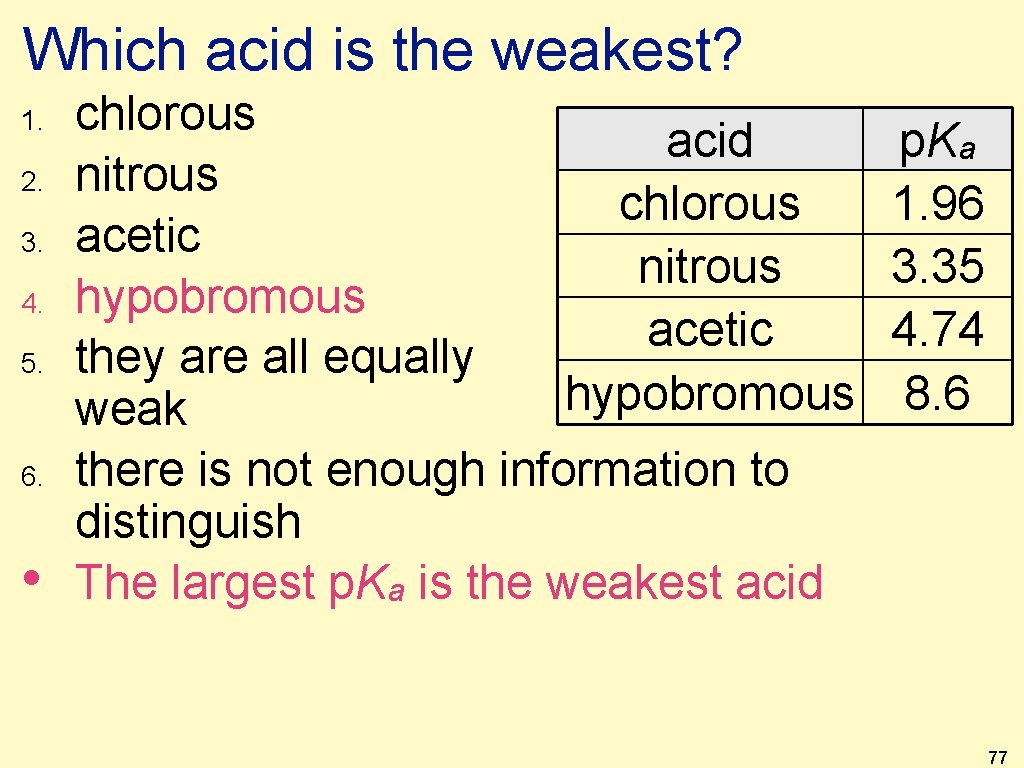
Which acid is the weakest? 1. 2. 3. 4. 5. 6. • chlorous acid nitrous chlorous acetic nitrous hypobromous acetic they are all equally hypobromous weak there is not enough information to distinguish The largest p. Ka is the weakest acid p. Ka 1. 96 3. 35 4. 74 8. 6 77
- Slides: 77