Problem Formulation to DoseResponse Advances via the ARA

Problem Formulation to Dose-Response: Advances via the ARA Beyond Science and Decisions Workshops Presented by: M. E. (Bette) Meek bmeek@uottawa. ca 1

NAS Committee: Advancing Risk Assessment - Background • “Chemical Risk assessment at a crossroads” • Facing substantial challenges, e. g. , – long delays in completing complex risk assessments, some of which take decades – lack of data – the need to address the many unevaluated chemicals in the marketplace • Recommendations for practical improvements to the U. S. Environmental Protection Agency (EPA) – Shorter (2 -5 y) and – longer (10 -20 y) term 2
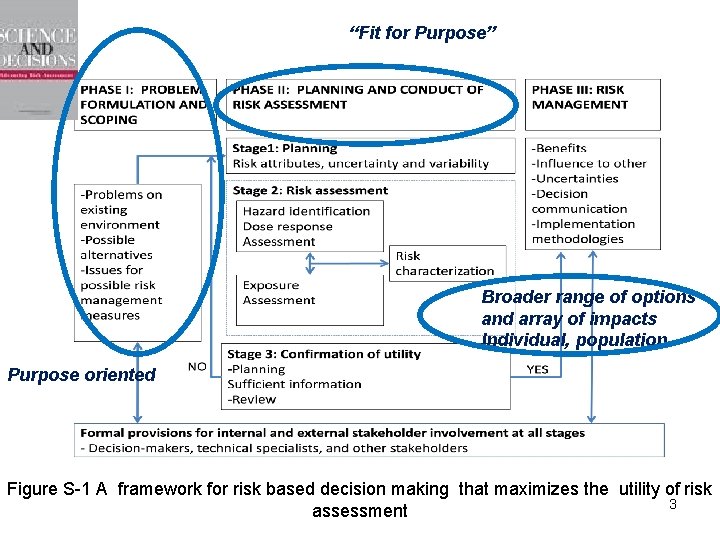
“Fit for Purpose” Broader range of options and array of impacts Individual, population Purpose oriented Figure S-1 A framework for risk based decision making that maximizes the utility of risk 3 assessment

Unified Approach to “Default” Dose Response Assessment; Use of “Defaults” • “A consistent approach to risk assessment for cancer and noncancer effects is scientifically feasible and needs to be implemented” • Predicated principally on the basis of perceived need to quantify risks for risk-risk and risk-benefit comparisons • “EPA should develop clear, general standards for the level of evidence needed to justify the use of agent-specific data and not resort to default” 4
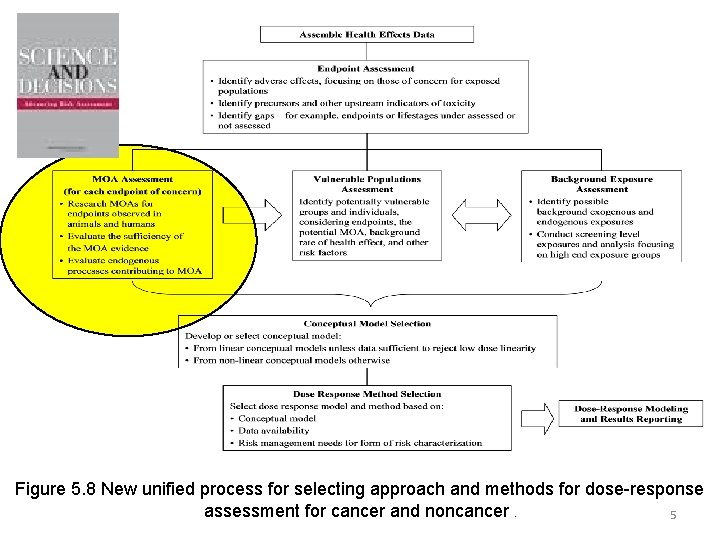
Figure 5. 8 New unified process for selecting approach and methods for dose-response assessment for cancer and noncancer. 5

Challenge: • Extending these considerations to address value of information and purpose specific assessment strategies including mode of action (MOA) understanding that: – More efficient assessment as a basis to address the many unevaluated chemicals in the marketplace requires: • Moving to more predictive, mode of action based approaches & tiered assessment strategies • Recognizing the importance of interdisciplinary collaboration/communication/engagement – E. g. , significant contributions of EPA and the international community in considering adequacy of data in moving from default to more progressive MOA based approaches 6
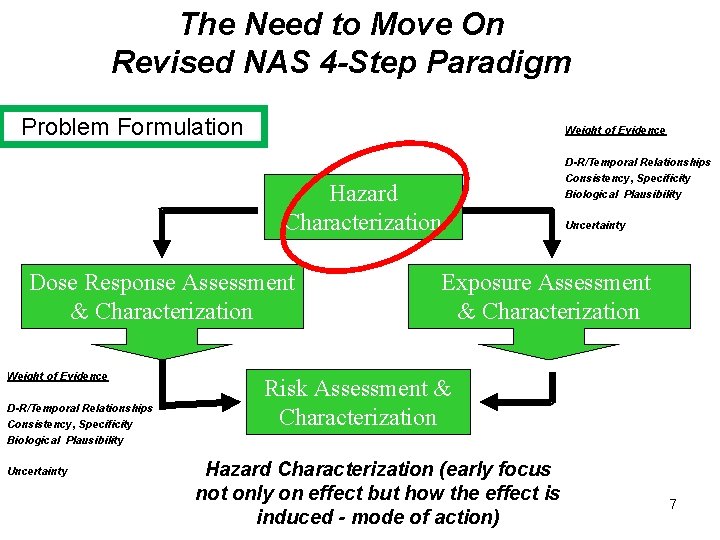
The Need to Move On Revised NAS 4 -Step Paradigm Problem Formulation Weight of Evidence Hazard Characterization Dose Response Assessment & Characterization Weight of Evidence D-R/Temporal Relationships Consistency, Specificity Biological Plausibility Uncertainty Exposure Assessment & Characterization Risk Assessment & Characterization Hazard Characterization (early focus not only on effect but how the effect is induced - mode of action) 7
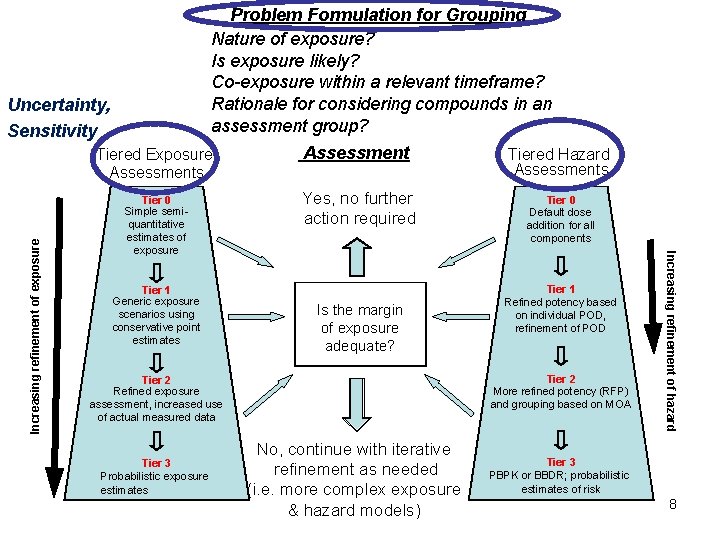
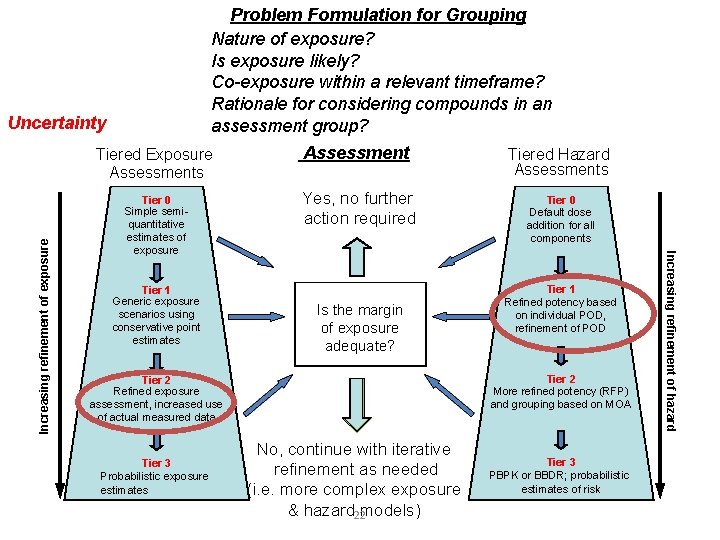
Problem Formulation for Grouping Nature of exposure? Is exposure likely? Co-exposure within a relevant timeframe? Rationale for considering compounds in an assessment group? Tiered Exposure Assessments Assessment Tiered Hazard Assessments Tier 0 Simple semiquantitative estimates of exposure Yes, no further action required Tier 0 Default dose addition for all components Tier 1 Generic exposure scenarios using conservative point estimates Is the margin of exposure adequate? Tier 2 More refined potency (RFP) and grouping based on MOA Tier 2 Refined exposure assessment, increased use of actual measured data Tier 3 Probabilistic exposure estimates Tier 1 Refined potency based on individual POD, refinement of POD No, continue with iterative refinement as needed (i. e. more complex exposure & hazard models) Increasing refinement of hazard Increasing refinement of exposure Uncertainty, Sensitivity Tier 3 PBPK or BBDR; probabilistic estimates of risk 8
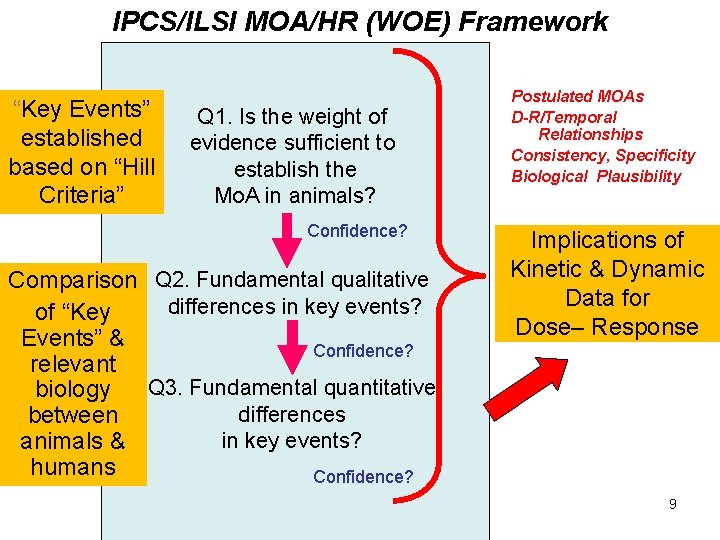
IPCS/ILSI MOA/HR (WOE) Framework “Key Events” established based on “Hill Criteria” Q 1. Is the weight of evidence sufficient to establish the Mo. A in animals? Confidence? Comparison Q 2. Fundamental qualitative differences in key events? of “Key Events” & Confidence? relevant Q 3. Fundamental quantitative biology differences between in key events? animals & humans Confidence? Postulated MOAs D-R/Temporal Relationships Consistency, Specificity Biological Plausibility Implications of Kinetic & Dynamic Data for Dose– Response 9

Engaging the Risk Assessment Community Evolution of MOA/HR Analysis • U. S. EPA ’ 99/’ 05 cancer guidelines • IPCS framework for WOE for MOA in animals – Sonich Mullin et al. , 2001 • ILSI RSI development of HR component and case studies – Meek et al. , 2003; Cohen et al. , 2004; Seed et al. , 2005 • IPCS revision of frameworks and addition of case studies – Boobis et al. , 2006; Boobis et al. , 2008 • ILSI RSI Key Events Dose Response Framework – Boobis et al. , 2010 • ECETOC Workshop - mapping MOAs & key events to chemical categories – Carmichael et al. (2011) • OECD Workshop (2010) – QSAR/MOA – Extending MOA/HR framework concepts as the coordinating construct between: • The ecological & health risk communities • The QSAR modelling and risk assessment communities • IPCS coordinating steering group on mode of action (constituted in October, 2010) – Revision of the MOA/HR framework – evolving methodologies – Database on MOAs/key events/”codification” of Bradford Hill criteria • Training workshops (2006 to present at locations in Canada, the U. S. , Europe & the Asian region) – ILSI RSI/Health Canada/U. S. EPA/IPCS/University of Ottawa 10

Objectives – ARA Project Engagement/Evolution • Sharing and additionally evolving a broad range of “fit for purpose” risk assessment tools • Coordinating & Extending specific recommendations in the NAS Report on Science & Decisions: Advancing Risk Assessment – Considering a broad range of (internationally available) tools & their potential evolution to address critical areas identified in the report • Considering Dose Response tailored to Need – Appropriate consideration of Mode Of Action (MOA) and Value of Information – Evolving consideration of human variability & biologically based methodology for determining probability of response – Tiered, “Purpose Oriented” Assessment, in appropriate context – Through consideration of case studies 11

Roles/Responsibilities • The Alliance for Risk Assessment Steering Committee (ARA SC) – representatives from state, tribal, and federal government, academia, and environmental NGOs – selected members of the Expert Panel after a review of publically solicited nominations • Dose Response Advisory Committee (DRAC) – sponsors including state, federal, industry, and NGO representatives – Developed workshop structure & charge questions, presenters, consulting with ARA Steering Committee • Science Panel – input on the utility of the case study methods to address specific problem formulations, and identify areas for additional development 12

Process/Output - 3 Workshops March 2010 Pre workshop: Broad solicitation and brainstorming regarding illustrative case studies Initial vetting and review of proposals for case studies October 2010 • Review of case studies • Recommendation for draft methods framework for “fit for purpose” dose-response analysis, reflecting: • different conceptual models, data availability & risk management needs May 2011 • Additional case studies and identified issues : – Problem formulation, Mode of action. Endogenous & background exposures 13

Process/Output/Learnings Recommendations: • additional dissemination of dose-response analysis techniques for a wide range of problem formulations or decision contexts • Development of templates for transparency in selecting doseresponse approaches, relevant to use in specified risk management • Additional case studies on: – combined exposures, – value of information – in vitro to in vivo extrapolation – an entire purpose driven risk assessment, from problem formulation to conclusion 14

Process/Output (Cont’d) Ongoing: • manuscript in preparation • Framework to be “evergreen” with a Standing Panel to review case studies/issue papers Learnings: • Need to have assessors considering context to address appropriate focus & complexity (problem formulation for assessment) Evolving Framework & 24 case studies Engagement Model 15
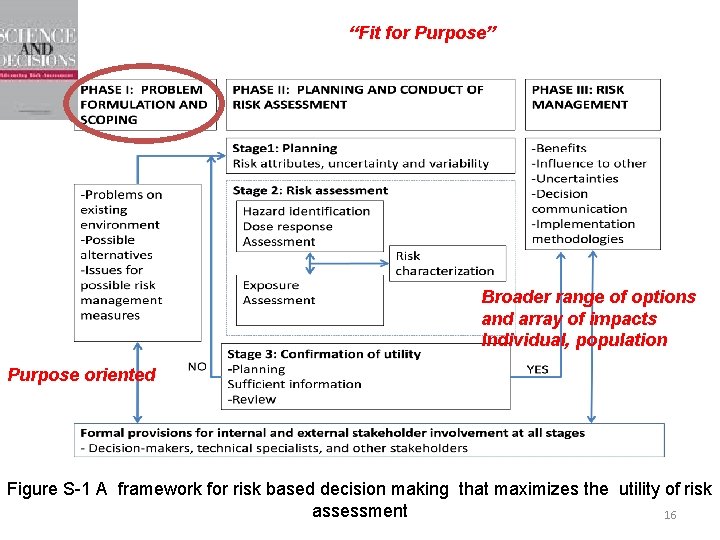
“Fit for Purpose” Broader range of options and array of impacts Individual, population Purpose oriented Figure S-1 A framework for risk based decision making that maximizes the utility of risk assessment 16

Organizational Framework 17
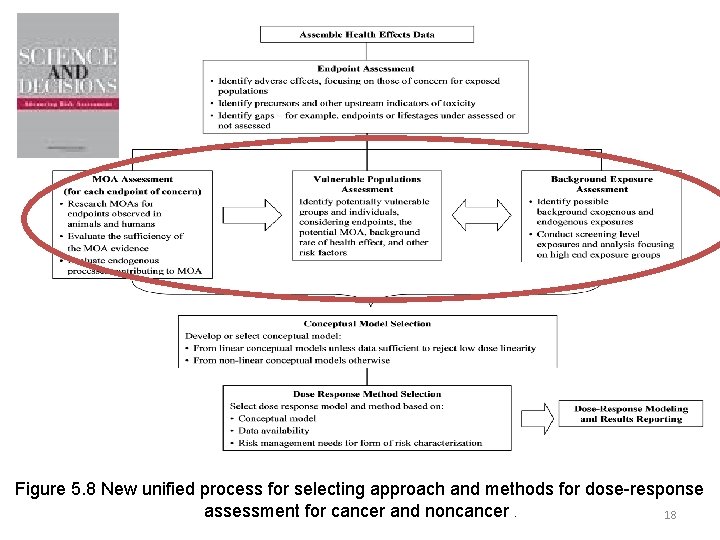
Figure 5. 8 New unified process for selecting approach and methods for dose-response assessment for cancer and noncancer. 18
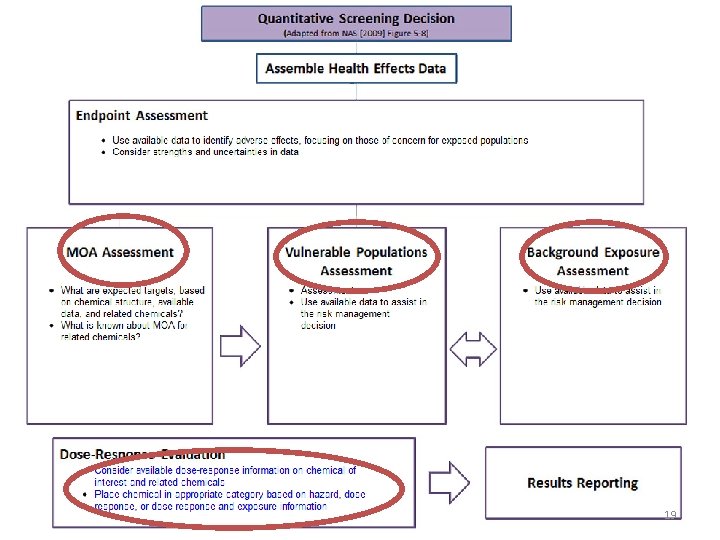
19

20
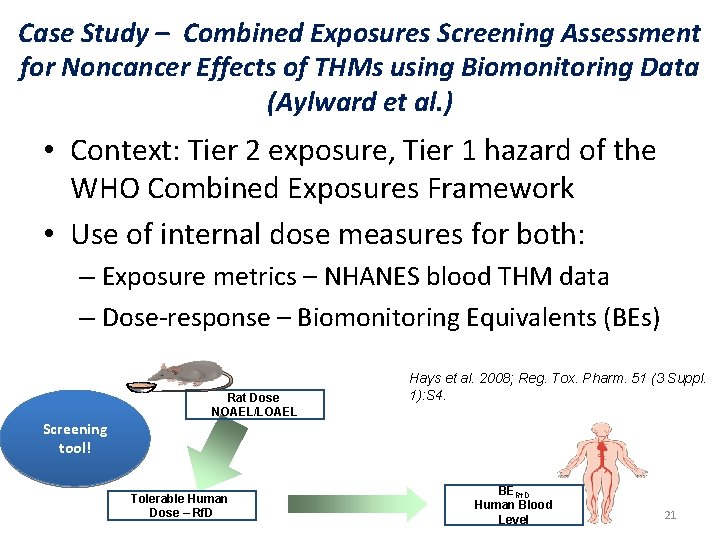
Case Study – Combined Exposures Screening Assessment for Noncancer Effects of THMs using Biomonitoring Data (Aylward et al. ) • Context: Tier 2 exposure, Tier 1 hazard of the WHO Combined Exposures Framework • Use of internal dose measures for both: – Exposure metrics – NHANES blood THM data – Dose-response – Biomonitoring Equivalents (BEs) Rat Dose NOAEL/LOAEL Hays et al. 2008; Reg. Tox. Pharm. 51 (3 Suppl. 1): S 4. Screening tool! Tolerable Human Dose – Rf. D BERf. D Human Blood Level 21
Problem Formulation for Grouping Nature of exposure? Is exposure likely? Co-exposure within a relevant timeframe? Rationale for considering compounds in an assessment group? Tiered Exposure Assessments Assessment Tiered Hazard Assessments Tier 0 Simple semiquantitative estimates of exposure Yes, no further action required Tier 0 Default dose addition for all components Tier 1 Generic exposure scenarios using conservative point estimates Is the margin of exposure adequate? Tier 2 More refined potency (RFP) and grouping based on MOA Tier 2 Refined exposure assessment, increased use of actual measured data Tier 3 Probabilistic exposure estimates Tier 1 Refined potency based on individual POD, refinement of POD No, continue with iterative refinement as needed (i. e. more complex exposure & hazard 22 models) Tier 3 PBPK or BBDR; probabilistic estimates of risk Increasing refinement of hazard Increasing refinement of exposure Uncertainty

Two Risk Assessment Approaches Investigated • Hazard quotient/Hazard index approach – Does not provide estimates of risk, just assessment of above/below Rf. D • Low dose risk extrapolation – Two approaches 23

Application of a Source-to-Outcome Model to Quantitatively Assess Variability in Dose and Sensitivity in Humans (Chlorpyrifos; Price et al. ) • Tier 3 analysis (probabilistic exposure estimates, PBPK & reliance on MOA-related precursor) – reserved for cases where there is a small margin between exposure and effect; combined effects • Relevant to substances that act by a similar mode of action (i. e. , ACh. E inhibition) • Addresses more generic issues raised by the NAS committee 24
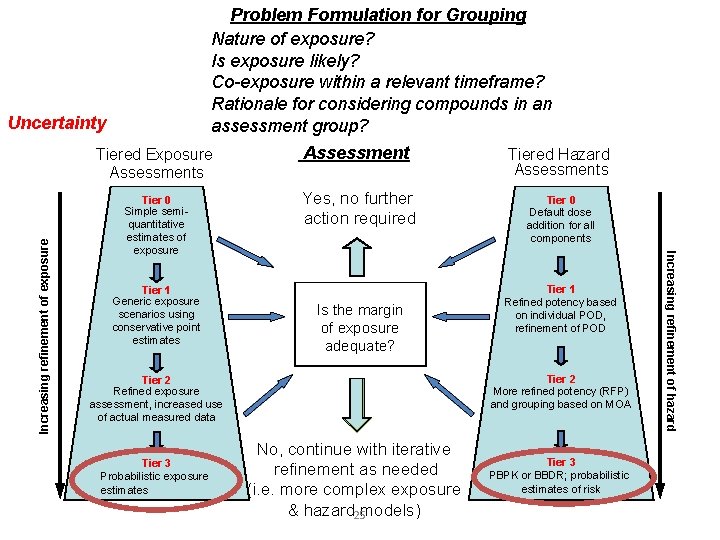
Problem Formulation for Grouping Nature of exposure? Is exposure likely? Co-exposure within a relevant timeframe? Rationale for considering compounds in an assessment group? Tiered Exposure Assessments Assessment Tiered Hazard Assessments Tier 0 Simple semiquantitative estimates of exposure Yes, no further action required Tier 0 Default dose addition for all components Tier 1 Generic exposure scenarios using conservative point estimates Is the margin of exposure adequate? Tier 2 More refined potency (RFP) and grouping based on MOA Tier 2 Refined exposure assessment, increased use of actual measured data Tier 3 Probabilistic exposure estimates Tier 1 Refined potency based on individual POD, refinement of POD No, continue with iterative refinement as needed (i. e. more complex exposure & hazard 25 models) Tier 3 PBPK or BBDR; probabilistic estimates of risk Increasing refinement of hazard Increasing refinement of exposure Uncertainty
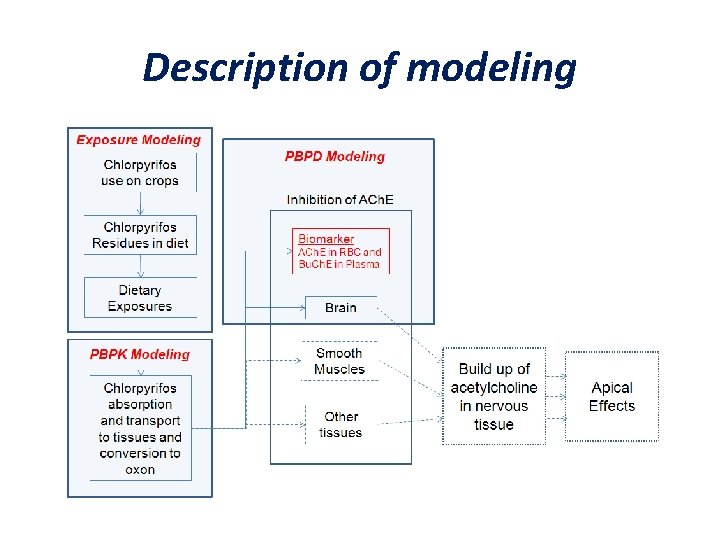
Description of modeling

Relevance to Advancements in Risk Assessment MOA Based: • Assessed variability in both – exposure (variation of residue levels across foods and variation in individual’s dietary consumptions) and – response (variation in physiology and metabolism) • Evaluated response to the range of actual human exposures • Assessed human sensitivity in multiple age groups (infants, children, adults) • Modeling was made more predictive by focusing on early “key event” - namely cholinesterase inhibition (Ch. EI)

Forward Looking Assessment • Public problem formulation with proposal for “fit for purpose” assessment – Assimilated Overview of Data – Proposed Focus – Efficiency – Proposed Process • Tiered assessment options drawing on predictive tools in early tiers – Importance of mechanistic underpinning • What’s the engagement strategy? 28

Collaborators 29

ARA Steering Committee • Barbara Harper, Confederated Tribes of the Umatilla Indian Reservation • William Hayes, State of Indiana • Bette Meek, University of Ottawa • Anita Meyer, United States Army Corps of Engineers • Edward Ohanian, U. S. Federal Government • Ruthann Rudel, Silent Spring • Phil Wexler, National Library of Medicine -----recused---- • Michael Dourson, Toxicology Excellence for Risk Assessment • Michael Honeycutt, Texas Commission on Environmental Quality 30

Dose-Response Advisory Committee • • Rick Becker, ACC Michael Dourson, TERA Julie Fitzpatrick, EPA Roberta Grant, TCEQ Lynne Haber, TERA Michael Honeycutt, TCEQ Lynn H. Pottenger, Dow Chemical Jennifer Seed, EPA 31

Expert Panel • • • Michael Bolger, U. S. FDA James S. Bus, Dow Chemical John Christopher, CH 2 M/Hill Rory Conolly, U. S. EPA Michael Dourson, TERA *Adam M. Finkel, UMDNJ William Hayes, Indiana DEM (Workshop II only) R. Jeffrey Lewis, Exxon. Mobil Biomedical Randy Manning, Georgia DNR • • Bette Meek, U of Ottawa (Chairperson) Paul Moyer, Minnesota DH (Workshop II only) *Greg Paoli, Risk Sciences International Rita Schoeny, U. S. EPA (Workshop III only) • *On NAS Science and Decisions panel 32

More Information? ARA Dose Response Framework – (working beta) http: //www. allianceforrisk. org/workshop/framework/ problemformulation. html Evolution of the ILSI/IPCS Frameworks – Mode of Action • Meek & Klaunig (2010) Chemico-Biological Interactions 184: 279 – 285 • Carmichael et al. (2011) Crit Rev Toxicol. 2011 Mar; 41(3): 175 -86 Guidance for CSAF • http: //www. who. int/ipcs/methods/harmonization/areas/un certainty/en/index. html Combined Exposures • Meek et al. (2011) Reg Tox Pharm 60: S 1 -S 14 33
- Slides: 33