Probing Solvation Effects in IBr CO 2n by

Probing Solvation Effects in IBr (CO 2)n by Photoelectron Spectroscopy Elisa Miller Leonid Sheps, Ryan Calvi, and W. Carl Lineberger JILA, Department of Chemistry University of Colorado AFOSR NSF 62 nd OSU Spectroscopy Symposium June 18, 2007 -
Introduction § Equilibrium structure State energies and geometry - § IBr (CO 2)n, n >1 § Dynamics n=8 Photodissociation and recombination § IBr (investigated by A. Sanov and T. Sanford) - n = 12 § IBr (CO 2)n, n >1 (investigated by V. Dribinski and T. Sanford)
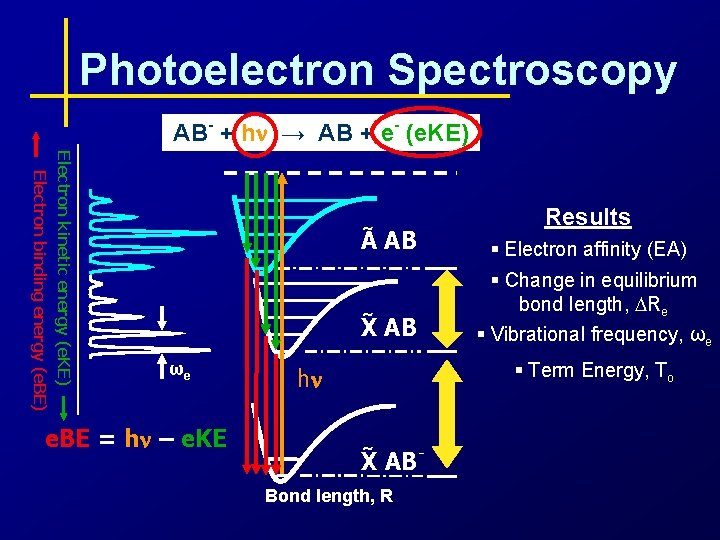
Photoelectron Spectroscopy AB- + hn → AB + e- (e. KE) Electron kinetic energy (e. KE) Electron binding energy (e. BE) ωe e. BE = hn – e. KE Results ~ A AB § Electron affinity (EA) ~ X AB § Change in equilibrium bond length, DRe § Vibrational frequency, ωe § Term Energy, To hn ~ X ABBond length, R

Experiment Source Chamber E/E 0. 020 Pulsed Valve IBr in CO 2 Time of Flight Ion Optics CCD camera Electron Imaging Mass Gate e- Acceleration Stack Nd: YAG/ OPO/ doubler Electron Gun Velocity Map Imaging

Velocity Map Imaging (VMI) - IBr (300 nm) photodetatchment v 1 v 2 2 -D raw image 2 -D slice through reconstructed 3 -D image Photoelectron spectrum

Energy Velocity Map Imaging (VMI) X 1 S+ ground state X 1 S+ X 2 S+1/2 I-Br. Bond. Distance Length v 1 v 2 2 -D raw image 2 -D slice through reconstructed 3 -D image Photoelectron spectrum

Velocity Map Imaging (VMI) Energy 3 P 2 excited state X 1 S+ ground state X 2 S+1/2 I-Br. Bond. Distance Length v 1 v 2 2 -D raw image 2 -D slice through reconstructed 3 -D image Photoelectron spectrum

Velocity Map Imaging (VMI) 3 P 3 P 1 excited state 1 Energy 3 P 2 excited state X 1 S+ ground state X 2 S+1/2 I-Br Bond Length v 1 v 2 2 -D raw image 2 -D slice through reconstructed 3 -D image Photoelectron spectrum

- Photoelectron Counts IBr (300 nm) Photoelectron Spectrum 3 P 1 ~4. 07 e. V 3 P 2 ~3. 96 e. V X 1 S+ ~3. 3 e. V Electron Binding Energy (e. V) Re = 0. 167 Ǻ 3 P 1 v=0 Re = 0. 183 Ǻ 3 P 2 v=0 Electron Binding Energy (e. V)

- IBr Bond Length IBr (3 P 1) Re = 2. 8583 Å Energy Re = 2. 842 Å IBr (3 P 2) IBr (1 S+) IBr- (2 S+1/2) I-Br Bond Length Re = 2. 469 Å Re = 3. 025 (. 01) Å s* p* p s

IBr Electron Affinity hn e. KE Energy IBr (3 P 1) EA (IBr) = hn - To- e. KE (v’ = 0) IBr (3 P 2) IBr (1 S+ ) To 1. 41 e. V To 1. 525 e. V Electron Affinity (EA) IBr- (2 S+1/2) I-Br Bond Length 2. 51 (. 01) e. V

- IBr Dissociation Energy - Do(IBr ) = EA(IBr) + Do(IBr) - EA(Br) I + Br Energy IBr (1 S+) Do(IBr) 1. 82 e. V I + Br EA(Br) 3. 36 e. V - EA(IBr) 2. 51(. 01) e. V Do(IBr ) IBr- (2 S+ 1/2) I-Br bond length 0. 97 (. 01) e. V

- - IBr (CO IBr Summary 2) Photoelectron Spectrum Results - IBr (CO 2) § Bond length (R ) IBr- = 3. 025(. 01) Å e § Electron Affinity (EA) IBr = 2. 51(1) e. V 3 P 1 e. BE = 0. 16 e. V §Dissociation Energy, Do (I - Br-) = 0. 97(. 01) e. V 1 S+ 3 P e. BE = 0. 16 e. V 2 e. BE = 0. 15 e. V EA (IBr) IBr -

- IBr (CO 2) Photoelectron Spectrum - EA(IBr(CO 2)) = EA(IBr) + Do(IBr (CO 2)) – Do(IBr(CO 2)) IBr(CO 2) IBr + CO 2 Known Properties: Energy EA(IBr) = 2. 51 (. 01) e. V Do(IBr-(CO 2)) ~ 0. 23 e. V EA(IBr(CO 2)) EA(IBr) Do(IBr(CO 2) ~ 0. 07 e. V EA(IBr(CO 2)) = 2. 67 (. 05) e. V IBr- + CO 2 IBr-(CO 2) Do(IBr-(CO 2))

Conclusions and Future Directions - § Re (IBr ) = 3. 025 (. 01) Ǻ EA (IBr) = 2. 51 (1) e. V Do (IBr ) = 0. 97 (. 01) e. V § EA (IBr(CO 2)) = 2. 67 (. 05) e. V § Investigate the effects of additional CO 2 solvation on IBr - § Pump-Probe photoelectron studies of IBr (CO 2)n § Probing potential energy surfaces and crossings during dissociation/recombination
- Slides: 15