Probing Excited States by Photoelectron Imaging Dyson Orbitals

Probing Excited States by Photoelectron Imaging: Dyson Orbitals within Equation-of-Motion Coupled -Cluster Formalism Anna I. Krylov University of Southern California, Los Angeles Ohio Spectroscopy Meeting, 2007

Acknowledgements: Dr. Melania Oana Inspiration from experiments of: Albert Stolow, Hanna Reisler, Klaus Muller-Dethlefs Albert Stolow, Hanna Reisler, USC Steacie Institute for Molecular Sciences
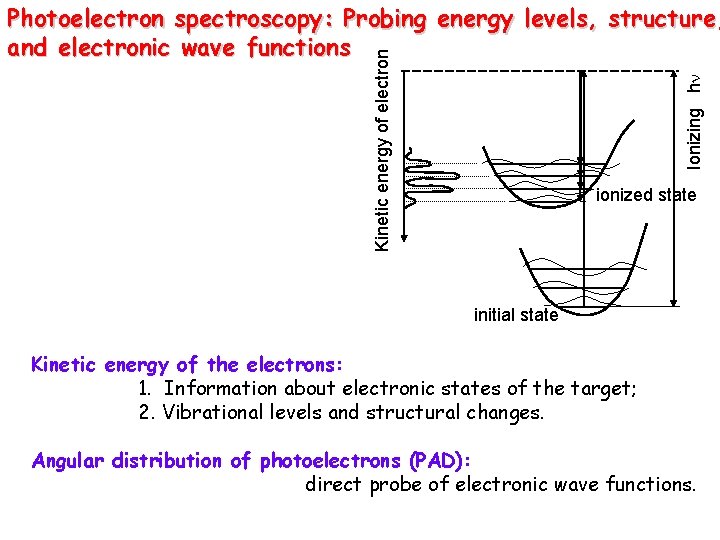
Ionizing hn Kinetic energy of electron Photoelectron spectroscopy: Probing energy levels, structure, and electronic wave functions ionized state initial state Kinetic energy of the electrons: 1. Information about electronic states of the target; 2. Vibrational levels and structural changes. Angular distribution of photoelectrons (PAD): direct probe of electronic wave functions.
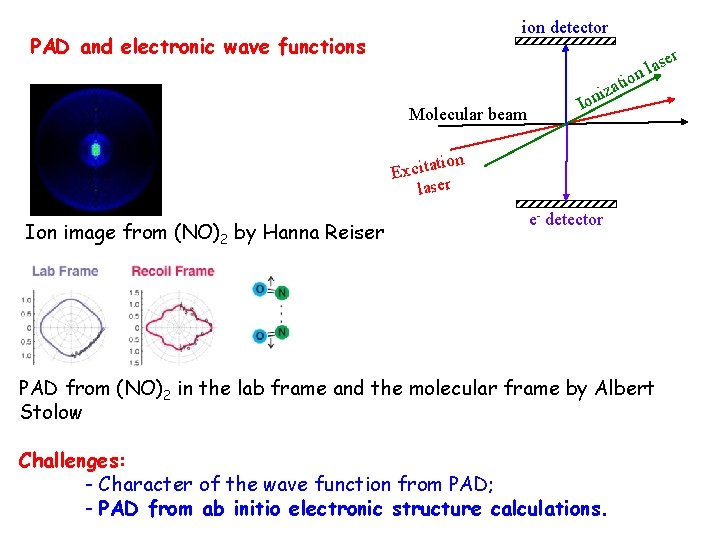
ion detector PAD and electronic wave functions er s a l n tio Molecular beam za oni I on i t a t i c Ex laser Ion image from (NO)2 by Hanna Reiser e- detector PAD from (NO)2 in the lab frame and the molecular frame by Albert Stolow Challenges: - Character of the wave function from PAD; - PAD from ab initio electronic structure calculations.

Outline: 1. From wave-function to PAD: Dyson orbitals. 2. Dyson orbitals for the ionization from the ground and excited states of formaldehyde: numerical examples. 3. From Dyson orbitals to PADs: Selection rules and averaging. 4. Dyson orbitals for NO dimer ionization. 5. Conclusions.

Photoelectron wave functions and PADs The probability of finding an electron in d. V at {r, θ, φ}: Ψel can d. V be expanded in the basis of spherical waves: Radial part: Rkl ~ Bessel functions, Jl+1/2: , with Angular part: Ylm – spherical harmonics

PAD and Dyson Orbitals |Cklm|2 - probability to find an ejected electron in the {klm} state. They are given by the ionization dipole moment matrix elements between the initial ( ΨN) and the final (ΨN-1 x Ψel) states: where ri - spatial coordinates of the ith electron Using permutational symmetry of the wave functions and integrating over N-1 coordinates: where fd(r) is Dyson orbital:

Dyson Orbitals: Summary 1. The “difference” or “overlap” between the N and N-1 e – wave functions of the neutral and the cation. 2. Can be used to calculate probability to find the ionized electron in a particular state. 3. Norm of the Dyson orbital ~ probability of the ionization event. 4. Can be interpreted as an initial state of the ionized electron, e. g. , for a one-electron system Dyson orbital is just the wave function. 5. For Hartree-Fock wave functions and within the Koopmans approximation: Φd = φ l k k j i M M+

Dyson Orbitals in EOM-CC Formalism EOM-IP/EE-CCSD: ΨM+/M* = (R 1 + R 2) F 0 REE R 1Ψref M* R 2Ψref RIP Ψref M R 1Ψref R 2Ψref M+ Coefficients of Dyson orbitals in MO basis - analogous to transition density matrix element:
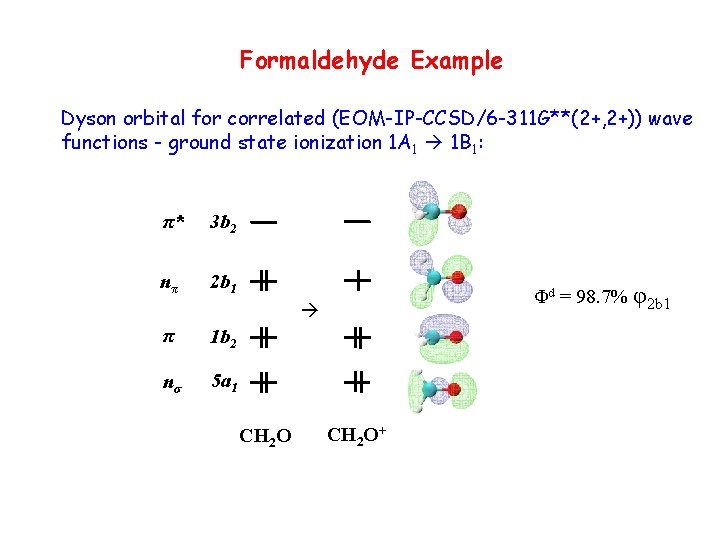
Formaldehyde Example Dyson orbital for correlated (EOM-IP-CCSD/6 -311 G**(2+, 2+)) wave functions - ground state ionization 1 A 1 1 B 1: π* 3 b 2 nπ 2 b 1 Φd = 98. 7% φ2 b 1 π 1 b 2 nσ 5 a 1 CH 2 O+
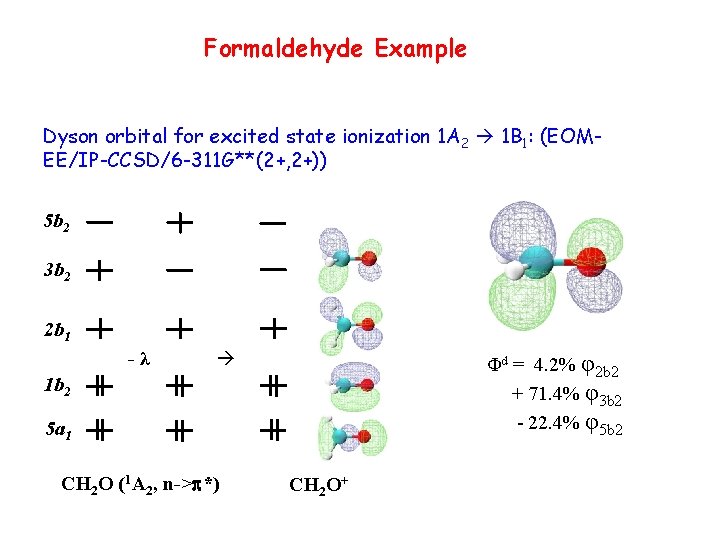
Formaldehyde Example Dyson orbital for excited state ionization 1 A 2 1 B 1: (EOMEE/IP-CCSD/6 -311 G**(2+, 2+)) 5 b 2 3 b 2 2 b 1 -λ Φd = 4. 2% φ2 b 2 + 71. 4% φ3 b 2 - 22. 4% φ5 b 2 1 b 2 5 a 1 CH 2 O (1 A 2, n->p*) CH 2 O+
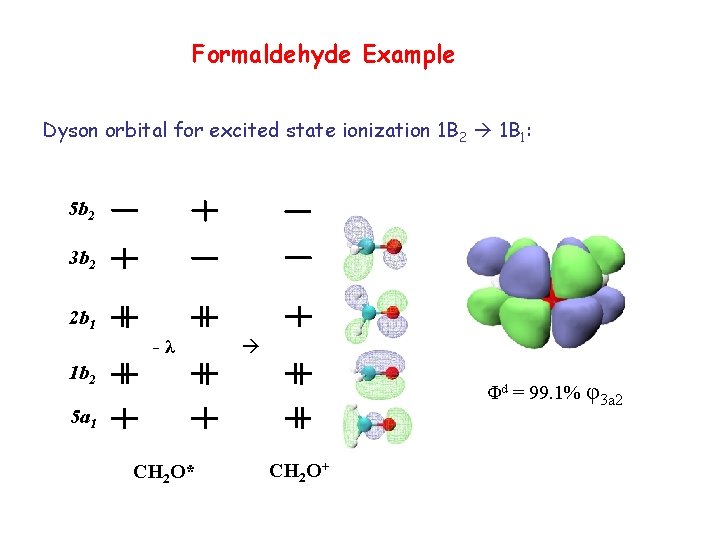
Formaldehyde Example Dyson orbital for excited state ionization 1 B 2 1 B 1: 5 b 2 3 b 2 2 b 1 -λ 1 b 2 Φd = 99. 1% φ3 a 2 5 a 1 CH 2 O* CH 2 O+
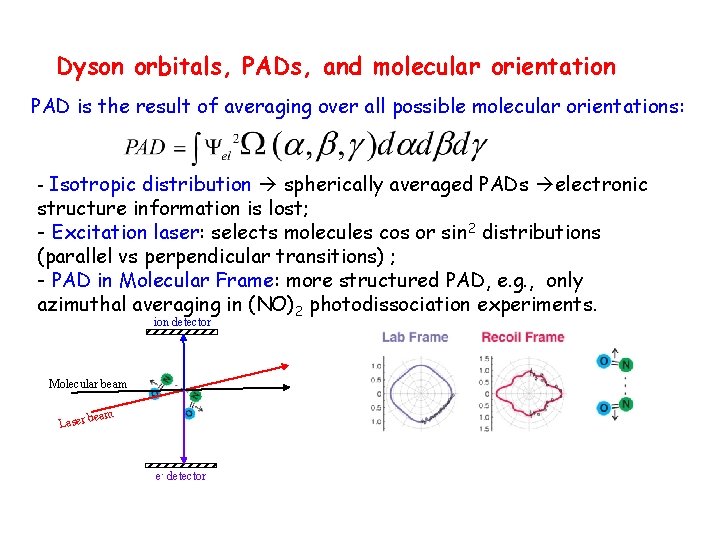
Dyson orbitals, PADs, and molecular orientation PAD is the result of averaging over all possible molecular orientations: - Isotropic distribution spherically averaged PADs electronic structure information is lost; - Excitation laser: selects molecules cos or sin 2 distributions (parallel vs perpendicular transitions) ; - PAD in Molecular Frame: more structured PAD, e. g. , only azimuthal averaging in (NO)2 photodissociation experiments. ion detector Molecular beam Laser e- detector

Electron Angular Momentum States: Selection Rules Dyson orbital r (x, y, z) Φd(r)·r Rkl. Ylm s (l = 0) l = 1 0 px (l = 1) ~ x x 0 x y z x 0 x 2 xy xz x l = 0, 2 Allowed electron angular momentum states: Δl = ± 1 Molecular Dyson orbital: more angular momentum states
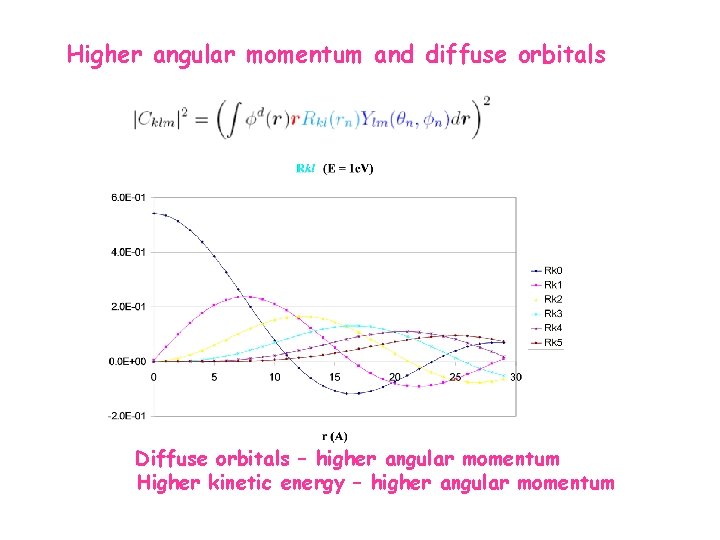
Higher angular momentum and diffuse orbitals Diffuse orbitals – higher angular momentum Higher kinetic energy – higher angular momentum
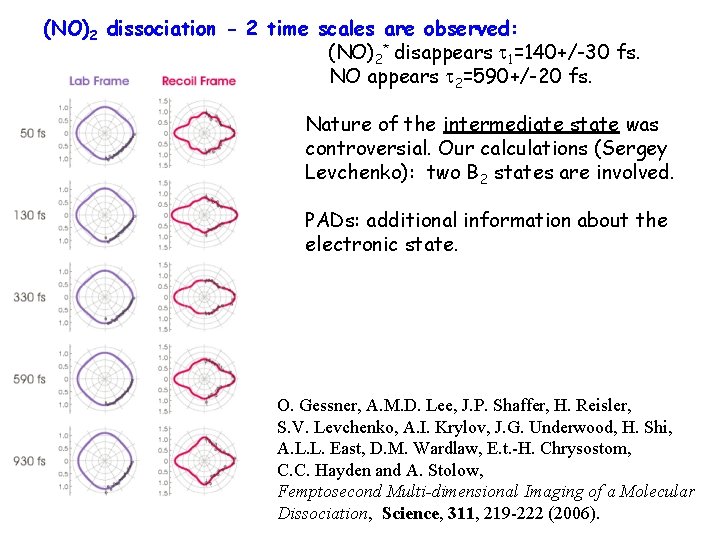
(NO)2 dissociation - 2 time scales are observed: (NO)2* disappears t 1=140+/-30 fs. NO appears t 2=590+/-20 fs. Nature of the intermediate state was controversial. Our calculations (Sergey Levchenko): two B 2 states are involved. PADs: additional information about the electronic state. O. Gessner, A. M. D. Lee, J. P. Shaffer, H. Reisler, S. V. Levchenko, A. I. Krylov, J. G. Underwood, H. Shi, A. L. L. East, D. M. Wardlaw, E. t. -H. Chrysostom, C. C. Hayden and A. Stolow, Femptosecond Multi-dimensional Imaging of a Molecular Dissociation, Science, 311, 219 -222 (2006).
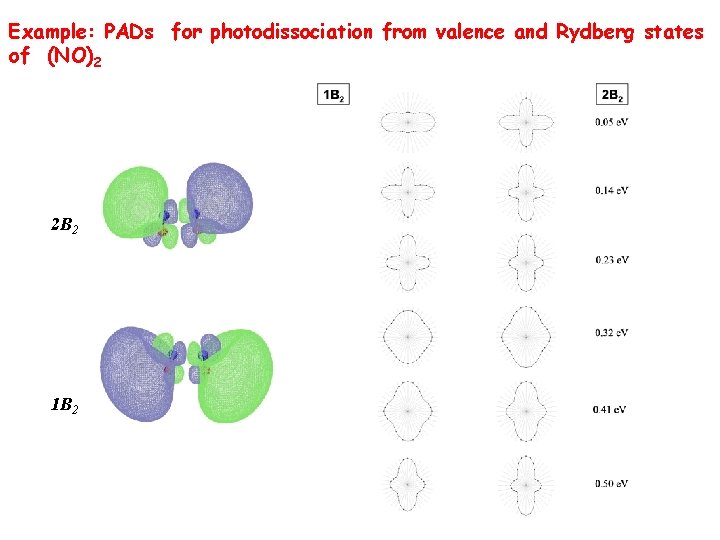
Example: PADs for photodissociation from valence and Rydberg states of (NO)2 2 B 2 1 B 2

Example: PADs for photodissociation from valence and Rydberg states of (NO)2

Conclusions 1. Dyson orbitals for the ground and excited state ionization are implemented within EOM-IP/EA/EE/SF-CCSD. 2. Dyson orbitals for one-electron ionizations – obey Koopmans like rules. 3. Quantitative analysis of Dyson orbitals: l, m angular momentum states accessible to the photoionized electron and the corresponding probabilities |Cklm|2. 4. PAD modeling: - within RPA: |Cklm|2 ↔ PAD; - beyond RPA: need the interference contributions - cross terms C*klm. Ckl’m. 5. Qualitative trends in molecular PADs: diffuse states – higher angular momentum; higher kinetic energy – higher angular momentum. 6. NO dimer: observed PADs are inconsistent with the A 1 state. More detailed comparison needs to take into account kinetic energy distribution and the phases.

THANKS: My group; Ab initio packages: Our codes: available in Q-CHEM & SPARTAN Additional calculations: ACES II, GAMESS Funding: 1. Center for Computational Studies of Open-Shell and Electronically Excited Species (NSF): http: //iopenshell. usc. edu bridging the gap between ab initio theory and experiment. 2. Department of Energy. 3. National Science Foundation. 4. WISE Research Fund (USC). 5. NIH-SBIR (w/Q-Chem).
- Slides: 20