Probing Buried Electrochemical Interfaces with Multiplex Broadband Sum

Probing Buried Electrochemical Interfaces with Multiplex Broadband Sum Frequency Generation Spectroscopy Alexei Lagutchev, Guo-Qiang Lu, Tomohiro Takeshita, Rachel Behrens, Andrzej Wieckowski and Dana Dlott School of Chemical Sciences, University of Illinois at Urbana-Champaign
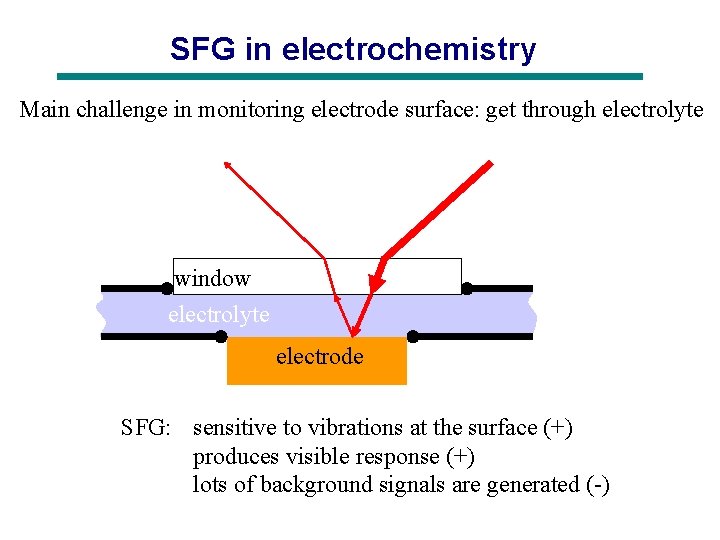
SFG in electrochemistry Main challenge in monitoring electrode surface: get through electrolyte window electrolyte electrode SFG: sensitive to vibrations at the surface (+) produces visible response (+) lots of background signals are generated (-)
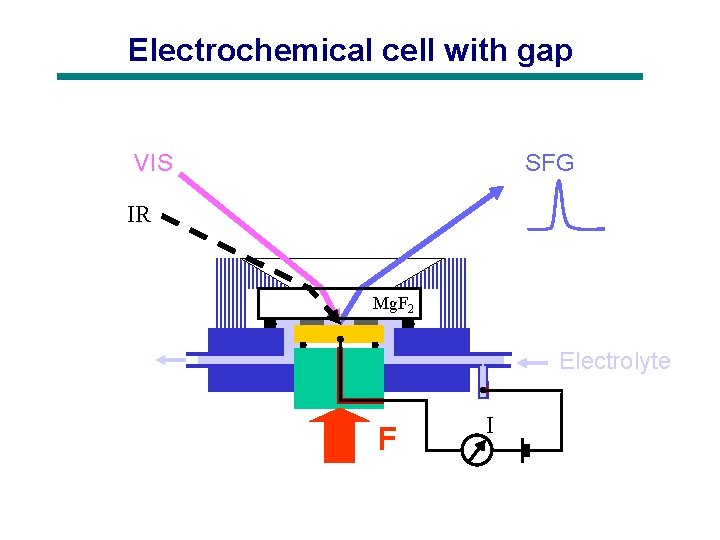
Electrochemical cell with gap VIS SFG IR Mg. F 2 Electrolyte F I
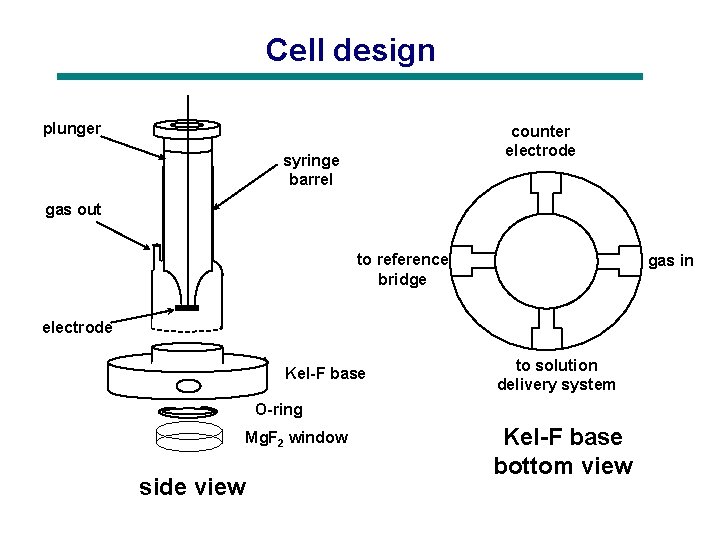
Cell design plunger counter electrode syringe barrel gas out to reference bridge gas in electrode Kel-F base to solution delivery system O-ring Mg. F 2 window side view Kel-F base bottom view

CO on Pt: SFG spectrum
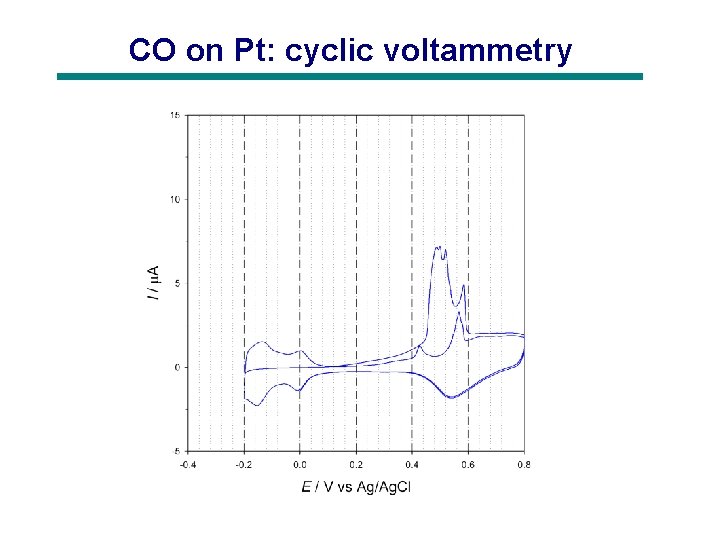
CO on Pt: cyclic voltammetry
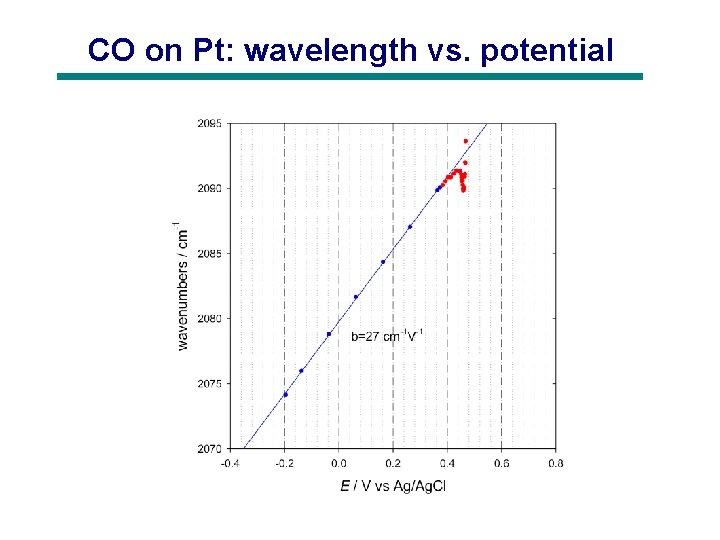
CO on Pt: wavelength vs. potential
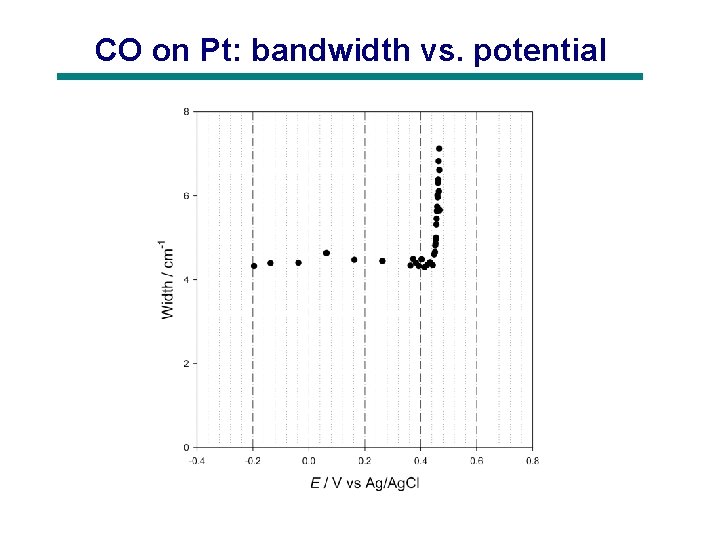
CO on Pt: bandwidth vs. potential

SFG vs. cyclic voltammetry
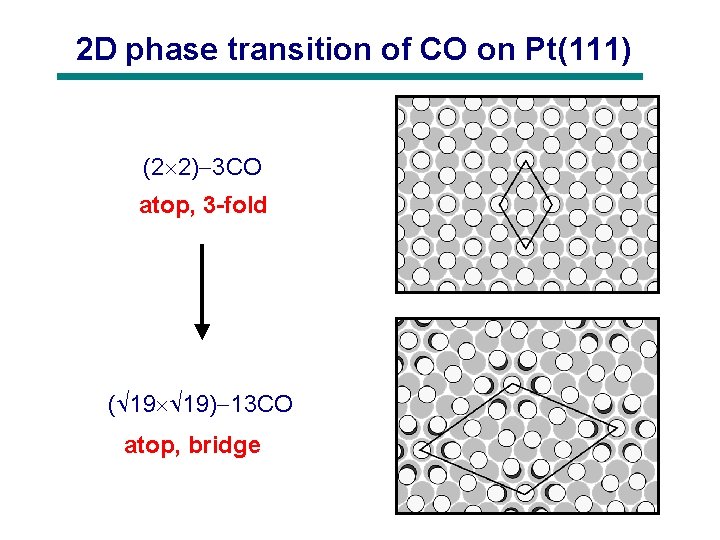
2 D phase transition of CO on Pt(111) (2 2) 3 CO atop, 3 -fold ( 19 19) 13 CO atop, bridge
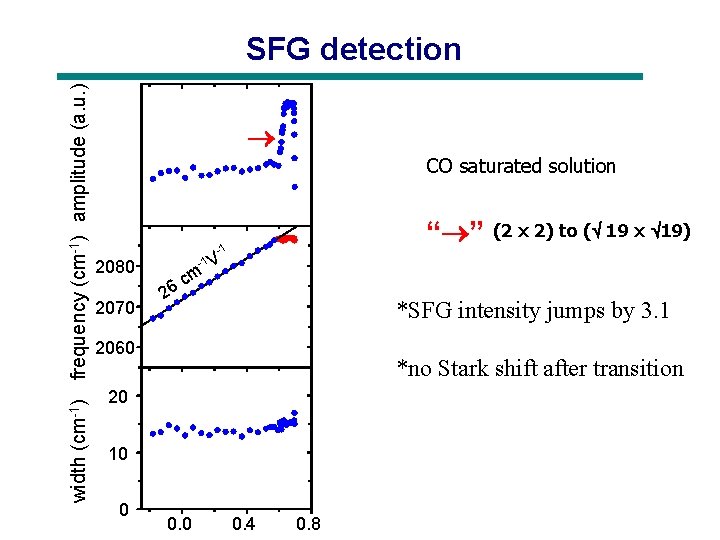
width (cm-1) frequency (cm-1) amplitude (a. u. ) SFG detection CO saturated solution 2080 2070 c 26 “ ” -1 -1 V m *SFG intensity jumps by 3. 1 2060 *no Stark shift after transition 20 10 0 (2 x 2) to ( 19 x 19) 0. 0 0. 4 0. 8
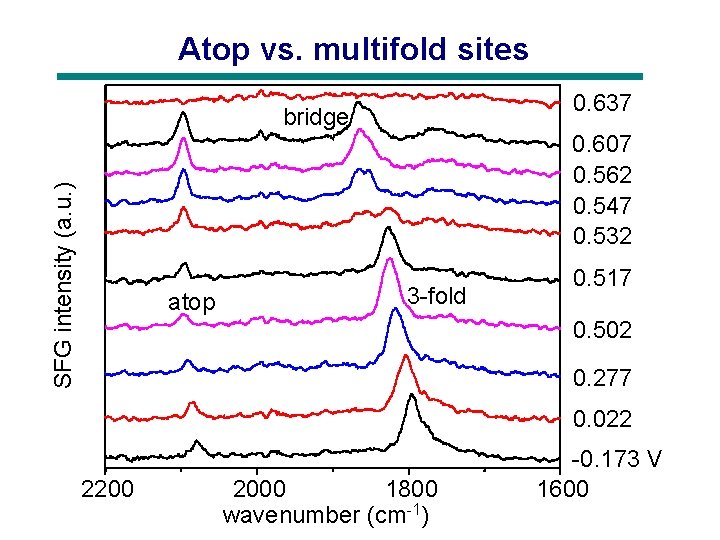
Atop vs. multifold sites 0. 637 SFG intensity (a. u. ) bridge atop 0. 607 0. 562 0. 547 0. 532 3 -fold 0. 517 0. 502 0. 277 0. 022 2200 2000 1800 wavenumber (cm-1) -0. 173 V 1600

SFG intensity and surface coverage Normally one expects Isfg~N 2 Experiment: From structure: When corrected for dipole-dipole interaction and changes in electron back-donation from Co to Pt
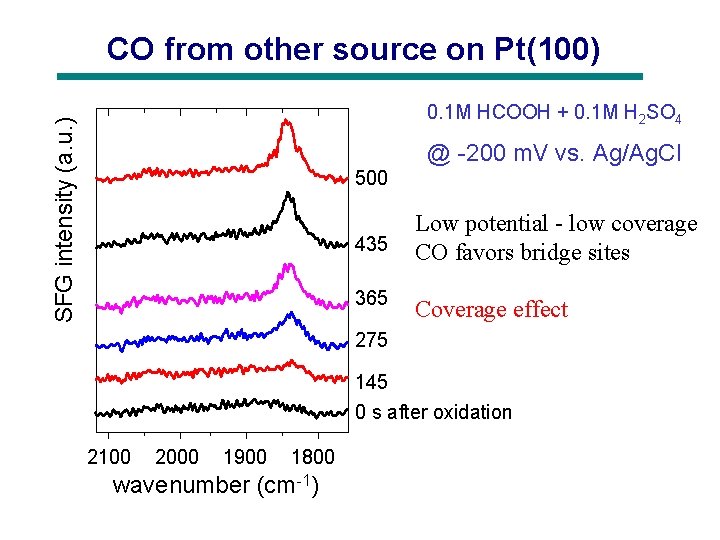
CO from other source on Pt(100) SFG intensity (a. u. ) 0. 1 M HCOOH + 0. 1 M H 2 SO 4 @ -200 m. V vs. Ag/Ag. Cl 500 435 365 Low potential - low coverage CO favors bridge sites Coverage effect 275 145 0 s after oxidation 2100 2000 1900 1800 wavenumber (cm-1)
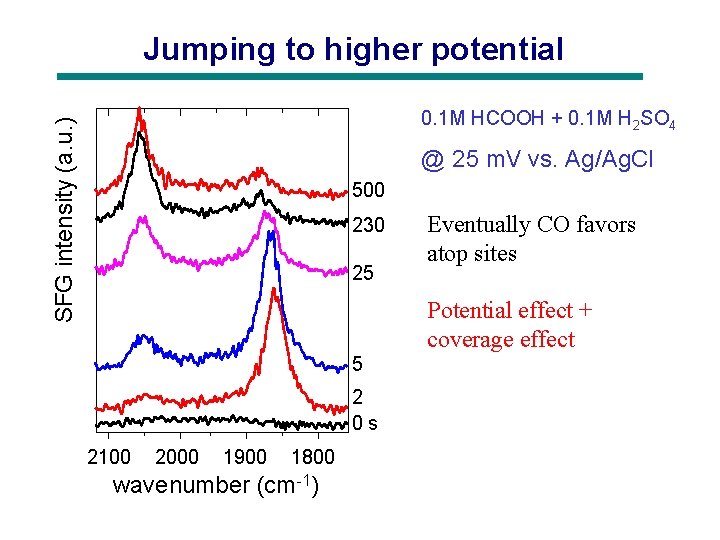
Jumping to higher potential SFG intensity (a. u. ) 0. 1 M HCOOH + 0. 1 M H 2 SO 4 @ 25 m. V vs. Ag/Ag. Cl 500 230 25 Eventually CO favors atop sites Potential effect + coverage effect 5 2 0 s 2100 2000 1900 1800 wavenumber (cm-1)
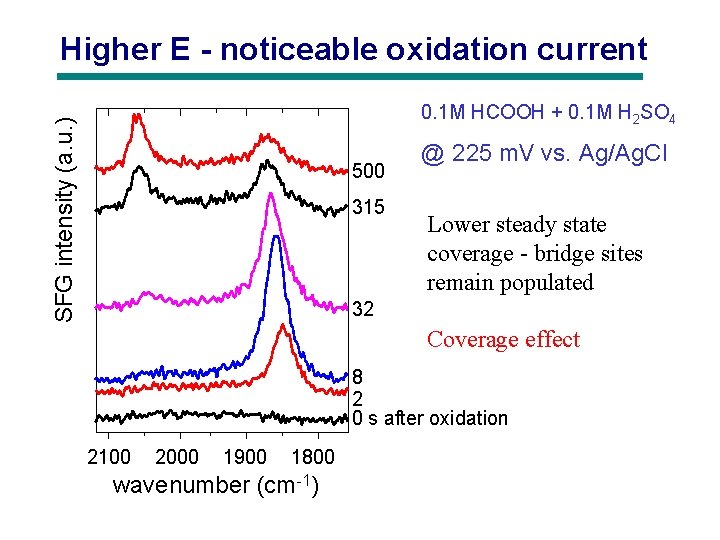
Higher E - noticeable oxidation current SFG intensity (a. u. ) 0. 1 M HCOOH + 0. 1 M H 2 SO 4 500 315 @ 225 m. V vs. Ag/Ag. Cl Lower steady state coverage - bridge sites remain populated 32 Coverage effect 8 2 0 s after oxidation 2100 2000 1900 1800 wavenumber (cm-1)
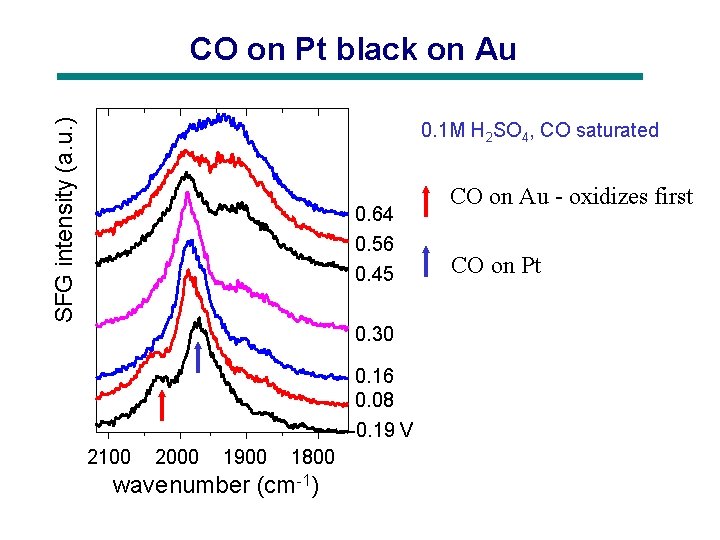
SFG intensity (a. u. ) CO on Pt black on Au 0. 1 M H 2 SO 4, CO saturated 0. 64 0. 56 0. 45 0. 30 0. 16 0. 08 -0. 19 V 2100 2000 1900 1800 wavenumber (cm-1) CO on Au - oxidizes first CO on Pt
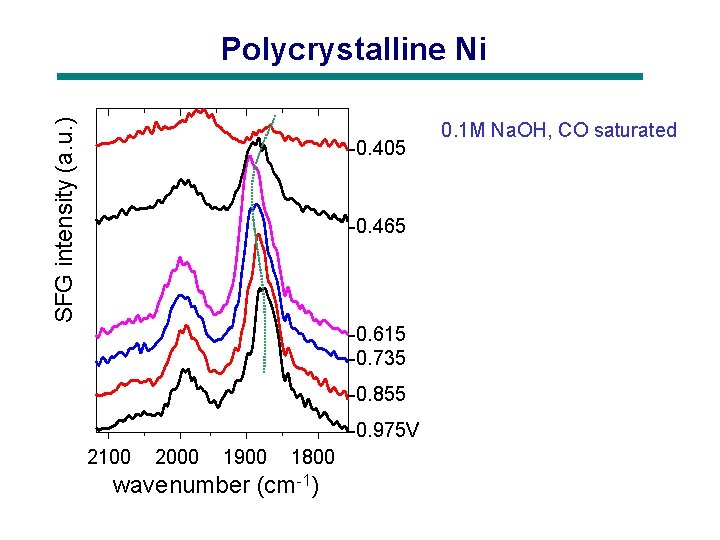
SFG intensity (a. u. ) Polycrystalline Ni -0. 405 -0. 465 -0. 615 -0. 735 -0. 855 -0. 975 V 2100 2000 1900 1800 wavenumber (cm-1) 0. 1 M Na. OH, CO saturated

Background vs. delay

Less obvious background effect

Time domain picture

Conclusions and future work • SFG is uniquely suitable to study electrochemical interfaces vibrational spectroscopy least restrictive for kinetics • Accurate quantitative analysis of electrode coverage is possible provided local field corrections are accounted for • Time domain manipulations can be successfully used to suppress background signals • CO on noble metals (Pt, Pd, Au) may be used as ultrathin detection layer in pump-probe experiments - fast shocking, heating. • CO adsorption on nanoparticles can be studied - direct application to electrocatalysis
- Slides: 22