Principles of spectroscopy Basics of atom Atom Atom

Principles of spectroscopy
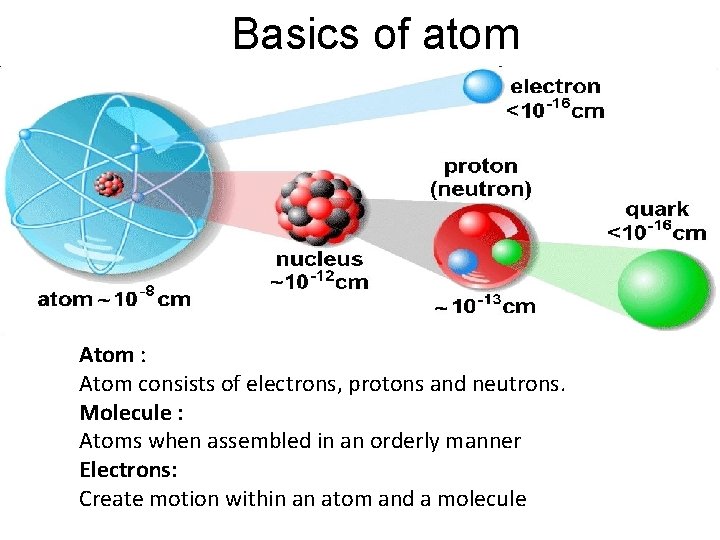
Basics of atom Atom : Atom consists of electrons, protons and neutrons. Molecule : Atoms when assembled in an orderly manner Electrons: Create motion within an atom and a molecule

Electrons • No. of electrons equals No. of protons in the nucleus • Very much smaller than are protons and neutrons • Add very little to the mass (1 g contain 10 x 1026 electrons) • Atom's properties based on the electrons. • Situated at large distances around the nucleus • Have a negative electric charge of 1. 6 x 10 -19 C • Electron mass is 0. 000549 amu (1 amu - 1. 6604 x 1027 kg)

Atomic Orbitals • • • Electrons are in constant motion Occupy a "volume" as they move Electrical attraction prevents them from flying off Electrons repel by one another. “Atomic orbitals“ - a cloud-like region in which there is a 90% probability of finding the electron in motion around the nucleus.

Energy shells • • • Electrons move around the nucleus in shells Shells are named K, L, M, N Each shell contain a maximum number of electrons. Number is determined by the formula 2 n 2 Shells are further divided into sub-shells, s, p, d, f… Orbital closest to the nucleus is filled first This orbital has lowest energy level This is spherical, so called "1 s" orbital Next higher energy level - four orbitals - 8 electrons. The "2 s” orbital is spherical Other 3 second-level orbitals are "dumbbell“, so termed "2 p" orbitals
Energy shells
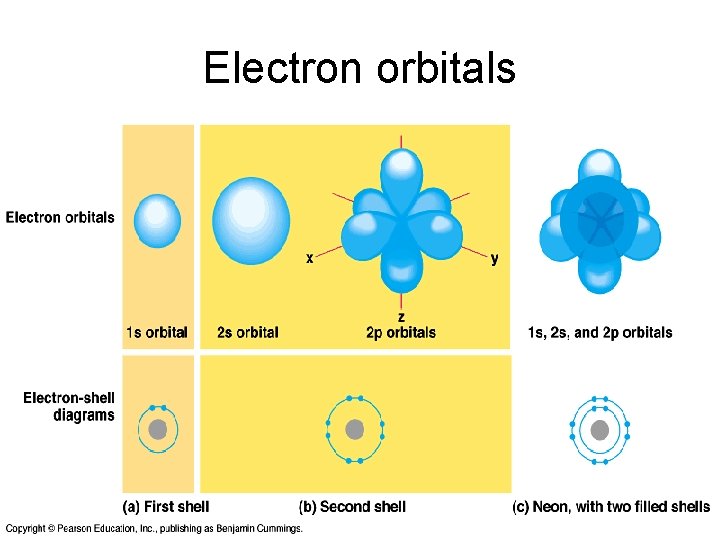
Electron orbitals

Light Quanta or Photons : Packets of energy in light, heat and EMR Propagate through space as electromagnetic waves Light : A collection of waves whose wavelengths are in a narrow range of between 4000 Ao to 8000 Ao [Ao = 10 -8 cm] (400 -800 nm) EMR : Radiation whose wavelengths of the order of meters (radiowaves) to less than 1 Ao (gamma rays).
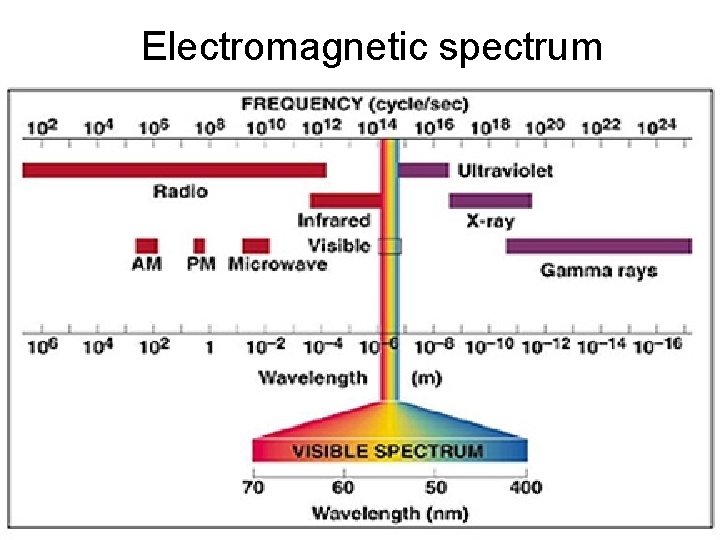
Electromagnetic spectrum

Electron energy level o When all the electrons are in an unexcited, or ground state, the atom is at its lowest energy level. o When the atom absorbs energy, electrons can be excited and move into higher-energy shells. o As electrons move from one shell to another, unique quanta of energy are absorbed or emitted. o Lights unique energy quanta dependent on the electronic structure of the atom.
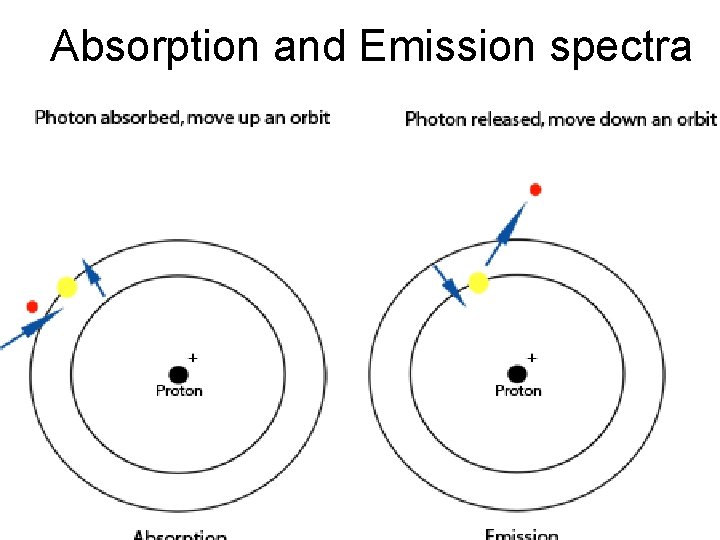
Absorption and Emission spectra
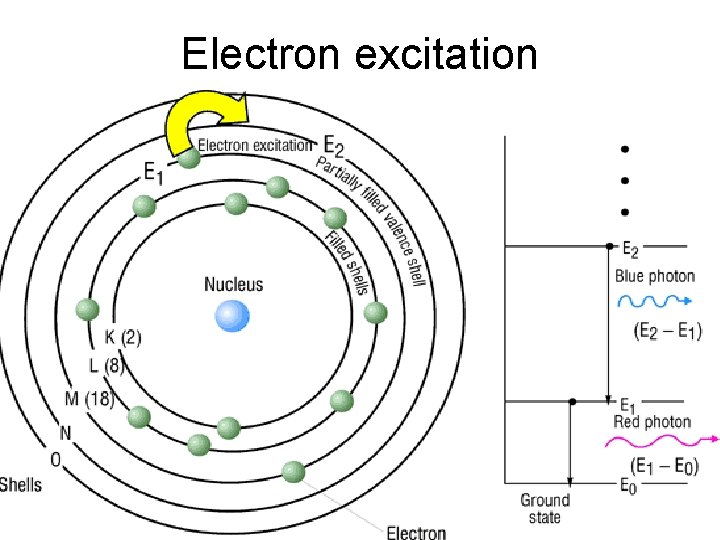
Electron excitation

Principles of Spectrophotometry v Beer's law v Measurement of absorbance v Action spectra v Difference spectra.
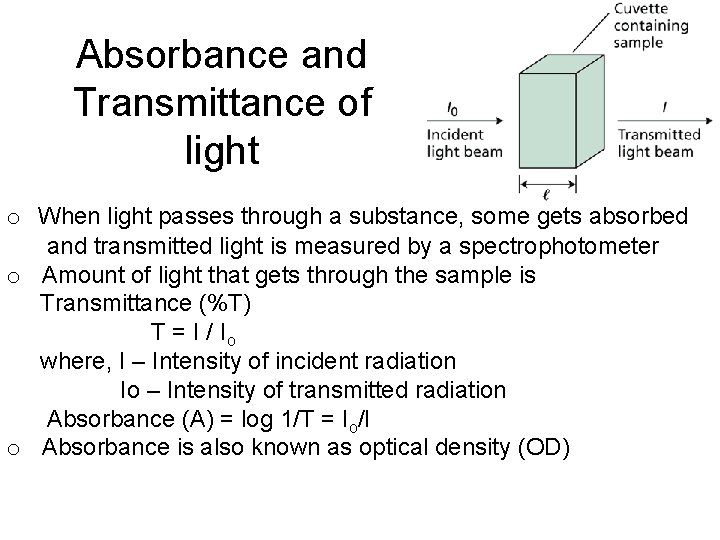
Absorbance and Transmittance of light o When light passes through a substance, some gets absorbed and transmitted light is measured by a spectrophotometer o Amount of light that gets through the sample is Transmittance (%T) T = I / Io where, I – Intensity of incident radiation Io – Intensity of transmitted radiation Absorbance (A) = log 1/T = Io/I o Absorbance is also known as optical density (OD)

Beers-Lambert Law o Absorbance is related to the concentration of the absorbing substance as well as the thickness of the layer o Mathematical relationship between concentration and absorbance is called Beer's Law o Mathematical relationship between concentration and thickness of the layer is called Lamberts Law A = εcl where A = absorbance ε = extinction coefficient (molar absorptivity) c = concentration, moles per liter l = path length, 1 cm o Transmittance is expressed on a range of 0 -100% o Absorbance has no units and varies from 0 to . o Normally between 0 -1 absorption, 90% of light is absorbed.

UV-Vis Spectrophotometer

UV-Vis Spectrophotometer • Absorption spectrum of a compound is a plot of the amount of light absorbed (extinction) against wavelength. • Colored compounds have one or more absorption maxima in visible region of the spectrum (400 -700 nm). • Absorption spectra in UV (200 -400 nm) and visible regions are due to energy transitions of both bonding and non-bonding outer electrons of the molecule.
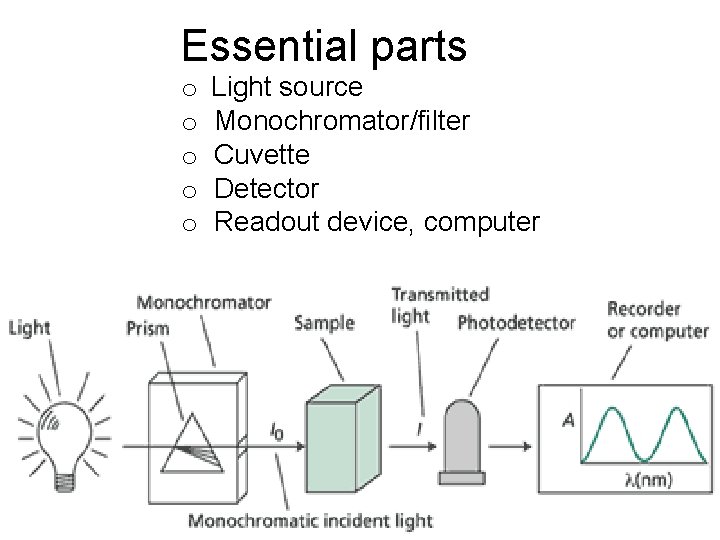
Essential parts o o o Light source Monochromator/filter Cuvette Detector Readout device, computer

Light source 1. Tungsten lamp - visible region 2. Hydrogen or deuterium lamp – UV region

Monochromators o An optical system which produces radiation of a single wavelength. o It is by refraction by a prism or diffraction by a grating o Prisms are made of glass for visible region but of quartz or silica for UV regions o Diffraction grating consist of a series of ruled lines on reflecting base and diffraction of white light gives a series of overlapping spectra. o Advantage of diffraction grating is their resolving power
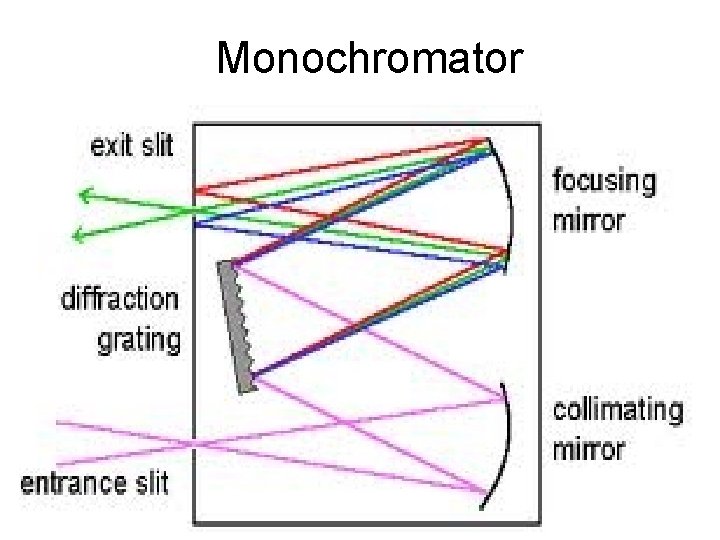
Monochromator

Internal parts of Spectroph otometer

Cuvettes • • • • Optically transparent cell (cuvette). Cuvette holder contain up to 4 cuvettes Need to have uniform dimensions Glass and plastic absorb below 310 nm Quartz or silica cells are transparent to wavelength > 180 nm Handling should be minimal Use pipette to fill and empty the cuvette Silica cuvettes susceptible to scratches Bubbles/ turbidity gives erroneous results Clean with mild sulphonic detergents, but not with concentrated acids or alkali. Common one have an optical path (l) length of 1 cm It holds 2. 5 -3. 0 cm 3 (ml) of sample Cells with path length of 100, 20, 5, 2 and 1 mm are available

Photocells § Convert quanta of radiation to electrical energy and it is then amplified, detected and recorded § Sensitive to radiation with a wavelength of about 400 nm and insensitive to wavelengths >550 nm. § Theoretically accuracy is 1± 0. 003. § Photons impinging on metal surface in a vacuum cause emission of electrons in proportion to the intensity of radiation. A positive electrode attracts these emitted electrons and hence a current flows, which causes a potential difference across a resistor. This potential is electronically amplified and the light absorbed is measured. § Photomultiplier tube are more sensitive than photocells.

Photomultiplier tube PMTs are very sensitive detectors used in fluorescence spectroscopy. • o o o Consist of a photocathode and dynodes in an evacuated chamber. Photons that strikes the photoemissive cathode emits electrons due to the photoelectric effect. Electrons are accelerated towards a series of additional electrodes called dynodes. Additional electrons are generated at each dynode. This cascading effect creates 105 to 107 electrons for each photon This amplified signal is finally collected at the anode where it can be measured.
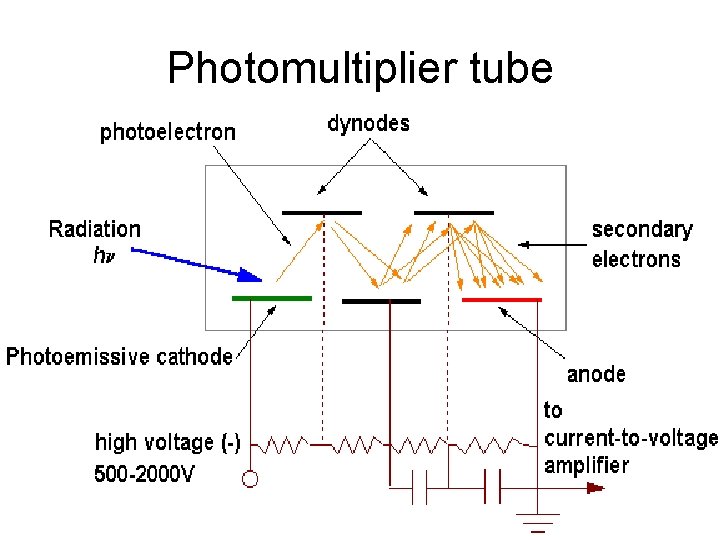
Photomultiplier tube
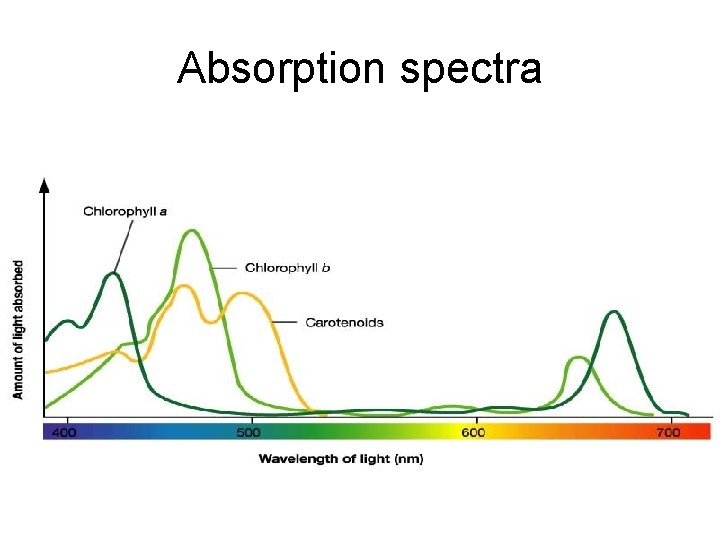
Absorption spectra

Applications 1. Measurement of chromatophore produced in color reaction. eg. Inorganic phosphates, aminoacids, proteins, phenols, tyrosine, carbohydrates, reducing sugars, pentoses, hexoses, glucose, hexosamines, DNA, RNA, sterols and cholesterols. 2. Visible & UV spectra helps to identify classes of compounds 3. Enzyme assays and kinetic analysis 4. Used in turbidometry and nephelometry
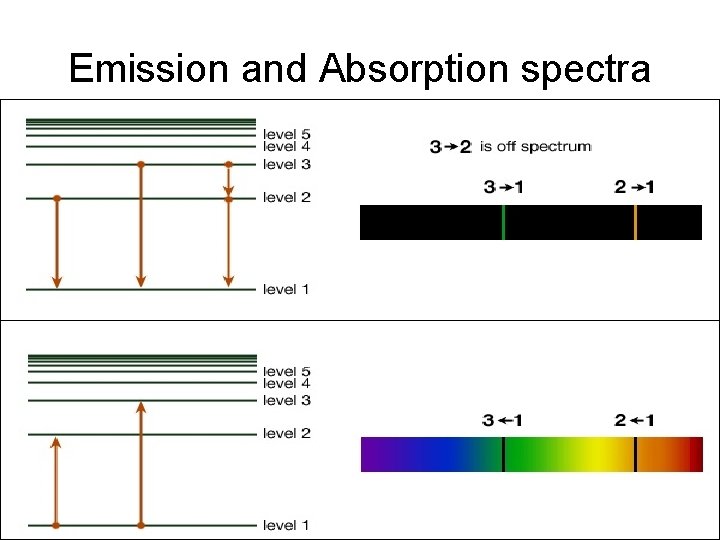
Emission and Absorption spectra

Emission spectra There actually three specific series of emission lines: • The Balmer series - emission that occurs within the visible spectrum • The Lyman series - emission that occurs within the ultraviolet spectrum • The Paschen series - emission that occurs within the infrared spectrum
- Slides: 30