Principles of Evidencebased Oncology European School of Oncology

Principles of Evidence-based Oncology European School of Oncology July 22 -24, 2004 Benjamin Djulbegovic, M. D. , Ph. D. Professor of Medicine and Oncology H. Lee Moffitt Cancer Center University of South Florida

The purpose of evidence-based medicine To learn how to: • distinguish evidence from propaganda (advertisement) • probability from certainty • data from assertions • rational belief from superstitions • science from folklore • theory from dogma (P. D. Hurd, Sci Am 1999; 281: 87 -92)

Part I. Understanding evidence

What is Evidence-Based Medicine? • Evidence is that “which enables the mind to see truth” (Webster’s Dictionary)

What Works in Health Care? • Well documented variation in clinical practice • “…actual treatments are often dictated by authority, personal experience, and habit” • Evidence-based medicine movement was born out of an attempt “to find out what works and what doesn’t in medicine”…through systematic, standardized analysis of available evidence. (Science 1996; 272: 22 -24)

EBM-historical perspectives Three movements (~1990): • Clinical Practice Guidelines and the need to standardize the practice (US) • Evidence Based Medicine and the need for critical appraisal (Canada) • Systematic reviews and the need for research synthesis-Cochrane Collaboration (UK)

Evidence-Based Medicine (EBM) • EBM is an approach to practicing medicine in which the clinicians are aware of the evidence in support of his/her practice, and the strength of that evidence • EBM is conscientious and judicious use of current best evidence from clinical care research in the management of individual patients (BMJ 1996; 312: 71)

Evidence-based Medicine: an Idea of the Year • A notion that claims of health intervention effects should be founded on scientifically valid empirical evidence was dubbed by the New York Times as one of the “Ideas of the Year” in 2001

Not All Evidence Is Created Equal • Understanding the hierarchy of clinical evidence • Developing rules of evidence

106 Number of Systems to Rate the Quality of Individual Studies AHRQ Report 2002 No. 02 -E 016

Important Domains and Elements for Systems to Rate Grade the Strength of Evidence Quality: the aggregate of quality ratings for individual studies, predicated on the extent to which bias was minimized. Quantity: magnitude of effect, numbers of studies, and sample size or power. Consistency: for any given topic, the extent to which similar findings are reported using similar and different study designs.

New system to unify/ improve on the existing systems of rating the strength of scientific evidence • GRADE system

Part II. Type of evidence

The nature of Medical Evidence Direct Evidence (Acting on basis of results of randomized controlled trials, case-control studies, etc) Indirect Evidence (Acting on basis of an understanding of biology of a disease and mechanism of action of the intervention)

Relevance: Type of Evidence • POE: Patient-oriented evidence – mortality, morbidity (disease-free survival), quality of life • DOE: Disease-oriented evidence – pharmacology (CR, PR, level of markers), pathophysiology, etiology

Part III Practice of EBM

Evidence-based Medicine • C -clinical question • L - literature search • A- appraise the evidence • D - decision making

Evidence-based medicine process EBM is problem-oriented practice that seeks to • Convert information needs regarding patient care into answerable questions • Track down, with maximum efficiency, the best evidence with which to answer them • Critically appraise that evidence for its validity (closeness to the truth), relevance (clinical applicability), and power (precision) • summarize and integrate this appraisal with clinical expertise to apply results in practice • evaluate performance and the impact of the process on clinical practice Sackett, 1995

Part IV Knowledge Resources

How much do we know? N=1851 Clinical Evidence, BMJ Publishing Group, 2003
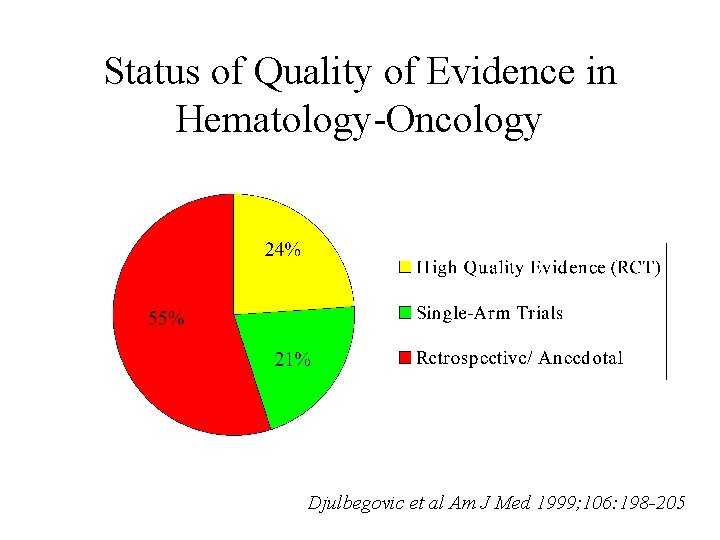
Status of Quality of Evidence in Hematology-Oncology Djulbegovic et al Am J Med 1999; 106: 198 -205

Information explosion and brain’s limited capacity for information processing and storage • 6 million articles is published in >20, 000 biomedical journals annually • Medical students are given to read > 11, 000 pages/yr • Practicing physicians need to instantaneously recall more than 2 million facts • MEDLINE Over 11 million indexed citations from~ 4, 500 journals – ~1. 5 million articles related to oncology – over 2, 000 completed references are added daily – Over 460, 000 added in 2001 • Randomized controlled trials – There about 60, 000 clinical trials currently under way with more than 200 clinical trials published in the scientific literature every year

Physicians’ unaided mind is sufficient for optimal decision-making • For every three patients seen physicians have 2 unanswered questions • This means that decisions could have been different 66% of time if the access to evidence (knowledge resources) had been optimal • – Ann Internal Med 1985; 103: 596 -599 – BMJ 1995; 310: 1085 -1086 JAMA 1998; 280: 1347 -1352)

Information explosion and need to filter and synthesize evidence • Need for filtering high-quality evidence • Systematic review/ Meta-analyses as a means to provide totality of evidence

The need for high-quality empirical evidence • To increase sensitivity in specificity of search for relevant literature, a variety of sources of primary and secondary evidence have been developed. • Catalog/database of the all randomized trials in a given field

Resources to Integrate Evidence • AHCPR Evidence-Based Practice Centers • ACP Journal Club/ Evidence-Based Medicine/Best Evidence • Journal of Family Practice “POEMs” • BMJ/ACP Clinical Evidence Series • Evidence-Based Oncology

Solving information paradox: SEARCHING FOR THE BEST EVIDENCE • Data consistently point that only about 1 -2% of published evidence meet criteria for validity and relevance to be of interest for practicing physicians (Evidence-based Oncology 2001; 2: 1)

Cochrane Database of Systematic Reviews The Cochrane Collaboration - an international network of individuals and institutions committed to preparing, maintaining, and promoting the accessibility of systematic reviews of the effects of health care interventions. Cochrane Systematic Reviews (3, 329) (March 2004) Database of Abstracts of Reviews of Effectiveness (4, 427) Registry of Randomized Controlled Trials (400, 976)

Knowledge Resources The Cochrane Library http: //www. update-software. com/cochrane/

Part VI Evidence and decision-making

HOW TO INTEGRATE BENEFITS AND RISKS OF AVAILABLE THERAPEUTIC OPTIONS • Should we always use the option with the best benefit/risk ratio? Efficacy=80% Toxicity=10% E/R=8 Efficacy=20% Toxicity=1% E/R=20

Understanding treatment benefits: framing effect There is a proposal to offer a coverage for breast screening program to women aged 20 -40 in your HMO. There are doubts about effectiveness of such a screening program. Here we present four statements derived from four randomized controlled trials published in the literature. On the basis of each statement you should indicate how likely you are to agree to the implementation of a breast screening program. Assume that the costs of each program are the same. Each result was deemed to be (statistically) significant. During a seven year follow up: • Program A reduced the rate of deaths from breast cancer by 33% • Program B produced an absolute reduction in deaths from breast cancer of 0. 06% • Program C increased the rate of patients surviving breast cancer from 99. 82% to 99. 88% • Program D meant that 1666 women needed to be screened to prevent one death from breast cancer Which program do you recommend? BMJ 1995; 311: 1056

Clinical Decision Making Patient circumstances Evidence from research Preferences, values and rights
- Slides: 33