PRINCIPLES OF ENVIRONMENTAL ENGINEERING AND SUSTAINABILITY LECTURE TWO

PRINCIPLES OF ENVIRONMENTAL ENGINEERING AND SUSTAINABILITY LECTURE TWO: TWO DIFFERENT TOPICS DEPARTMENT OF ENVIRONMENTAL ENGINEERING

OUTLINE This lecture will cover two different topics: ØUnits, Dimensions and Conversions. ØBiogeochemical Cycles 2

Units, Dimensions and Conversions Units • SI (Système International) Units: • mks: L = meters (m), M = kilograms (kg), T = seconds (s) • British Units: • L = inches, feet, miles, M = slugs (pounds), T = seconds • We will use mostly SI units, but you may run across some problems using British units. You should know how to convert back & forth. 3

Measurements and Units Length • The meter is presently defined as “the length of the path traveled by light in a vacuum during a time interval of 1/299, 792, 458 of a second. ” • Operational definition: measurement standard based on a laboratory procedure – can be reproduced “easily” 4

Length Distance Length (m) Radius of Visible Universe To Andromeda Galaxy To nearest star Earth to Sun Radius of Earth 1 x 1026 2 x 1022 4 x 1016 1. 5 x 1011 6. 4 x 106 Sears Tower Football Field Tall person Thickness of paper Wavelength of blue light Diameter of hydrogen atom Diameter of proton 4. 5 x 102 1. 0 x 102 2 x 100 1 x 10 -4 4 x 10 -7 1 x 10 -10 1 x 10 -15 5

Measurements and Units Time • The second was once defined by the rotation of the earth – but this rotation is not constant! • In 1967, the second was given an operational definition based on the transitions of the cesium atom. • The second is currently defined as “the duration of 9, 192, 631, 770 periods of the radiation corresponding to the transition between the two hyperfine levels of the ground state of the cesium-133 atom” 6

Time Interval Age of Universe Age of Grand Canyon Avg age of college student One year One hour Light travel from Earth to Moon One cycle of guitar A string One cycle of FM radio wave One cycle of visible light Time for light to cross a proton Time (s) 5 x 1017 3 x 1014 6. 3 x 108 3. 2 x 107 3. 6 x 103 1. 3 x 100 2 x 10 -3 6 x 10 -8 1 x 10 -15 1 x 10 -24 7

Measurements and Units • Mass • The least “convenient” of the defined units. • The kilogram is defined by an object – not by an experiment reproducible in laboratories. • At the moment, the prototype kilogram is made of a special platinum-iridium alloy that does not corrode. • Work is in progress to define the kilogram based on the mass of a single atom – this would lead to an operational definition 8

Mass Object visible universe Milky Way galaxy Sun Earth Boeing 747 Car Student Dust particle Bacterium Proton Electron Mass (kg) ~ 1052 7 x 1041 2 x 1030 6 x 1024 4 x 105 1 x 103 7 x 101 1 x 10 -9 1 x 10 -15 2 x 10 -27 9 x 10 -31 9

Measurements and Units • Other SI units • Throughout this course and the second course in the sequence, we will meet more SI units • Some will be unique, e. g. • ampère (A): measures electric current • kelvin (K): measures temperature • radian (rad): measures angles • Some will be combinations of the “main three, ” e. g. • Newton (N = kg·m/s 2): measures force • Pascal (Pa = kg/m·s 2): measures pressure 10
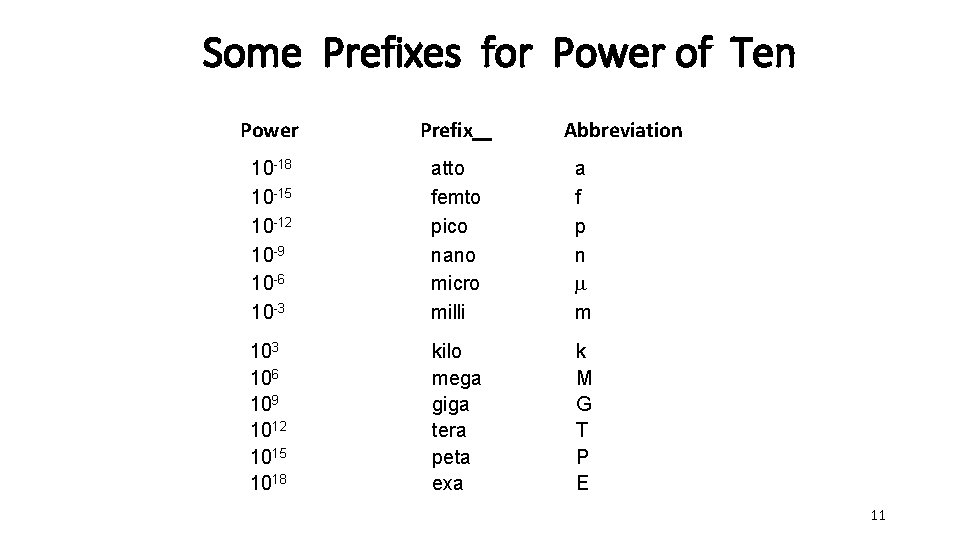
Some Prefixes for Power of Ten Power Prefix Abbreviation 10 -18 10 -15 10 -12 10 -9 10 -6 10 -3 atto femto pico nano micro milli a f p n m m 103 106 109 1012 1015 1018 kilo mega giga tera peta exa k M G T P E 11

Converting between different systems of units • Useful Conversion factors: • 1 inch = 2. 54 cm • 1 m = 3. 28 ft • 1 mile = 5280 ft • 1 mile = 1. 61 km • Example: convert miles per hour to meters per second: 12

Converting between different systems of units • When on travel in Europe you rent a small car which consumes 6 liters of gasoline per 100 km. What is the MPG of the car ? • Useful Conversion factors: • 1 gallon = 4 liters • 1 mile = 1. 61 km Solution = 41 MPG ! 13

Dimensional Analysis • This is a very important tool to check your work • It’s also very easy! • Example: Doing a problem you get the answer for distance d = v t 2 ( velocity x time 2 ) Quantity on left side = L Quantity on right side = L / T x T 2 = L x T • Left units and right units don’t match, so answer must be wrong !! 14

There is a famous Einstein's equation connecting energy and mass (relativistic). Using dimensional analysis find which is the correct form of this equation : (a) l (b) (c) Note : çc is speed of light (L/T) ç E is energy (M L 2 / T 2) Solution -> (b) 15

Biogeochemical Cycles Ø A biogeochemical cycle is the cyclic movement of a substance (for example, water, carbon, nitrogen, or phosphate) through the biotic (living) and abiotic (nonliving) environments. Ø Biogeochemical cycles are a key component of ecosystems ecology. 16
Nature reuses everything. All of the matter that cycles through living organisms are important in maintaining the health of the ecosystem. 17
• Biogeochemical cycles of matter involve biological processes, geological processes, and chemical processes. • As matter moves through these cycles, it is never created or destroyed—just changed. 18
* In almost all biochemical cycles, there is much less of the substance in the living reservoir than the nonliving reservoir. 19
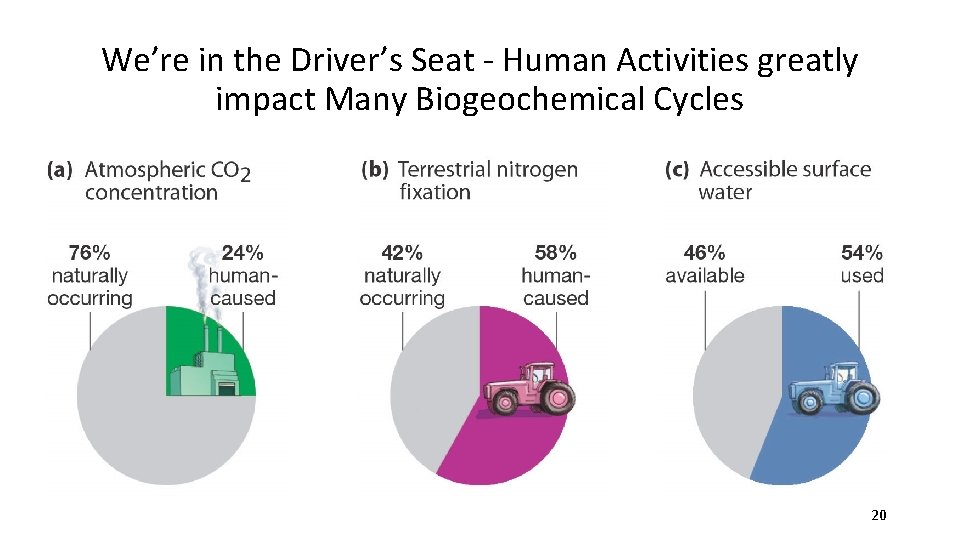
We’re in the Driver’s Seat - Human Activities greatly impact Many Biogeochemical Cycles 20

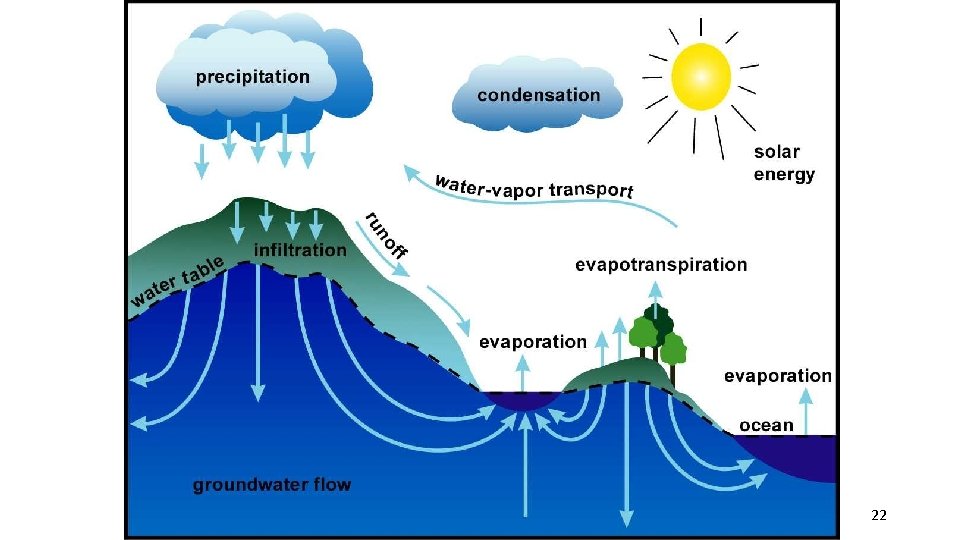
1. The Water (hydrologic) Cycle üPlants absorb water from the soil. üWater may percolate down through the soil and eventually collect in pools (groundwater). üTranspiration (release of water through leaves due to the sun) and respiration return water to the atmosphere. üPrecipitation (rain, snow, etc) returns water to the soil 21
22
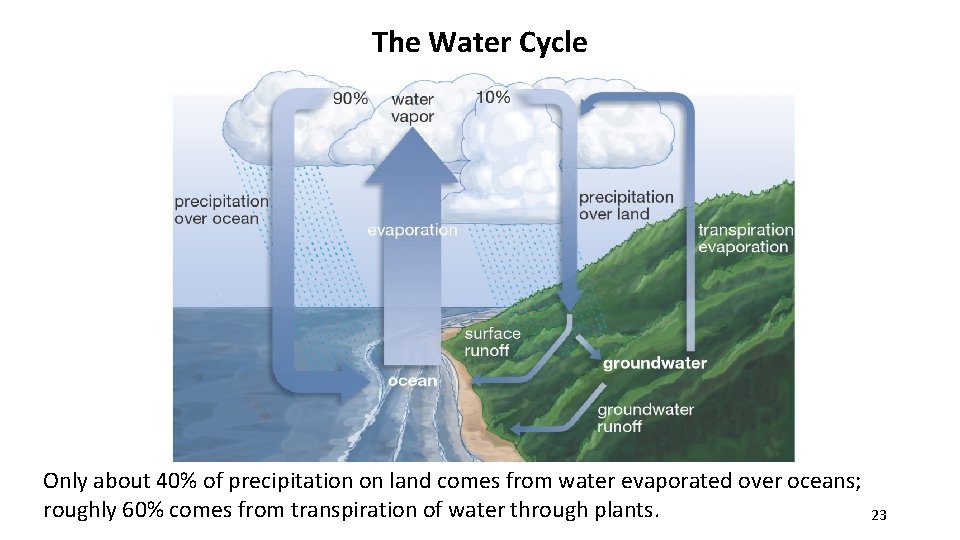
The Water Cycle Only about 40% of precipitation on land comes from water evaporated over oceans; roughly 60% comes from transpiration of water through plants. 23

Human Impact on the Water Cycle • Withdraw large quantities of fresh water – water diversion, groundwater depletion, wetland drainage. • Clearing vegetation from land • Increases runoff, reduces infiltration, increases flooding, increases soil erosion • Modify water quality-adding nutrients (phosphates, nitrates). 24

2. The Carbon Cycle Photosynthesis and Cell Respiration cycle carbon and oxygen through the environment • Carbon is fixed by plants • 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 • Carbon is given off by consumers • C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O • Organisms containing carbon form fossil fuels • Burning fossil fuels releases carbon • 2 C 8 H 18 + 25 O 2 16 CO 2 + 18 H 2 O 25
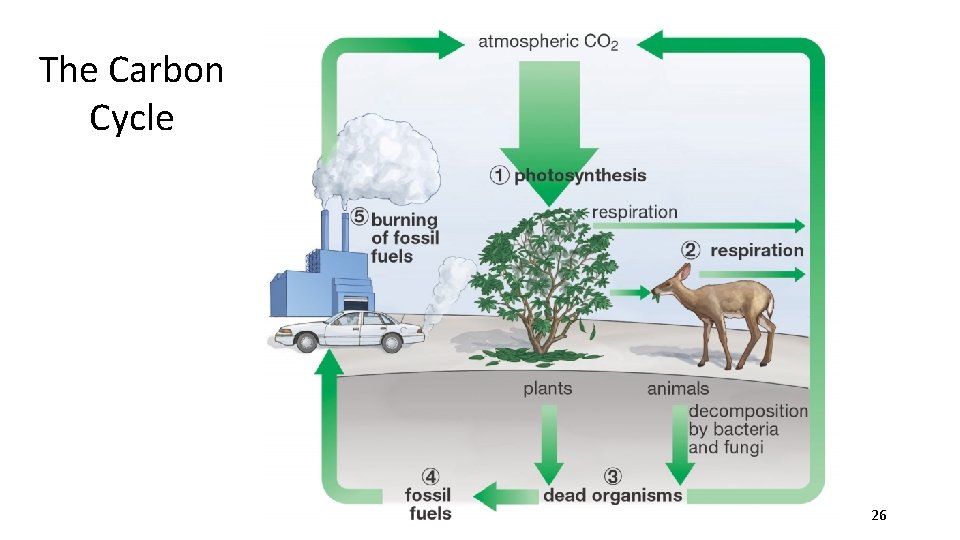
The Carbon Cycle 26
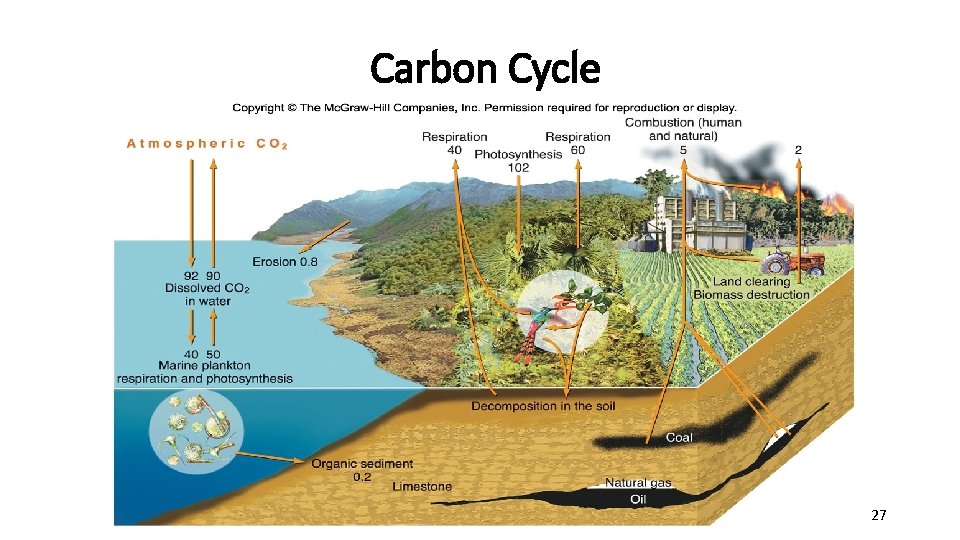
Carbon Cycle 27

Human Impact on the Carbon Cycle • Burning of fossil fuels & biomass (wood) generates huge amounts of carbon dioxide that cannot be taken up fast enough by the carbon sinks (oceans, forests). • This excess carbon dioxide contributes to global warming, which may lead to loss of biodiversity globally, rising sea levels, more violent storms, and changes in precipitation patterns. • Removal of vegetation – decreases primary production (decreases carbon fixation) and subsequently, biodiversity. 28

3. The Nitrogen cycle Atmospheric nitrogen (N 2) makes up nearly 78%-80% of air. Organisms cannot use it in that form. Lightning and some species of bacteria convert nitrogen into usable forms. 29

ü Nitrogen fixation-convert atmospheric nitrogen (N 2) into ammonium (NH 4+) which can be used to make organic compounds like amino acids. N 2 + NH 4 ü Only in certain types of bacteria and industrial technologies can fix nitrogen. üAmmonia is further changed by other types of soil bacteria (nitrification) into nitrate, which is taken up by plants and passed on to animals as they consume the plants. üEventually this is returned to the nonliving reservoir when the organism dies and decays. 30
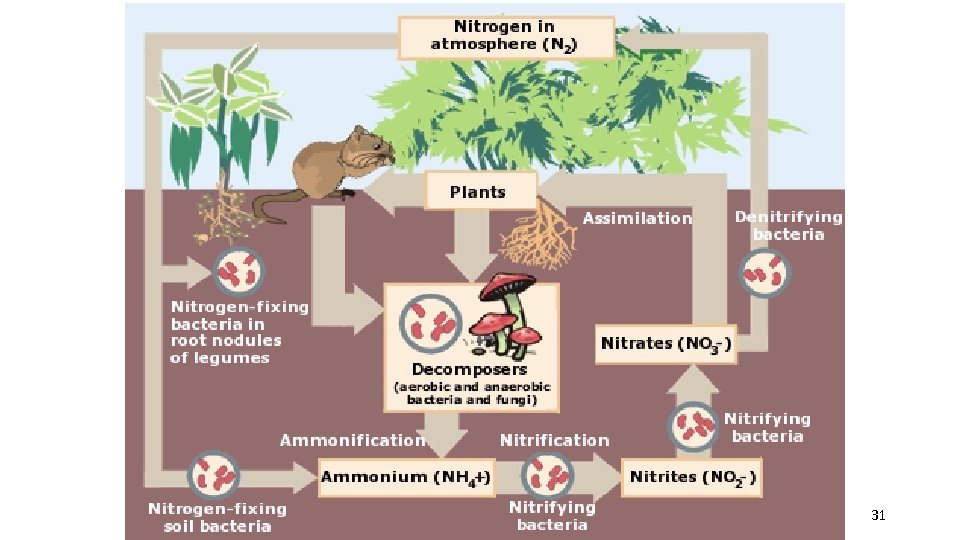
31
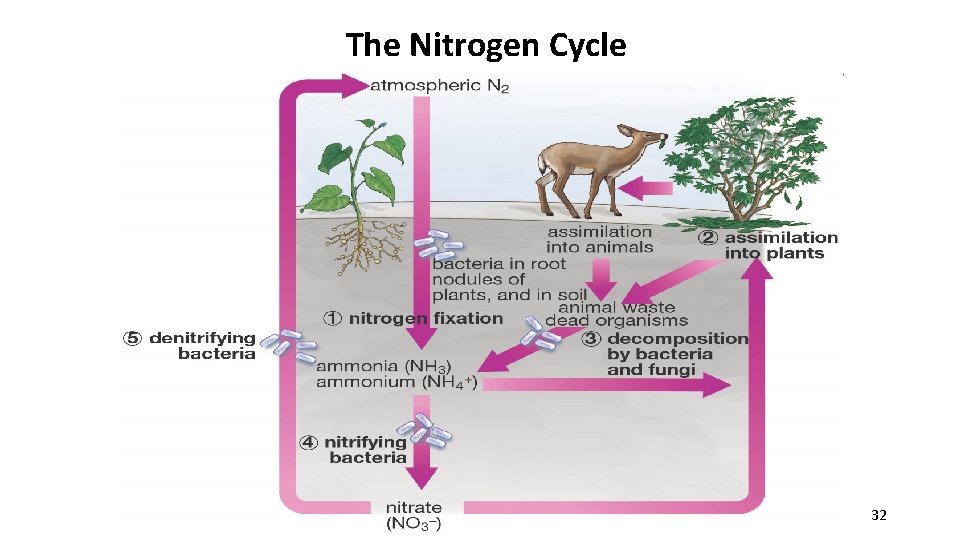
The Nitrogen Cycle 32

Humans Play a Major Role in the Nitrogen Cycle • Use of synthetic nitrogen fertilizers may stimulate algal blooms (due to agricultural runoff), which depletes oxygen and decreases biodiversity. Aerial fertilization (with nitrogen) of sugar beets. 33

Human impact on the Nitrogen Cycle Continued…. • Land management: Nitrogen-fixing crops (legumes) add more useable nitrogen to the soil. (Example of crop rotation: corn, soybeans, corn, soybeans) ØOn the other hand, overplanting of crops depletes nitrogen from soil. • Burning fossil fuels- forms nitrogen dioxide (NO 2) in atmosphere, which can react with water to form nitric acid (HNO 3) & causes acid rain 34

The Phosphorous Cycle Phosphorus - usually found in soil and rocks in the form of calcium phosphate. • Calcium phosphate dissolves in water and forms phosphate ions. We can’t use this inorganic form of phosphorous. ** Phosphorous is an important component of DNA, bones and teeth • Plants take up inorganic phosphate via their roots. It is then used to build organic phosphate. • Consumers get organic phosphate from plants. • Decomposers return phosphate to soil • Phosphate leaches into water supply • May form new phosphate containing rock 35
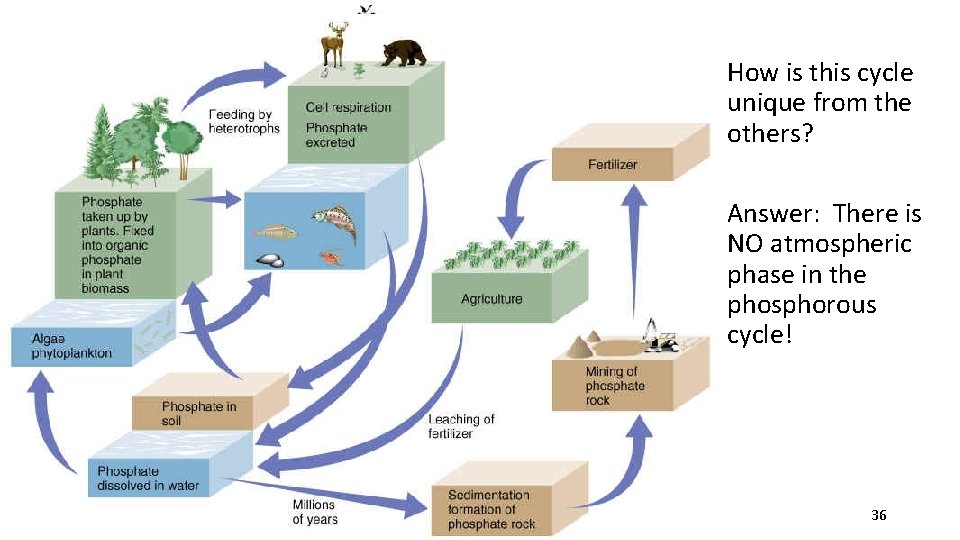
How is this cycle unique from the others? Answer: There is NO atmospheric phase in the phosphorous cycle! 36

Phosphorous Cycle 37

Human Impact on the Phosphorous Cycle • Mining of large quantities of phosphate rock-used for organic fertilizers and detergents • Runoff of wastes and fertilizers causes accumulation in lakes and ponds killing aquatic organisms Øleads to excessive algal growth, depletion of oxygen, & decrease in biodiversity; eutrophication ("over nourishment") 38

End of Lecture Two 39
- Slides: 39