Principles of chemistry Macromolecules of life Lecture 3
























- Slides: 43

Principles of chemistry Macromolecules of life Lecture 3

Carbon: The Backbone of Life • Living organisms consist mostly of carbon-based compounds • Carbon is unparalleled in its ability to form large, complex, and diverse molecules • Proteins, DNA, carbohydrates, and other molecules that distinguish living matter are all composed of carbon compounds • Biological diversity has its molecular basis in carbon’s ability to form a huge number of molecules with particular shapes and chemical properties

Organic chemistry is the study of carbon compounds • Organic chemistry is the study of compounds that contain carbon • Organic compounds range from simple molecules to colossal ones • Most organic compounds contain hydrogen atoms in addition to carbon atoms

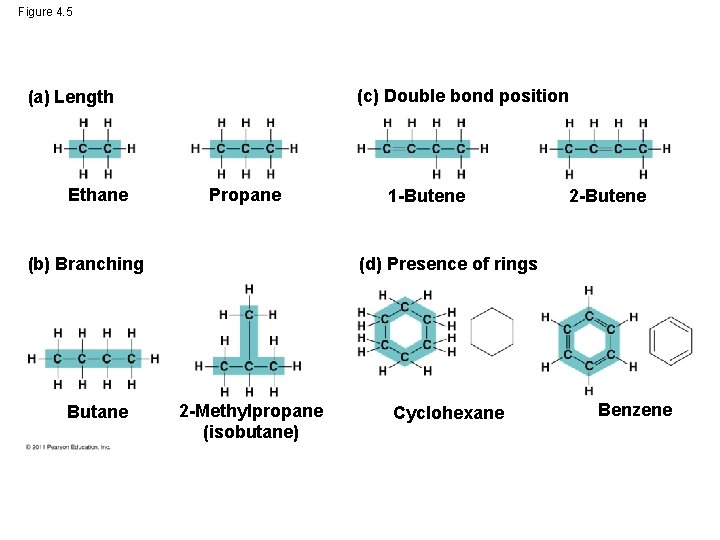
Molecular Diversity Arising from Carbon Skeleton Variation • Carbon chains form the skeletons of most organic molecules • Carbon chains vary in length and shape • Because carbon atoms can form up to four covalent bonds, molecules containing carbon can form straight chains, branches, or even rings, balls, tubes, and coils

Figure 4. 5 (c) Double bond position (a) Length Ethane Propane (b) Branching Butane 1 -Butene 2 -Butene (d) Presence of rings 2 -Methylpropane (isobutane) Cyclohexane Benzene

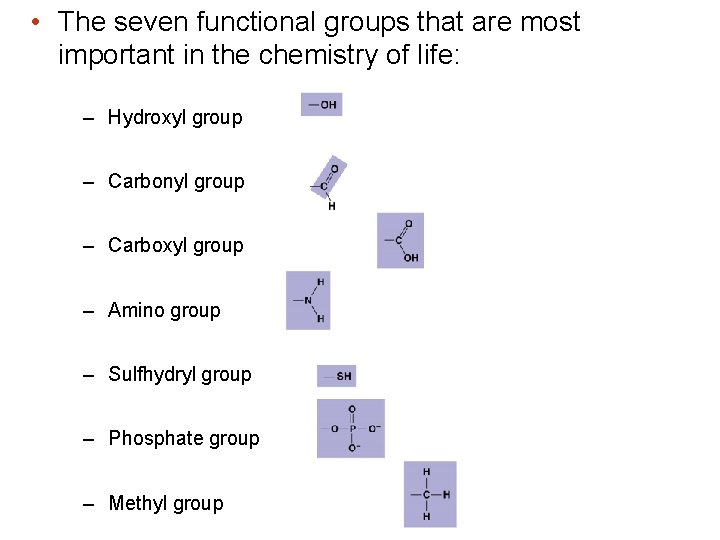
A few chemical groups are key to the functioning of biological molecules • Distinctive properties of organic molecules depend on the carbon skeleton and on the molecular components attached to it • Functional groups are the components of organic molecules that are most involved in chemical reactions • The number and arrangement of functional groups give each molecule its unique properties • A structural building block that determines the characteristics of the compound

Figure 4. UN 02 Estradiol Testosterone

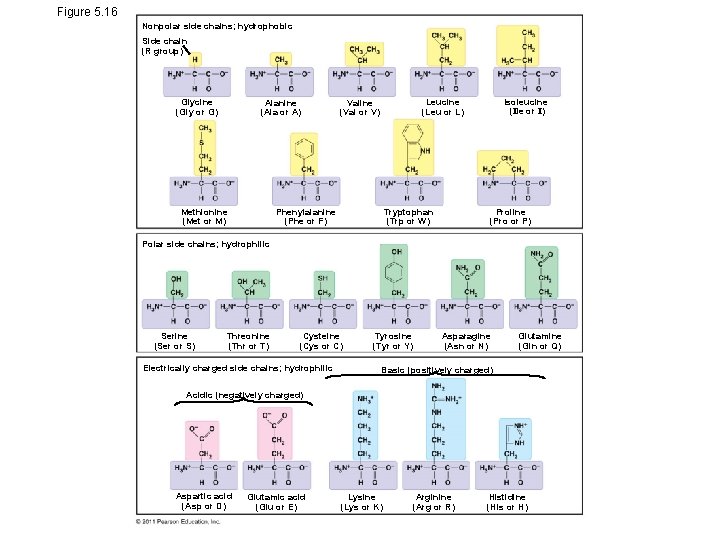
• The seven functional groups that are most important in the chemistry of life: – Hydroxyl group – Carbonyl group – Carboxyl group – Amino group – Sulfhydryl group – Phosphate group – Methyl group

The Molecules of Life • All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic acids • Macromolecules are large molecules composed of thousands of covalently connected atoms • Molecular structure and function are inseparable

• Four main types of organic macromolecules: Carbohydrates Lipids Proteins Nucleic Acids

: Macromolecules are polymers, built from monomers • A polymer is a long molecule consisting of many similar building blocks • These small building-block molecules are called monomers • Three of the four classes of life’s organic molecules are polymers – Carbohydrates – Proteins – Nucleic acids

Carbohydrates serve as fuel and building material • Carbohydrates include sugars and the polymers of sugars • The simplest carbohydrates are monosaccharides, or single sugars (Glucose (C 6 H 12 O 6) is the most common monosaccharide • Carbohydrate macromolecules are polysaccharides, polymers composed of many sugar building blocks • Monosaccharides serve as a major fuel for cells and as raw material for building molecules


Figure 5. 3 Aldoses (Aldehyde Sugars) Ketoses (Ketone Sugars) Trioses: 3 -carbon sugars (C 3 H 6 O 3) Glyceraldehyde Dihydroxyacetone Pentoses: 5 -carbon sugars (C 5 H 10 O 5) Ribose Ribulose Hexoses: 6 -carbon sugars (C 6 H 12 O 6) Glucose Galactose Fructose

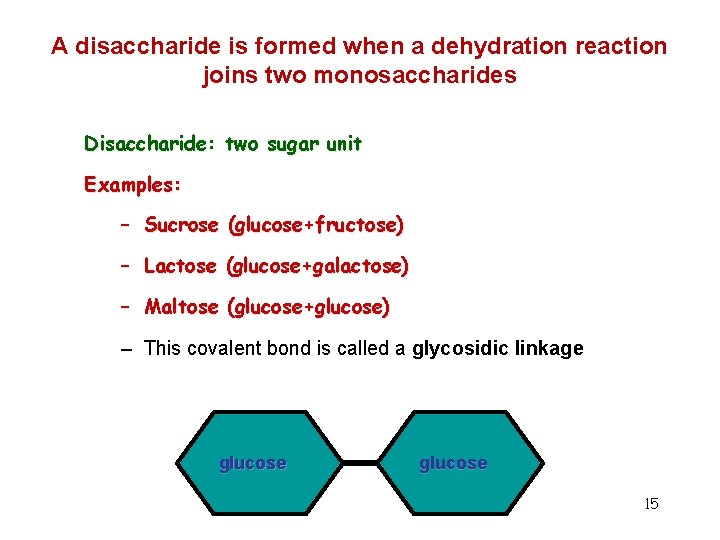
A disaccharide is formed when a dehydration reaction joins two monosaccharides Disaccharide: two sugar unit Examples: – Sucrose (glucose+fructose) – Lactose (glucose+galactose) – Maltose (glucose+glucose) – This covalent bond is called a glycosidic linkage glucose 15

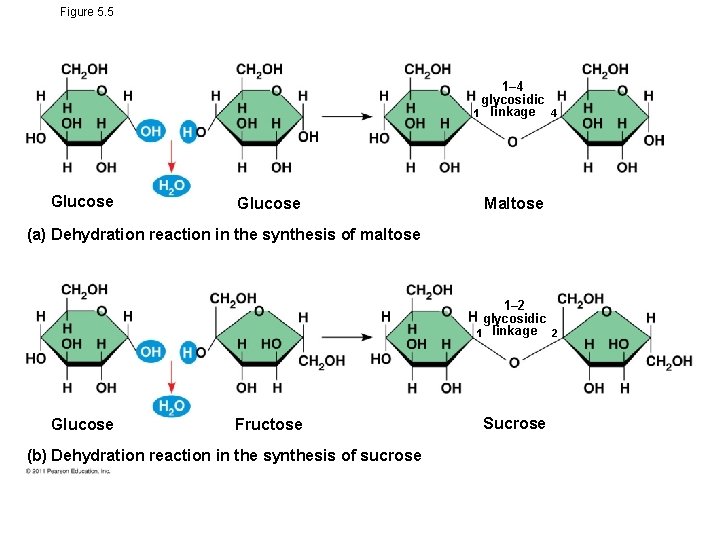
Figure 5. 5 1– 4 glycosidic 1 linkage 4 Glucose Maltose (a) Dehydration reaction in the synthesis of maltose 1– 2 glycosidic 1 linkage 2 Glucose Fructose (b) Dehydration reaction in the synthesis of sucrose Sucrose

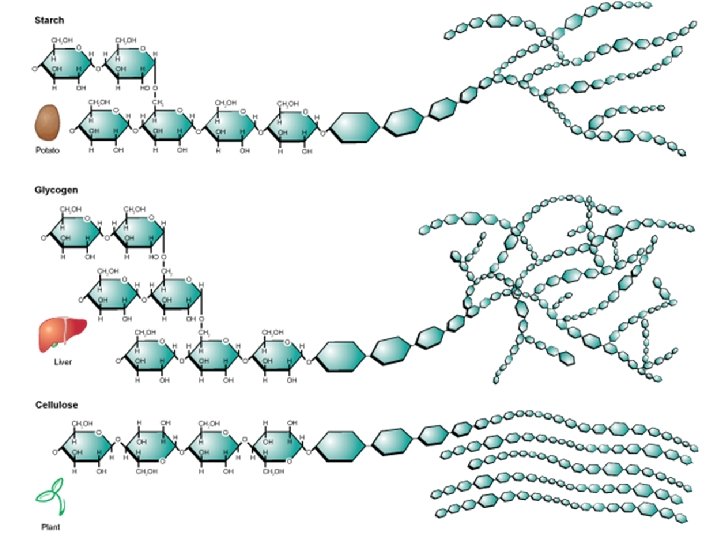
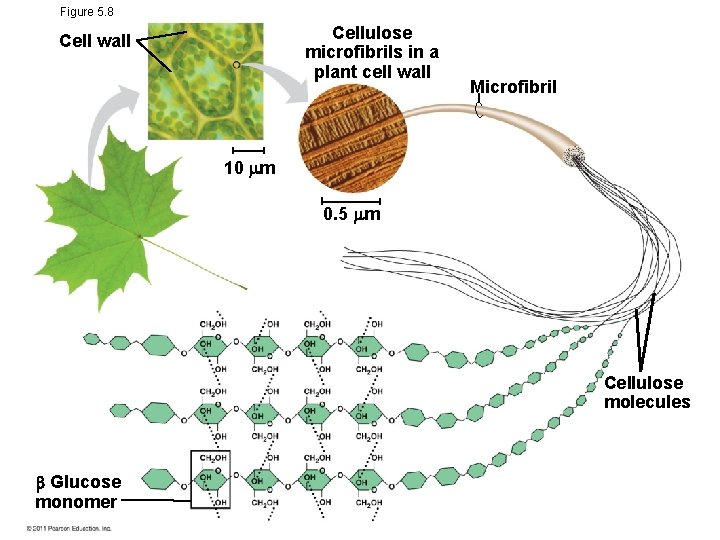
Storage Polysaccharides • Starch, a storage polysaccharide of plants, consists entirely of glucose monomers • Plants store surplus starch as granules within chloroplasts and other plastids • The polysaccharide cellulose is a major component of the tough wall of plant cells • Glycogen is a storage polysaccharide in animals • Humans and other vertebrates store glycogen mainly in liver and muscle cells

Figure 5. 8 Cellulose microfibrils in a plant cell wall Cell wall Microfibril 10 m 0. 5 m Cellulose molecules Glucose monomer

• Enzymes that digest starch by hydrolyzing linkages can’t hydrolyze linkages in cellulose • Cellulose in human food passes through the digestive tract as insoluble fiber • Some microbes use enzymes to digest cellulose • Many herbivores, from cows to termites, have symbiotic relationships with these microbes

Lipids are a diverse group of hydrophobic molecules • Lipids are the one class of large biological molecules that do not form polymers • The unifying feature of lipids is having little or no affinity for water • Lipids are hydrophobic because they consist mostly of hydrocarbons, which form nonpolar covalent bonds • The most biologically important lipids are fats, phospholipids, and steroids

Functions: • Energy storage • membrane structure • Protecting against desiccation (drying out). • Insulating against cold. • Absorbing shocks. • Regulating cell activities by hormone actions.

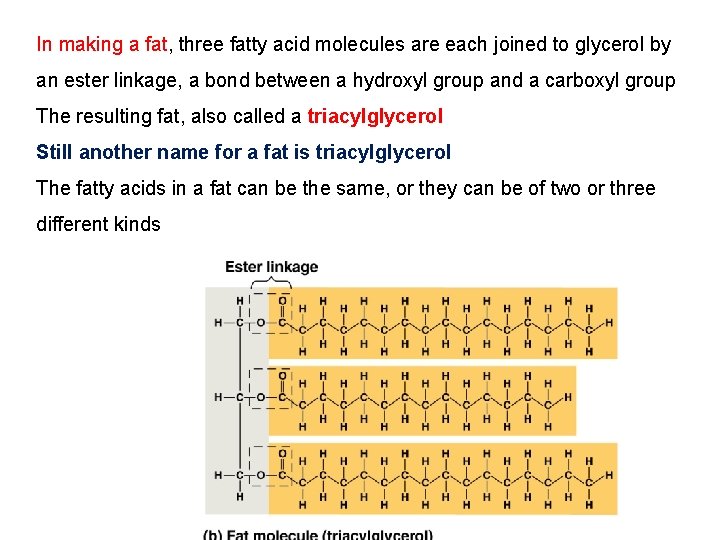
Fats • Fats are constructed from two types of smaller molecules: glycerol and fatty acids • Glycerol is a three-carbon alcohol with a hydroxyl group attached to each carbon • A fatty acid consists of a carboxyl group attached to a long carbon skeleton

In making a fat, three fatty acid molecules are each joined to glycerol by an ester linkage, a bond between a hydroxyl group and a carboxyl group The resulting fat, also called a triacylglycerol Still another name for a fat is triacylglycerol The fatty acids in a fat can be the same, or they can be of two or three different kinds

• A fatty acid has a long carbon skeleton, usually 16 or 18 carbon atoms in length • The carbon at one end of the skeleton is part of a carboxyl group, the functional group that gives these molecules the name fatty acid. • The rest of the skeleton consists of a hydrocarbon chain • The relatively nonpolar C¬H bonds in the hydrocarbon chains of fatty acids are the reason fats are hydrophobic • Fats separate from water because water molecules form hydrogen bonds with each other and exclude the fats

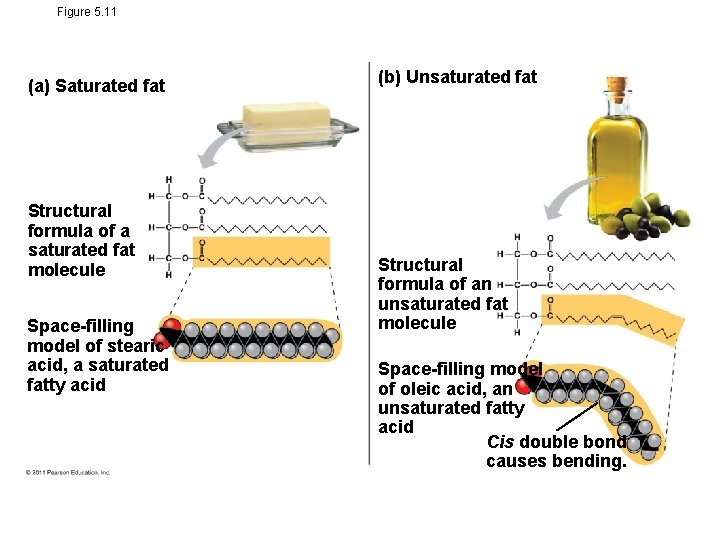
• Fatty acids vary in length (number of carbons) and in the number and locations of double bonds • Saturated fatty acids have the maximum number of hydrogen atoms possible and no double bonds • Unsaturated fatty acids have one or more double bonds

Figure 5. 11 (a) Saturated fat Structural formula of a saturated fat molecule Space-filling model of stearic acid, a saturated fatty acid (b) Unsaturated fat Structural formula of an unsaturated fat molecule Space-filling model of oleic acid, an unsaturated fatty acid Cis double bond causes bending.

Saturated Fatty Acids: • Solid at room temperature • Come from animals • Full (saturated) of hydrogens on single bonded carbons

Unsaturated fatty acids • Are not full of hydrogen because of double bonded carbons • Come from plants • Liquid at room temperature • Monounsaturated = 1 double bond between carbons; Polyunsaturated = more than 1 double bond between carbons

In contrast, the fats of plants and fishes are generally unsaturated, meaning that they are built of one or more types of unsaturated fatty acids. Usually liquid at room temperature, plant and fish fats are referred to as oils—olive oil and cod liver oil are examples. The kinks where the cis double bonds are located prevent the molecules from packing together closely enough to solidify at room temperature. The phrase “hydrogenated vegetable oils” on food labels means that unsaturated fats have been synthetically converted to saturated fats by adding hydrogen. Peanut butter, margarine, and many other products are hydrogenated to prevent lipids from separating out in liquid (oil) form. A diet rich in saturated fats is one of several factors that may contribute to the cardiovascular disease known as atherosclerosis. In this condition, deposits called plaques develop within the walls of blood vessels, causing inward bulges that impede blood flow and reduce the resilience of the vessels. Recent studies have shown that the process of hydrogenating vegetable oils produces not only saturated fats but also unsaturated fats with trans double bonds. These trans fats may contribute more than saturated fats to atherosclerosis and other problems

• Certain unsaturated fatty acids are not synthesized in the human body • These must be supplied in the diet • These essential fatty acids include the omega-3 fatty acids, required for normal growth, and thought to provide protection against cardiovascular disease

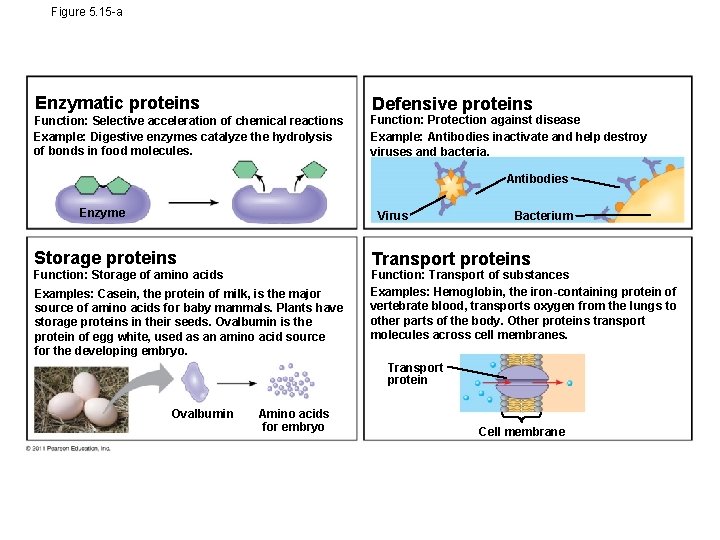
Proteins include a diversity of structures, resulting in a wide range of functions • Nearly every dynamic function of a living being depends on proteins • Proteins account for more than 50% of the dry mass of most cells • Protein functions include speed up chemical reaction, structural support, storage, transport, cellular communications, movement, and defense against foreign substances

Figure 5. 15 -a Enzymatic proteins Defensive proteins Function: Selective acceleration of chemical reactions Example: Digestive enzymes catalyze the hydrolysis of bonds in food molecules. Function: Protection against disease Example: Antibodies inactivate and help destroy viruses and bacteria. Antibodies Enzyme Virus Storage proteins Bacterium Transport proteins Function: Storage of amino acids Examples: Casein, the protein of milk, is the major source of amino acids for baby mammals. Plants have storage proteins in their seeds. Ovalbumin is the protein of egg white, used as an amino acid source for the developing embryo. Function: Transport of substances Examples: Hemoglobin, the iron-containing protein of vertebrate blood, transports oxygen from the lungs to other parts of the body. Other proteins transport molecules across cell membranes. Transport protein Ovalbumin Amino acids for embryo Cell membrane

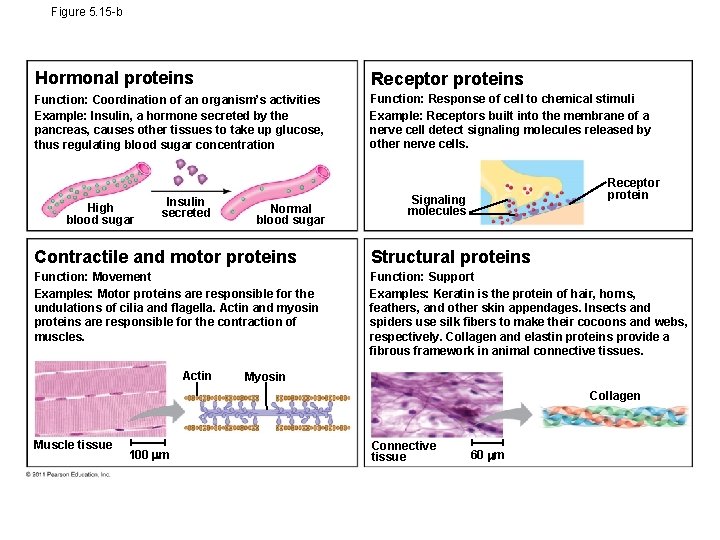
Figure 5. 15 -b Hormonal proteins Receptor proteins Function: Coordination of an organism’s activities Example: Insulin, a hormone secreted by the pancreas, causes other tissues to take up glucose, thus regulating blood sugar concentration Function: Response of cell to chemical stimuli Example: Receptors built into the membrane of a nerve cell detect signaling molecules released by other nerve cells. High blood sugar Insulin secreted Normal blood sugar Receptor protein Signaling molecules Contractile and motor proteins Structural proteins Function: Movement Examples: Motor proteins are responsible for the undulations of cilia and flagella. Actin and myosin proteins are responsible for the contraction of muscles. Function: Support Examples: Keratin is the protein of hair, horns, feathers, and other skin appendages. Insects and spiders use silk fibers to make their cocoons and webs, respectively. Collagen and elastin proteins provide a fibrous framework in animal connective tissues. Actin Myosin Collagen Muscle tissue 100 m Connective tissue 60 m

• A human has tens of thousands of different proteins, each with a specific structure and function; proteins, in fact, are the most structurally sophisticated molecules known. • Consistent with their diverse functions, they vary extensively in structure, each type of protein having a unique three-dimensional shape.

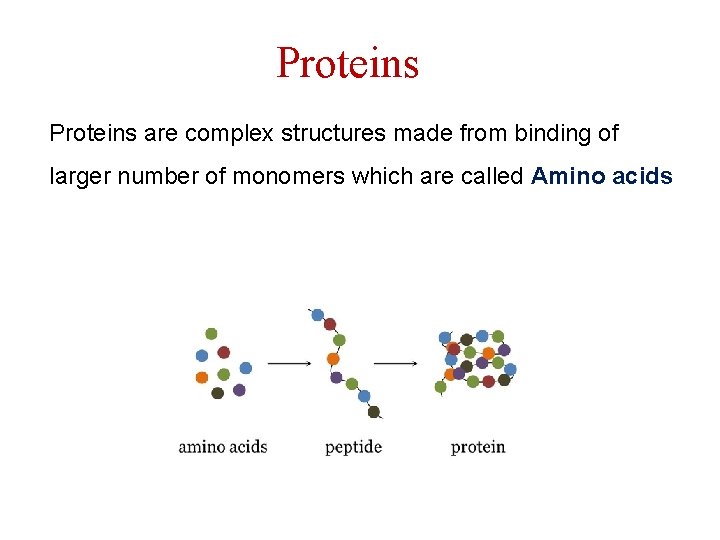
Proteins are complex structures made from binding of larger number of monomers which are called Amino acids

Figure 5. 16 Nonpolar side chains; hydrophobic Side chain (R group) Glycine (Gly or G) Alanine (Ala or A) Methionine (Met or M) Isoleucine (Ile or I) Leucine (Leu or L) Valine (Val or V) Phenylalanine (Phe or F) Tryptophan (Trp or W) Proline (Pro or P) Polar side chains; hydrophilic Serine (Ser or S) Threonine (Thr or T) Cysteine (Cys or C) Electrically charged side chains; hydrophilic Tyrosine (Tyr or Y) Asparagine (Asn or N) Glutamine (Gln or Q) Basic (positively charged) Acidic (negatively charged) Aspartic acid (Asp or D) Glutamic acid (Glu or E) Lysine (Lys or K) Arginine (Arg or R) Histidine (His or H)

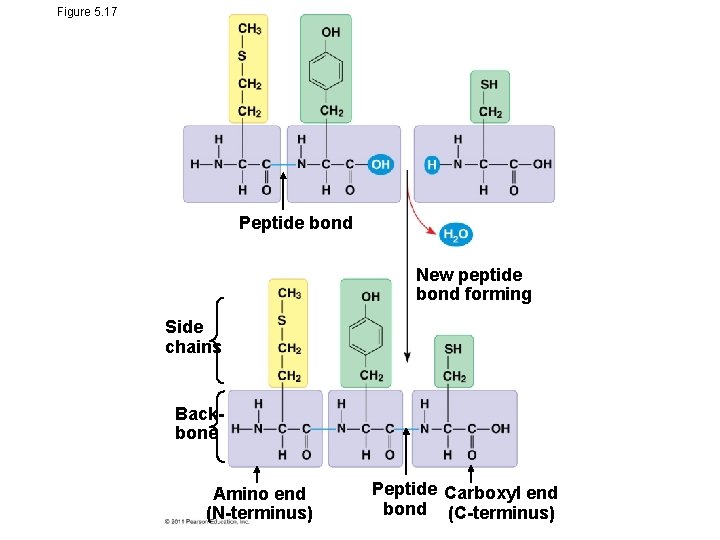
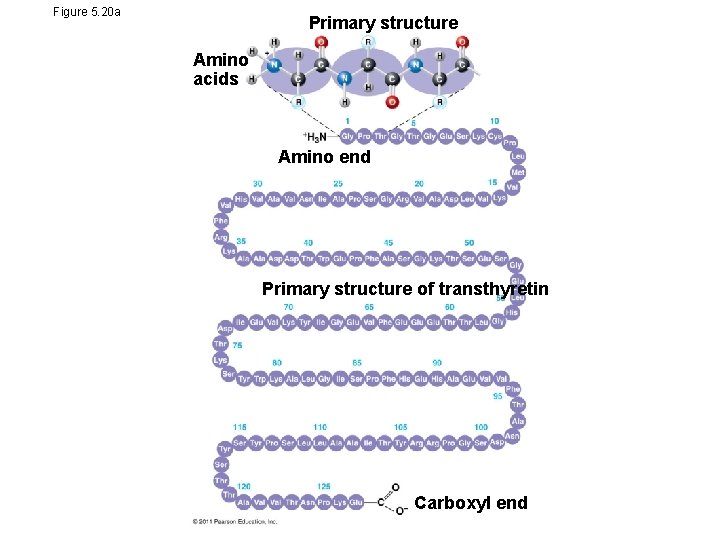
Amino Acid Polymers • Amino acids are linked by peptide bonds • A polypeptide is a polymer of amino acids • Polypeptides range in length from a few to more than a thousand monomers • Each polypeptide has a unique linear sequence of amino acids, with a carboxyl end (C-terminus) and an amino end (N-terminus)

Figure 5. 17 Peptide bond New peptide bond forming Side chains Backbone Amino end (N-terminus) Peptide Carboxyl end bond (C-terminus)

Peptide, polypeptide and protein • A peptide is a compound consisting of 2 or more amino acids. • Oligopeptides have 10 or fewer amino acids. • Polypeptides and proteins are chains of 10 or more amino acids; however, peptides consisting of more than 50 amino acids are classified as proteins

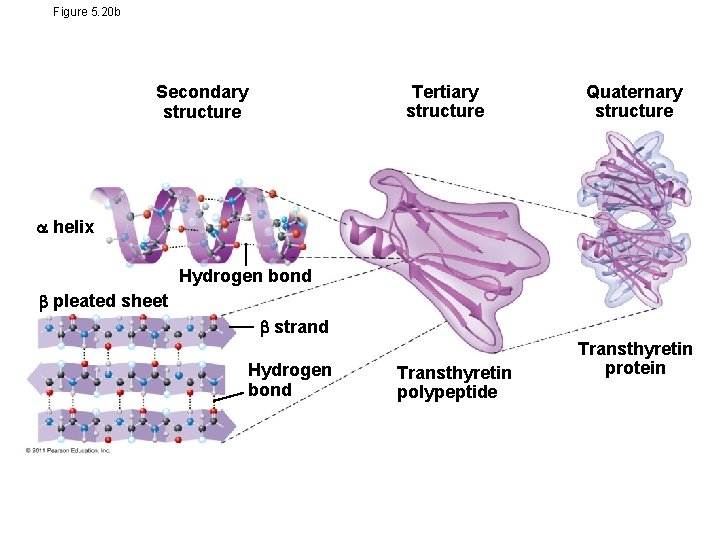
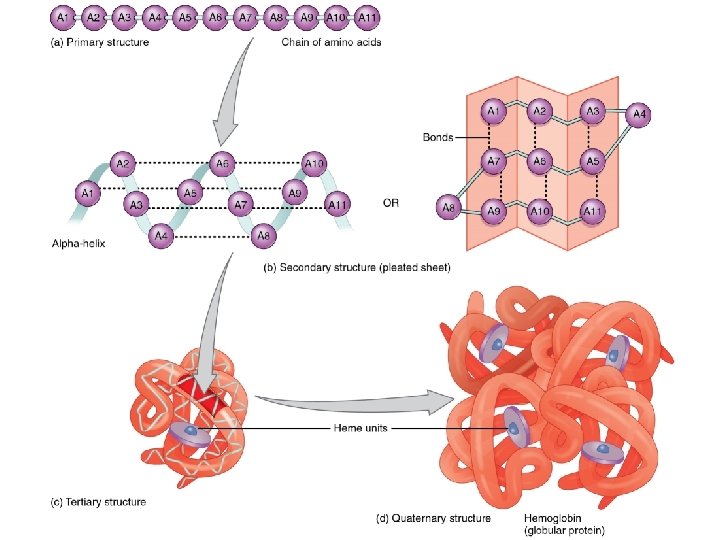
Four Levels of Protein Structure • The primary structure of a protein is its unique sequence of amino acids • Secondary structure, found in most proteins, consists of coils and folds in the polypeptide chain • Tertiary structure is determined by interactions among various side chains (R groups) • Quaternary structure results when a protein consists of multiple polypeptide chains

Figure 5. 20 a Primary structure Amino acids Amino end Primary structure of transthyretin Carboxyl end

Figure 5. 20 b Tertiary structure Secondary structure Quaternary structure helix Hydrogen bond pleated sheet strand Hydrogen bond Transthyretin polypeptide Transthyretin protein
