Principles of Bioinorganic Chemistry 2004 The grade for

Principles of Bioinorganic Chemistry - 2004 The grade for this course will be determined by a term exam (35%), a written research paper with oral presentation (55%), and problem sets (10%). The oral presentations will be held in research conference style at an all-day symposium at MIT on Saturday, October 30 th. Please reserve the date for there are no excused absences. Papers are due October 28 th. WEB SITE: web. mit. edu/5. 062/www/

Principles of Bioinorganic Chemistry Two Main Avenues of Study • Understand the roles of naturally occurring inorganic elements in biology. By weight, > 50% of living matter is inorganic. Metal ions at the core of biomolecules control many key life processes. • Use metals as probes and drugs Examples: Cisplatin, auranofin as pharmaceuticals Cardiolyte (99 m. Tc) and Gd, imaging agents Mo. S 42 -, Wilson’s disease; cancer? ?

Respiration - Three O 2 Carriers in Biology deoxy. Hb, Mb deoxy. Hc deoxy. Hr oxy. Hb, Mb oxy. Hc oxy. Hr

The Heme Group; the Defining E� xample of a Bioinorganic Chip Peripheral carboxylates and axial ligands matter!
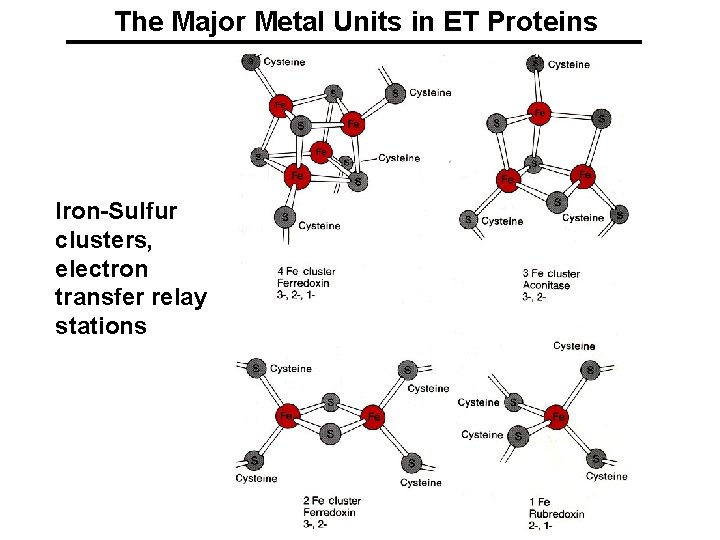
The Major Metal Units in ET Proteins Iron-Sulfur clusters, electron transfer relay stations
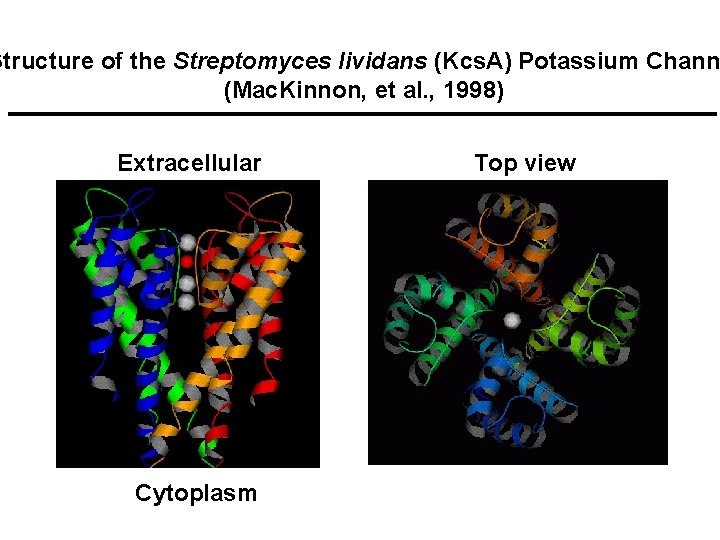
Structure of the Streptomyces lividans (Kcs. A) Potassium Chann (Mac. Kinnon, et al. , 1998) Extracellular Cytoplasm Top view

Cobalamin structures

Three Inorganic Compounds Used in Modern Medicine

Course Organization • What metals? How taken up? How assemble? • How do cells regulate metal ion concentrations? Homeostasis • How do metal ions fold biopolymers? • How is the correct metal ion inserted into its site? • What physical methods are used and how do they work? • Electron transfer metalloproteins. • Substrate binding and activation, non-redox. • Bioorganometallic chemistry is now established. • Atom and group transfer (mainly oxygen chemistry). • Protein tuning of active sites.

Choice, Uptake and Assembly of Metal Ions in Cells PRINCIPLES: • Relatively abundant metal ions used (geosphere/biosphere) • Labile metals used (nature works at a kilohertz)� • Low abundance metals concentrated by ATP driven processe • Entry to the cell controlled by specific channels and pumps • Co-factors employed: bioinorganic chips (porphyrins) • Self-assembling units form - from geosphere • Metallochaperones assure that metal ions find their proteins ILLUSTRATIONS: • The selectivity filter of the potassium channel • Uptake of iron

Relative abundance of metal ions in the earth’s crust and seawater Kinetics of H 2 O exchange: Ø 108 sec-1, labile Ø 10 -3 -10 -6 sec-1, inert

Iron Uptake in the Cell The Challenge: • Iron is the second most abundant metal after aluminum • Its Fe(II) and Fe(III) redox states render it functionally useful • At p. H 7, iron is insoluble (10 -18 M) • The challenge: How to mobilize iron in the biosphere? The Solutions: In bacteria, siderophores In humans, transferrin

Synthesis and Structure of Dinuclear Ferric Citrate Complexes “It will be interesting to determine whether solutions of 1 or 2 are taken up by living cells. ” Shweky et. al. Inorg. Chem. 1994, 33, 51615162.
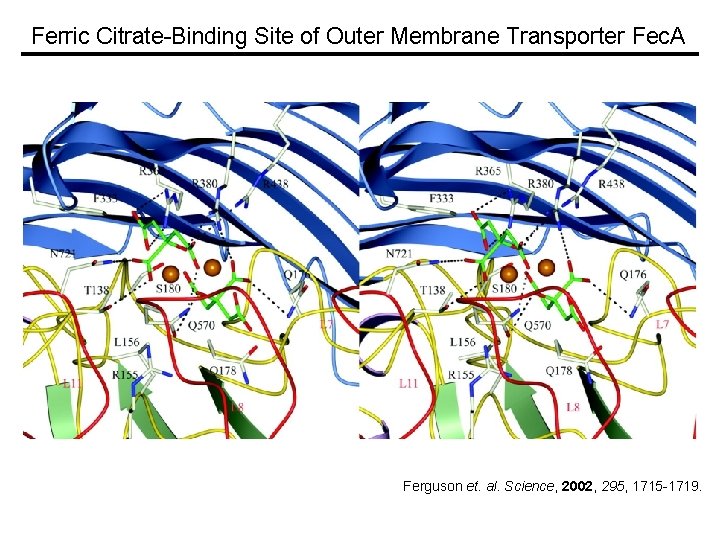
Ferric Citrate-Binding Site of Outer Membrane Transporter Fec. A Ferguson et. al. Science, 2002, 295, 1715 -1719.

Diiron Core of the Outer Membrane Transporter Fec. A 1. 98 Å 2. 00 Å Fe 2. 01 Å 2. 02 Å Fe 2. 05 Å 2. 00 Å 2. 01 Å 1. 96 Å

Enterobactin: a Bacterial Siderophore

Enterobactin, a Cyclic Triserine Lactone A specific cell membrane receptor exists for ferric enterobactin. Release in the cell can occur by hydrolysis of the lactone, reduction to Fe(II), and/or lowering the p. H.

Structure of Vanadium(IV) Enterobactin
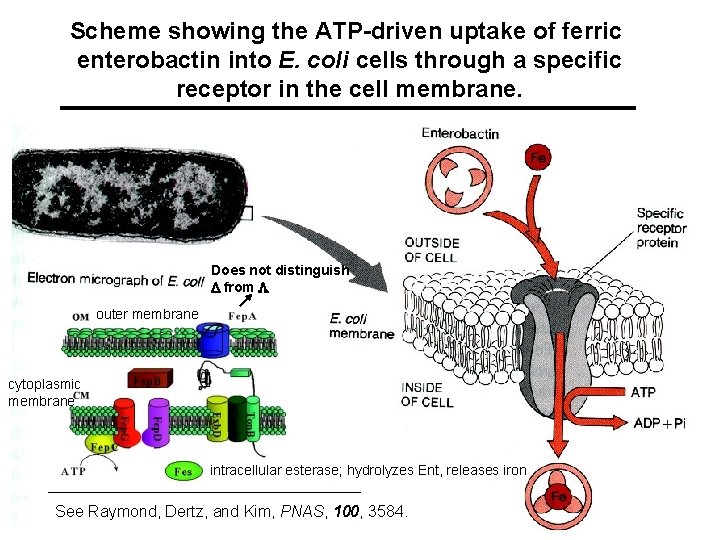
Scheme showing the ATP-driven uptake of ferric enterobactin into E. coli cells through a specific receptor in the cell membrane. Does not distinguish D from L outer membrane cytoplasmic membrane intracellular esterase; hydrolyzes Ent, releases iron See Raymond, Dertz, and Kim, PNAS, 100, 3584.
- Slides: 20