Principles of Bioinorganic Chemistry 2004 Recitations are held

Principles of Bioinorganic Chemistry - 2004 Recitations are held on Mondays at 5 PM, or a little later on seminar days, in 18 -475

Metallochaperones; Metal Folding PRINCIPLES: • Metallochaperones guide and protect metals to natural sites • Chaperone and target receptor protein structurally homologou ILLUSTRATION: • Copper insertion into metalloenzymes Useful references: • Copper Delivery by Metallochaperone Proteins. A. C. Rosenzweig, Acc. Chem. Res. , 2001, 34, 119 -128. • Perspectives in Inorganic Structural Genomics: A Trafficking Route for Copper. F. Arnesano, L. Banci, I. Bertini, and S. Ciofi-Baffoni, Eur. J. Inorg. Chem. , 2004, 1583 -1593.
![Copper Uptake and Transport in Cells The puzzles: The total cellular [Cu] in yeast Copper Uptake and Transport in Cells The puzzles: The total cellular [Cu] in yeast](http://slidetodoc.com/presentation_image_h/14ccb984221bc16b3b6172b1ad09eb25/image-3.jpg)
Copper Uptake and Transport in Cells The puzzles: The total cellular [Cu] in yeast is 0. 07 m. M, none free. How does copper find its way into metalloproteins? The implications: Mn, Fe, Zn have similar systems; understanding one in detail has implications for all
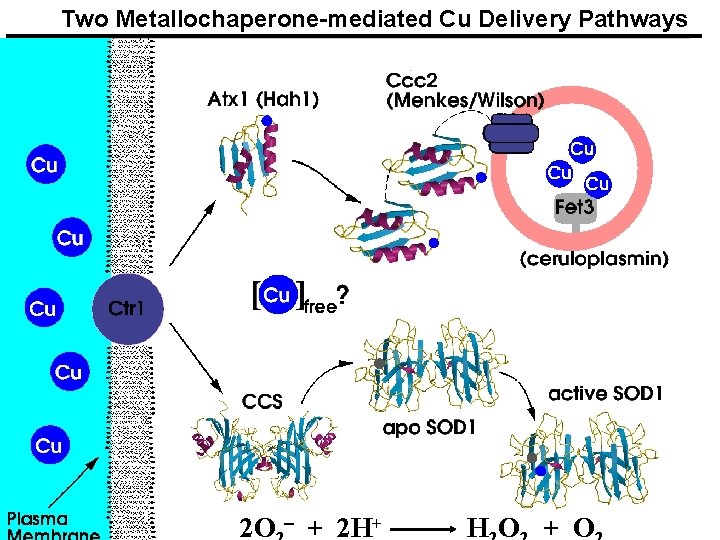
Two Metallochaperone-mediated Cu Delivery Pathways 2 O - + 2 H+ HO + O

Two well characterized pathways Atx 1 delivers Cu to transport ATPases in the secretory pathway, which translocates it into vesicles for insertion into multicopper oxidases such as ceruloplasmin Mutations in human forms of these ATPases lead to Menkes and Wilson diseases CCS delivers copper to Cu, Zn SOD Human Cu/Zn SOD is linked to ALS

Copper Uptake and Transport in Cells The players: SOD, superoxide dismutase, a copper enzyme, a dimer containing two His-bridged Cu/Zn sites CCS, a copper chaperone for superoxide dismutase Ctr, family of membrane proteins that transport copper across the plasma membrane, delivering it to at least three chaperones: CCS, Cox 17, Atx 1 N-terminus has 8 putative Cu motifs (MXMXXM) C-terminus has 2 CXC motifs Atx 1, the copper chaperone for Ccc 2, a cation transporting ATPase; has CXXC sites Fet 3, a multicopper ferroxidase Note the connection between Fe and Cu trafficking

Key Questions Address by Structural Bioinorganic Chemistry (Rosenzweig, O’Halloran, Culotta) ¨What are the details of copper binding by these proteins, including stoichiometry and coordination geometry? ¨How do these chaperones interact with their copper receptor proteins? ¨What features of the copper binding and protein-protein interactions render each chaperone specific for its target protein?
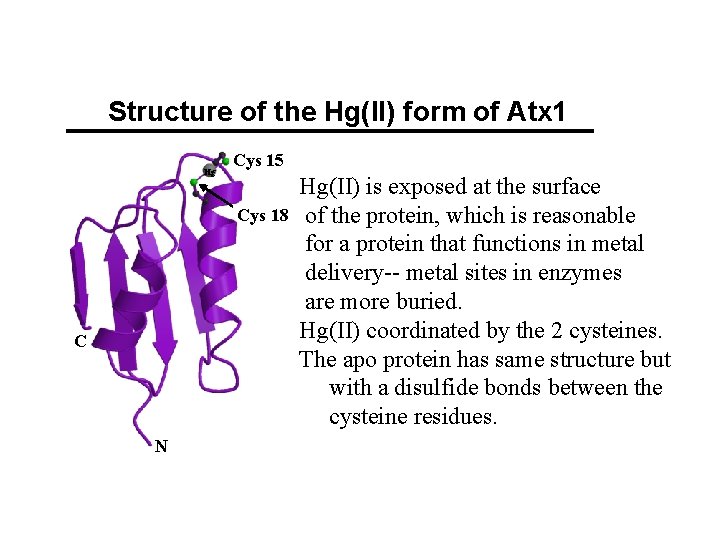
Structure of the Hg(II) form of Atx 1 Hg Cys 15 Cys 18 C N Hg(II) is exposed at the surface of the protein, which is reasonable for a protein that functions in metal delivery-- metal sites in enzymes are more buried. Hg(II) coordinated by the 2 cysteines. The apo protein has same structure but with a disulfide bonds between the cysteine residues.
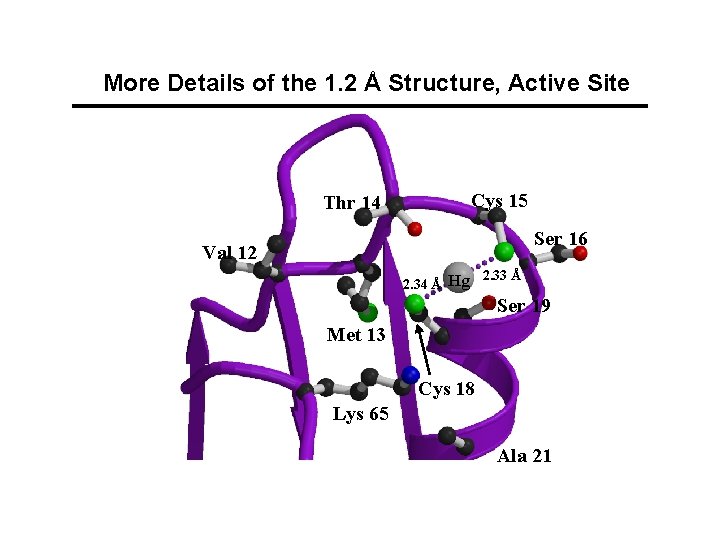
More Details of the 1. 2 Å Structure, Active Site Cys 15 Thr 14 Ser 16 Val 12 2. 34 Å Hg 2. 33 Å Ser 19 Met 13 Cys 18 Lys 65 Ala 21
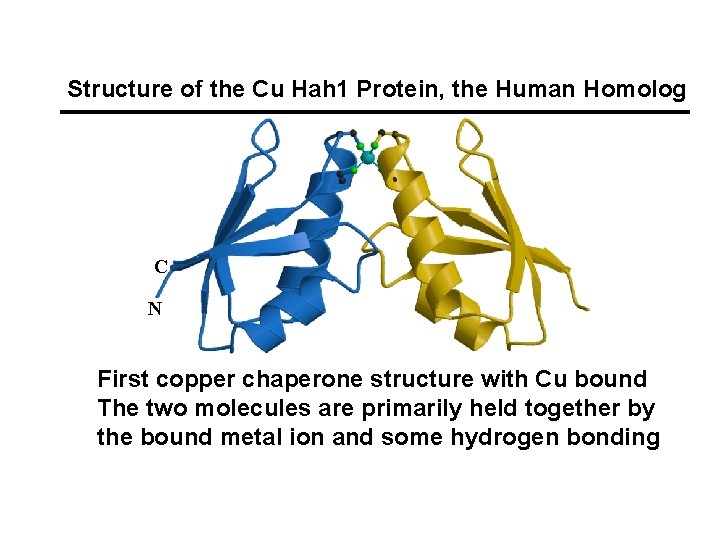
Structure of the Cu Hah 1 Protein, the Human Homolog C N First copper chaperone structure with Cu bound The two molecules are primarily held together by the bound metal ion and some hydrogen bonding

Extended H-Bonding Interactions Stabilize the Structure T 11 B is conserved in most related domains. When it is not there it is replaced by His, which could serve the same function.
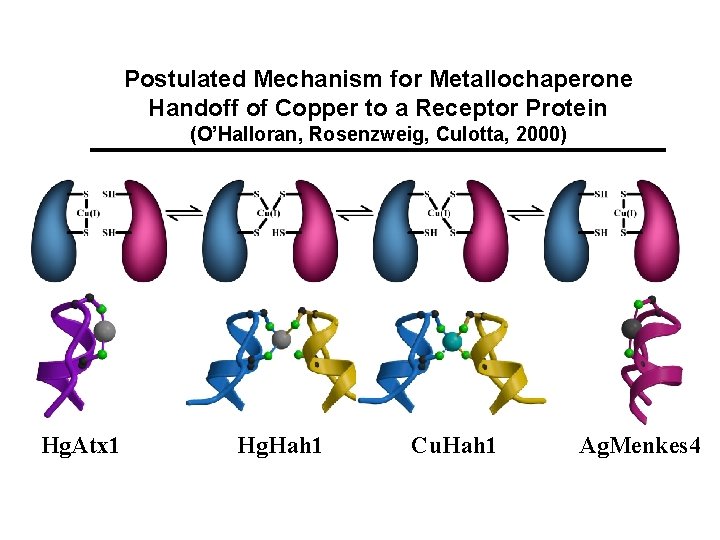
Postulated Mechanism for Metallochaperone Handoff of Copper to a Receptor Protein (O’Halloran, Rosenzweig, Culotta, 2000) Hg. Atx 1 Hg. Hah 1 Cu. Hah 1 Ag. Menkes 4
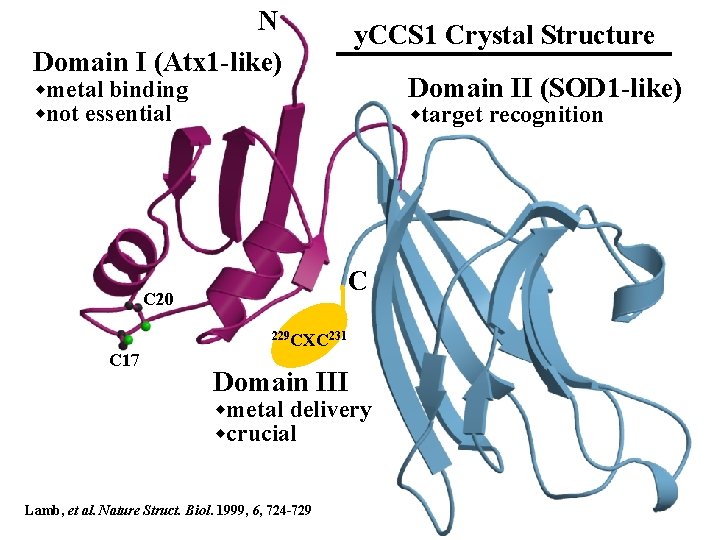
N Domain I (Atx 1 -like) y. CCS 1 Crystal Structure Domain II (SOD 1 -like) wmetal binding wnot essential wtarget recognition C C 20 229 CXC 231 C 17 Domain III wmetal delivery wcrucial Lamb, et al. Nature Struct. Biol. 1999, 6, 724 -729
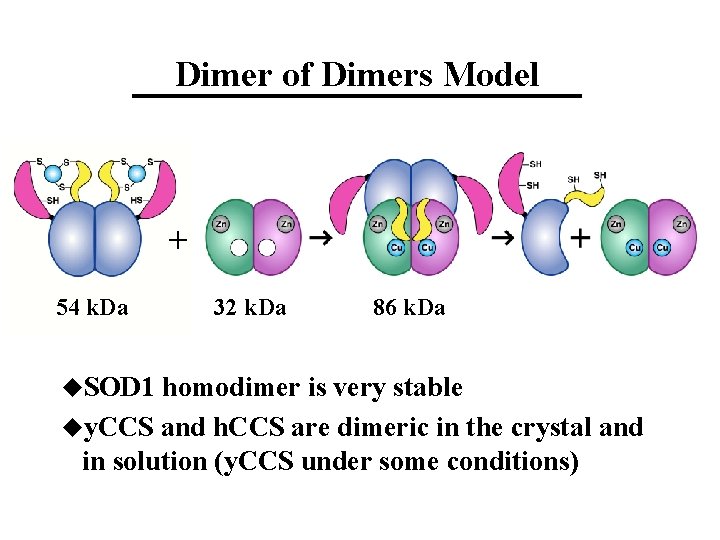
Dimer of Dimers Model + 54 k. Da u. SOD 1 32 k. Da 86 k. Da homodimer is very stable uy. CCS and h. CCS are dimeric in the crystal and in solution (y. CCS under some conditions)
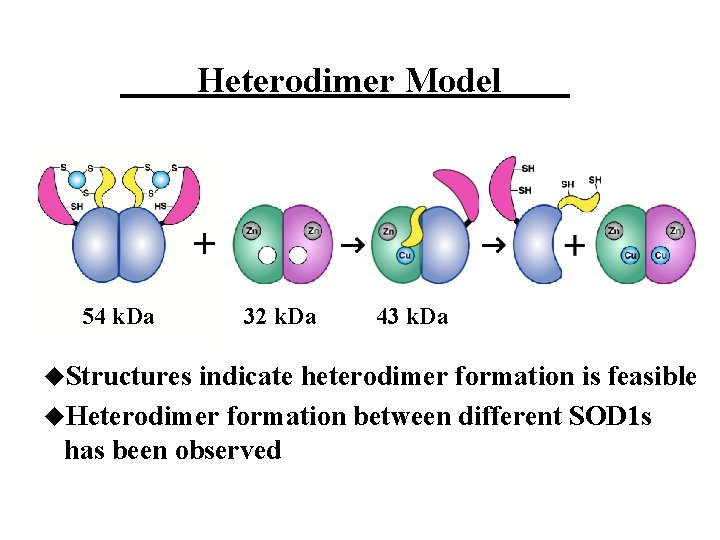
Heterodimer Model + 54 k. Da u. Structures 32 k. Da 43 k. Da indicate heterodimer formation is feasible u. Heterodimer formation between different SOD 1 s has been observed

Biophysical and biochemical studies of complex formation u. According to gel filtration chromatography, dynamic light scattering, analytical ultracentrifugation, and chemical crosslinking experiments, y. CCS and SOD 1 form a specific protein-protein complex u. The molecular weight of the complex, ~43 k. Da, is most consistent with a heterodimer u. Higher order complexes, such as a dimer of dimers, were not detected 86 k. Da Lamb, et al. Biochem. 2000, 39, 14720 -14727 43 k. Da

Factors Affecting Heterodimer Formation u. The heterodimeric complex formed with a mutant of SOD 1 that cannot bind copper, H 48 F-SOD 1, is more stable u. Heterodimer formation is facilitated by zinc u. Heterodimer formation is apparently independent of whether copper is bound to y. CCS u. Heterodimer formation between Cu-y. CCS and wt. SOD 1 in the presence of zinc is accompanied by SOD 1 activation u. These data suggest that in vivo copper loading occurs via a heterodimeric intermediate Lamb, et al. Biochem. 2000, 39, 14720 -14727

Crystals of the y. CCS/H 48 F-SOD 1 heterodimeric complex P 3221 a = b = 104. 1 Å, c = 233. 7 Å Solved by molecular replacement Lamb, et al. , Nature Structural Biology (2001), 8(9), 751 -755.
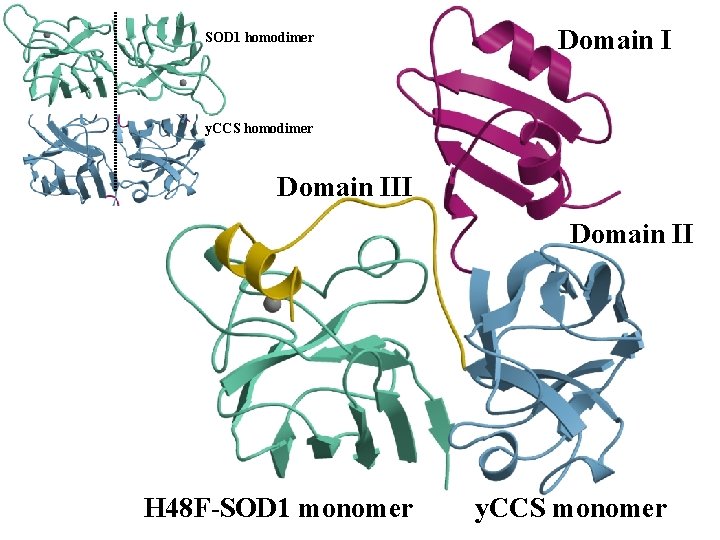
SOD 1 homodimer Domain I y. CCS homodimer Domain III Domain II H 48 F-SOD 1 monomer y. CCS monomer
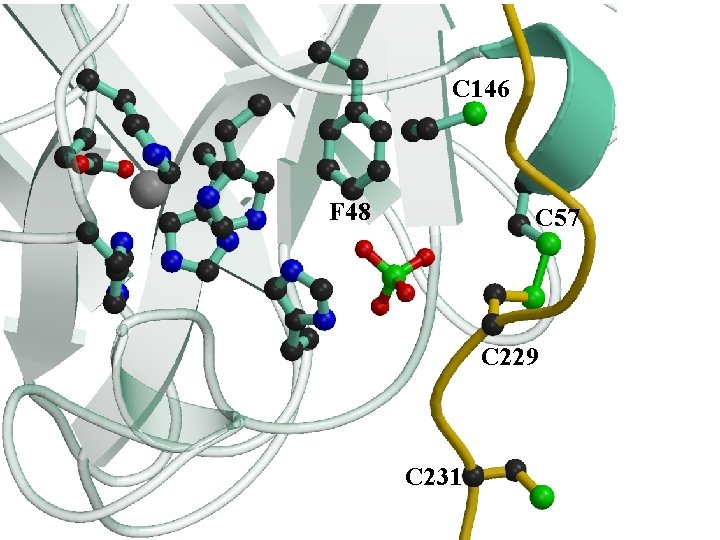
C 146 F 48 C 57 C 229 C 231
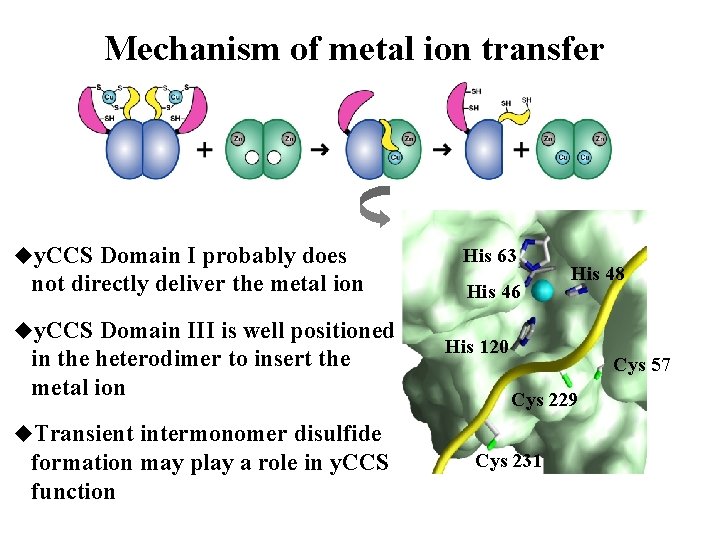
Mechanism of metal ion transfer uy. CCS Domain I probably does not directly deliver the metal ion uy. CCS Domain III is well positioned in the heterodimer to insert the metal ion His 63 His 46 His 120 Cys 57 Cys 229 u. Transient intermonomer disulfide formation may play a role in y. CCS function His 48 Cys 231

Metal Folding of Biopolymers PRINCIPLES: • Metal ions organize the structures of biopolymers • In binding proteins, metal ions typically shed water molecules • In binding nucleic acids, aqua ligands remain for H-bonding • Metal-mediated biopolymer folding facilitates interactions • Cross-link formation underlies metallodrug action • High coordination numbers are used for function ILLUSTRATIONS: • Zinc finger proteins control transcription • Ca 2+, a second messenger and sentinel at the synapse • Cisplatin, an anticancer drug
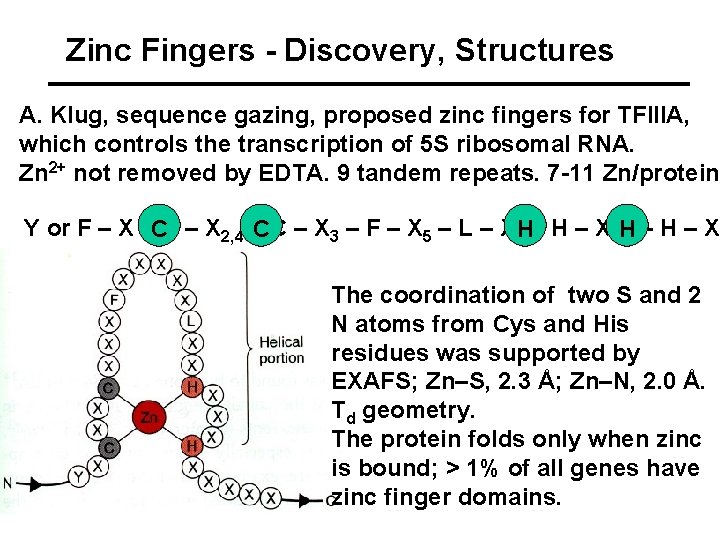
Zinc Fingers - Discovery, Structures A. Klug, sequence gazing, proposed zinc fingers for TFIIIA, which controls the transcription of 5 S ribosomal RNA. Zn 2+ not removed by EDTA. 9 tandem repeats. 7 -11 Zn/protein. Y or F – X –CC – X 2, 4 –CC – X 3 – F – X 5 – L – X 2 H– H – X 3, 4 H– H – X The coordination of two S and 2 N atoms from Cys and His residues was supported by EXAFS; Zn–S, 2. 3 Å; Zn–N, 2. 0 Å. Td geometry. The protein folds only when zinc is bound; > 1% of all genes have zinc finger domains.
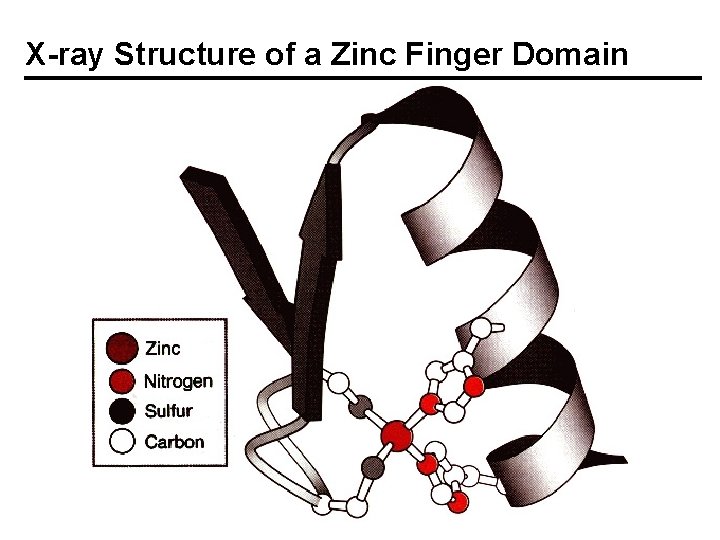
X-ray Structure of a Zinc Finger Domain
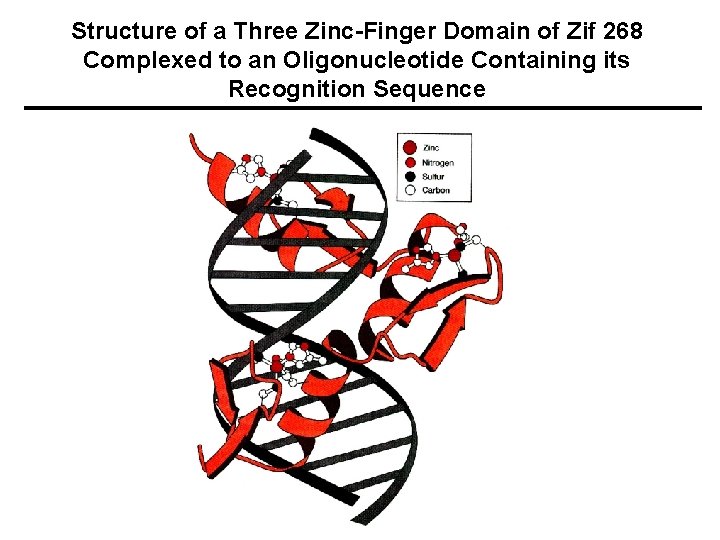
Structure of a Three Zinc-Finger Domain of Zif 268 Complexed to an Oligonucleotide Containing its Recognition Sequence

The Specificity of Zinc for Zinc-finger Domains Kd value: Metal ion: 2 p. M 5 n. M 2 m. M 3 m. M Zn 2+ Co 2+ Ni 2+ Fe 3+
- Slides: 26