Principles of Bioinorganic Chemistry 2004 Note The course

Principles of Bioinorganic Chemistry - 2004 Note: The course seminar presentations will be held on Sunday, October 31, 2004 in the Bush Room. Please remember that daylight savings time ends that day.

Dioxygen Carriers: Hb, Mb, Hc, Hr Examples of Atom- and Group-Transfer Chemistry PRINCIPLES: • Both substrate binding and redox changes occur • Coupled proton-electron transfer steps set the redox potentials • Closely positioned redox/acid-base units work in concert • Interactions with substrates/other proteins gate electron transf • Two-electron transfer strategies include 2 metals, M-porphyrin • Metal centers used to create or destroy radical species • Changes in metal coordination spheres can facilitate allostery • Bioinorganic chemistry of dioxygen paramount example ILLUSTRATIONS: • O 2 Binding and Transport: hemoglobin (Hb), myoglobin (Mb), hemocyanin (Hc), and hemerythrin (Hr) • O 2 Activation: cytochrome P-450, tyrosinase, methane monooxygenase; dioxygenases

Properties of Protein Dioxygen Carriers
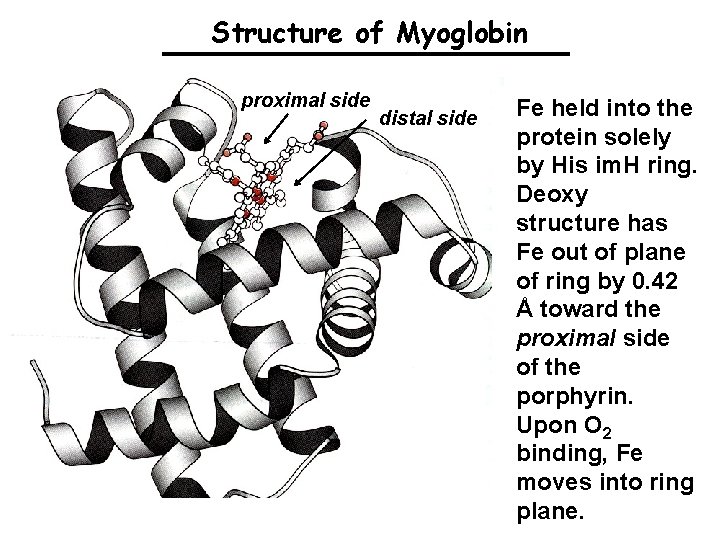
Structure of Myoglobin proximal side distal side Fe held into the protein solely by His im. H ring. Deoxy structure has Fe out of plane of ring by 0. 42 Å toward the proximal side of the porphyrin. Upon O 2 binding, Fe moves into ring plane.
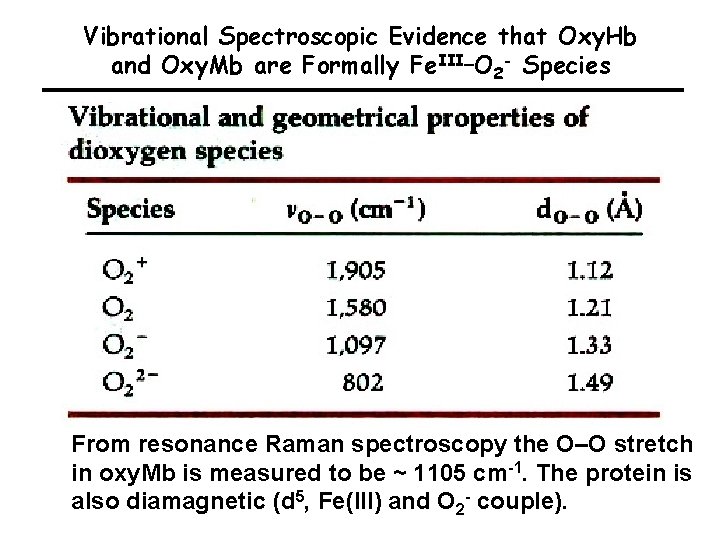
Vibrational Spectroscopic Evidence that Oxy. Hb and Oxy. Mb are Formally Fe. III–O 2 - Species From resonance Raman spectroscopy the O–O stretch in oxy. Mb is measured to be ~ 1105 cm-1. The protein is also diamagnetic (d 5, Fe(III) and O 2 - couple).
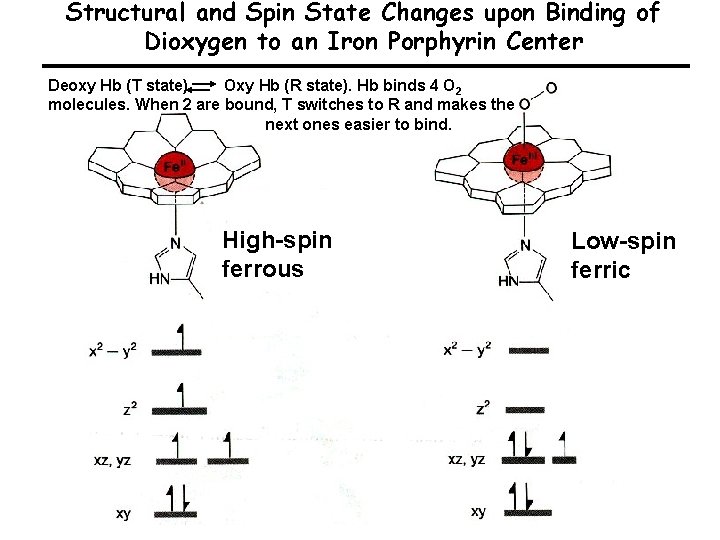
Structural and Spin State Changes upon Binding of Dioxygen to an Iron Porphyrin Center Deoxy Hb (T state) Oxy Hb (R state). Hb binds 4 O 2 molecules. When 2 are bound, T switches to R and makes the next ones easier to bind. High-spin ferrous Low-spin ferric

Model Chemistry for Oxy Hb and Oxy Mb The problem: Fe. IIP + O 2 IIP. . Fe Fe. IIIP–O 2 - PFe. III–O . . Fe. IIP 2 PFe. IV=O: PFe. III–O–Fe. IIIP m-oxo, “dimer” ferryl . . O–Fe. IIIP The solutions: Attach the porphyrin to a solid support to avoid the bimolecular reaction; or, use low T, non-aqueous solvents, and py or 1 -Me. Im complexes, but stability is lost at - 45 °C or above. The best solution was the construction of a sterically hindered cavity for dioxygen binding to avoid the intemolecular chemistry leading to thermodynamic sink of the system, the (moxo)diiron(III) species.
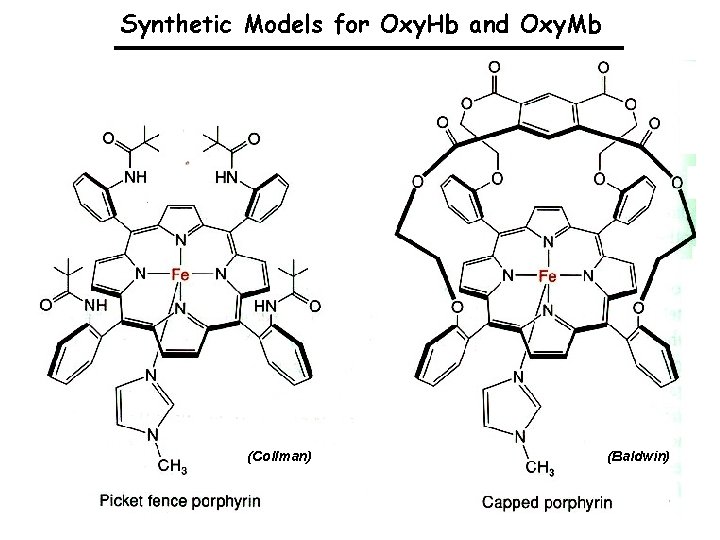
Synthetic Models for Oxy. Hb and Oxy. Mb (Collman) (Baldwin)
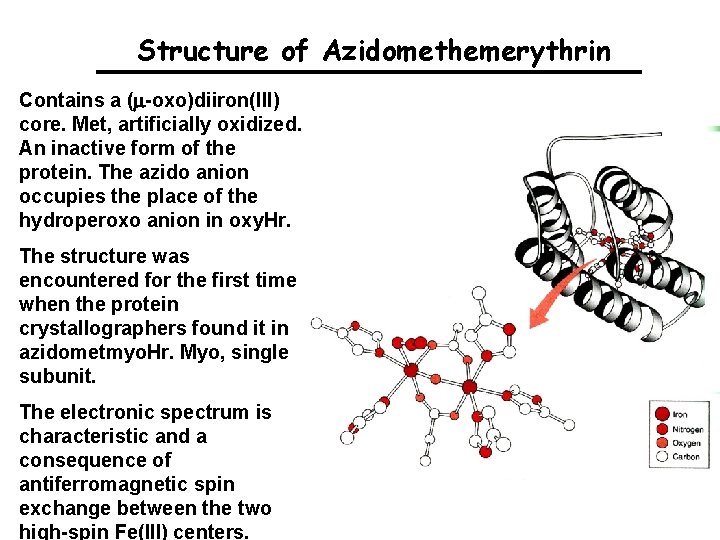
Structure of Azidomethemerythrin Contains a (m-oxo)diiron(III) core. Met, artificially oxidized. An inactive form of the protein. The azido anion occupies the place of the hydroperoxo anion in oxy. Hr. The structure was encountered for the first time when the protein crystallographers found it in azidometmyo. Hr. Myo, single subunit. The electronic spectrum is characteristic and a consequence of antiferromagnetic spin exchange between the two high-spin Fe(III) centers.

Hemerythrins - Diiron Dioxygen Carriers Properties: Mono- (myo Hr) and multi- (Hr) subunit proteins. Found in marine invertebrates. Easily isolated protein; crystallizes after one step!! Deoxy Hr, colorless, diiron(II) Oxy Hr, red, diiron(III) peroxo n. O–O, 844 cm-1 in the terminally bound peroxide region. n. Fe–O–Fe, 486 cm-1, resonance enhanced symmetric stretch. The asymmetric stretch occurs at 757 cm-1. Mixed-valent, semimet Hr, Fe(II)Fe(III): inactive.

Note proton-coupled electron transfer Evidence for proton transfer comes from resonance Raman work

Early Structural Models for Methemerythrin These and related complexes have no site for binding of azide or dioxygen related species such as hydroperoxide. The syntheses exemplify spontaneous self-assembly. The challenges are to make a site available, allow redox chemistry to occur, and avoid polymerization to rust or molecular ferric wheels and related complexes.

Early Structural Models for Deoxyhemerythrin None does the chemistry of the protein!
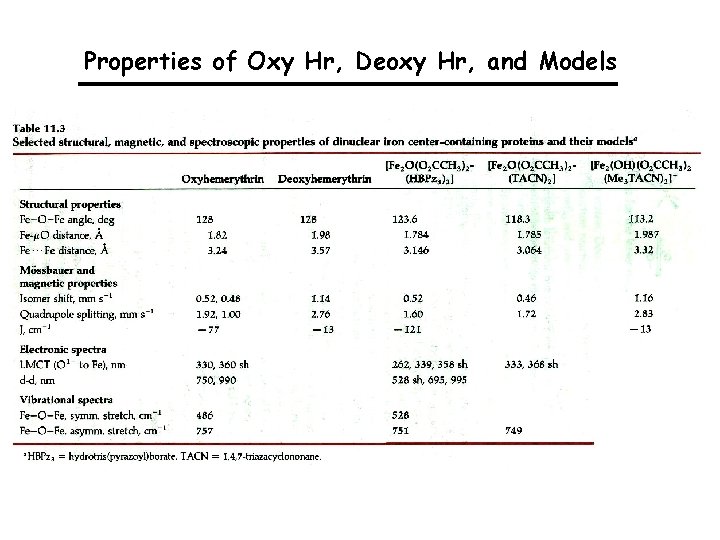
Properties of Oxy Hr, Deoxy Hr, and Models


Dioxygen Binding Chemistry of the Hr Model Complex Product matches protein Mössbauer, resonance Raman, UV-vis spectra

Hemocyanins - Dicopper Dioxygen Carriers Properties: Multi-subunit proteins, ranging in size up to 460 k. Da. Found in spiny lobsters, crayfish, and arachnids. Deoxy Hc, colorless, dicopper(I) Oxy Hc, blue, dicopper(II) peroxide n. O–O, 745 -750 cm-1 in the peroxide region, but low. Unusual structure, first established by model chemistry: O Cu Cu O
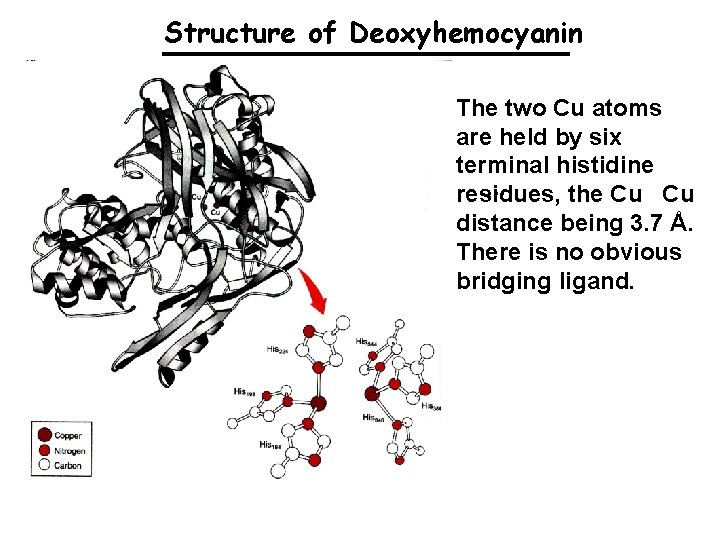
Structure of Deoxyhemocyanin The two Cu atoms are held by six terminal histidine. . . residues, the Cu Cu distance being 3. 7 Å. There is no obvious bridging ligand.
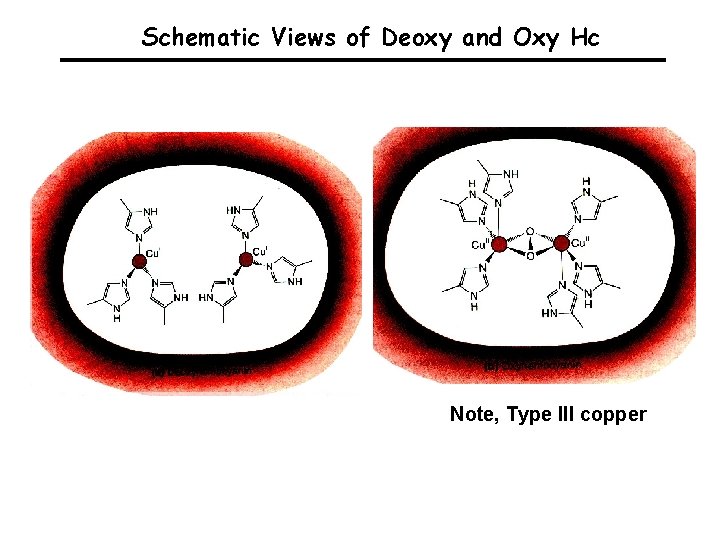
Schematic Views of Deoxy and Oxy Hc Note, Type III copper
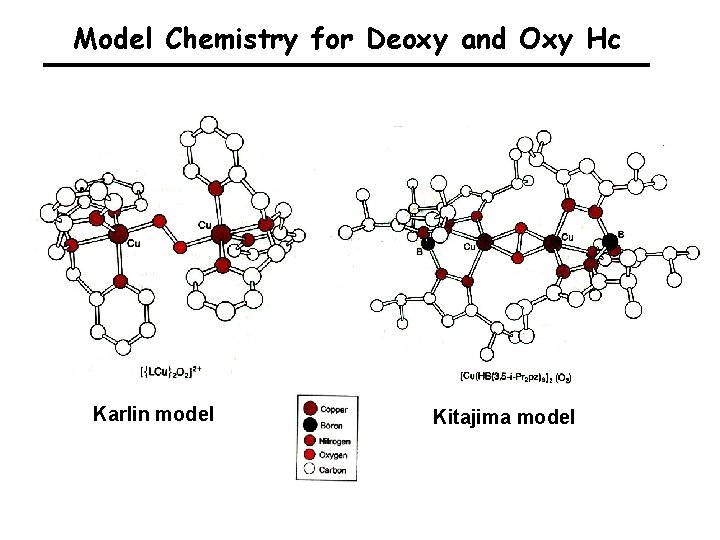
Model Chemistry for Deoxy and Oxy Hc Karlin model Kitajima model

Important Relationships Reversible O 2 binding • Iron porphyrin, Hb/Mb O 2 Activation Iron porphyrin, P-450 • Dicopper center, Hc tyrosinase Dicopper center, • Diiron center, Hr Diiron center, R 2, MMO WHAT CONTROLS THE FUNCTION? ?
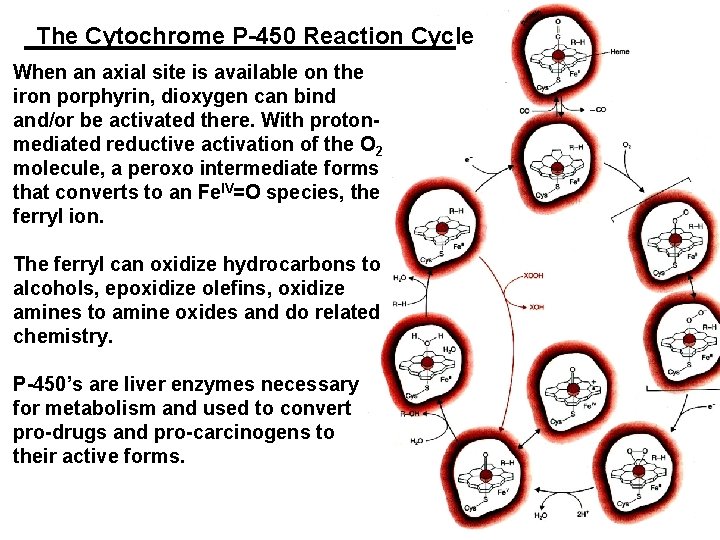
The Cytochrome P-450 Reaction Cycle When an axial site is available on the iron porphyrin, dioxygen can bind and/or be activated there. With protonmediated reductive activation of the O 2 molecule, a peroxo intermediate forms that converts to an Fe. IV=O species, the ferryl ion. The ferryl can oxidize hydrocarbons to alcohols, epoxidize olefins, oxidize amines to amine oxides and do related chemistry. P-450’s are liver enzymes necessary for metabolism and used to convert pro-drugs and pro-carcinogens to their active forms.

Principles Illustrated by these Cases Substrate binding and redox changes occur: In all three cases, O 2 binding is accompanied by electron transfer from one or two metal ions to dioxygen. Coupled proton-electron transfer steps set the potentials: In oxy. Hr a proton transfers from the bridging hydroxide to the peroxo ligand; this step appears to block further conversion to high-valent iron oxidase center(s). Metal center used to create or destroy radical species: Occurs in ribonucleotide reductase R 2 protein. Changes in metal coordination sphere facilitate allostery: Explains the cooperativity of O 2 binding in Hb.

The Mineral Springs in Bath, England, Source of Methylococcus capsulatus (Ba The Restitutive Contents of the WATER’s Concoctive Power: Solution of gaffes, chaos of Salts and mineral effluvia of subterranean expiration. It cleanses the body from all blotches, scurvical itchings and BREAKING OUTS WHATSOEVER!

Methanotrophs are Used in Bioremediation Prince William Sound, Alaska: After the Exxon Valdez oil spill, fertilizers were spread on the beaches and natural methanotrophs restored their pristine beauty. Plants recruit oil-detoxifying microbes, as discovered by scientists analyzing the recovery of the environment in the Persian Gulf region following the 1991 Gulf War. " In the root zone was a rich reservoir of well-known oil eating microbes. . . one family of which (Arthrobacter) accounted for fully 95 percent. . . " Science News, 148, 84 (August 5, 1995)
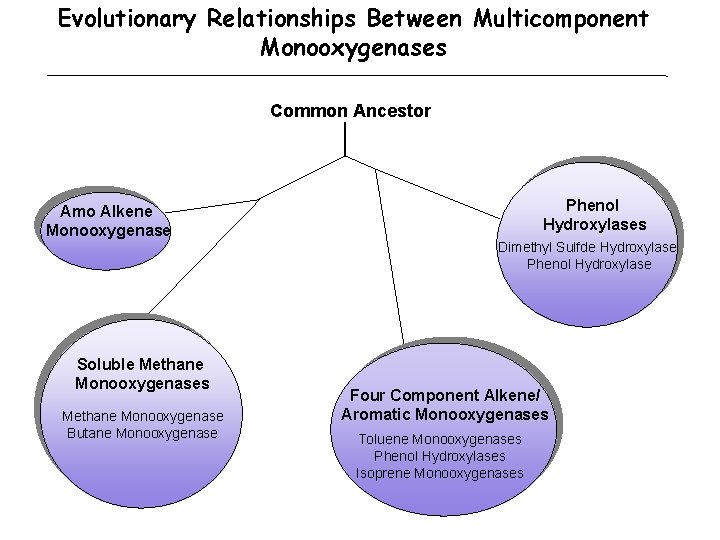
Evolutionary Relationships Between Multicomponent Monooxygenases Common Ancestor Phenol Hydroxylases Amo Alkene Monooxygenase Dimethyl Sulfde Hydroxylase Phenol Hydroxylase Soluble Methane Monooxygenases Methane Monooxygenase Butane Monooxygenase Four Component Alkene/ Aromatic Monooxygenases Toluene Monooxygenases Phenol Hydroxylases Isoprene Monooxygenases

Properties of Hydrocarbon Monooxygenases Containing Carboxylate-Bridged Diiron Centers Leahy, Batchelor, Morcomb, FEMS Microbiol. Rev. 2003, 770, 1 -31.

Properties of Methanotrophs p. MMO, Cu s. MMO, Fe • 5 -50 Tg CH 4/year consumed by soil methanotrophs (1 -10% of atmospheric CH 4), converting this greenhouse gas to biomass. • 104 kcal/mol BDE for methane makes it a challenge to activate. • Controlled oxidation to methanol at moderate temperatures in neutral aqueous solution is a remarkable chemical feat. • 500 billion barrels crude oil equivalent in recoverable but remote natural gas deposits might be made available.

Carboxylate-Bridged Diiron Proteins Global Research Goals • • What tunes the properties of the diiron centers? What are the electron transport pathways? What factors control dioxygen reactivity? How is substrate specificity achieved? Objectives for the s. MMO and Model Studies • • Determine structures of all components and complexes Understand hydroxylation and epoxidation reactions Synthesize and characterize structural/spectroscopic mode Achieve selective oxidation and catalysis

Soluble Methane Monooxygenase (s. MMO) Component Structures & Reaction Cycle
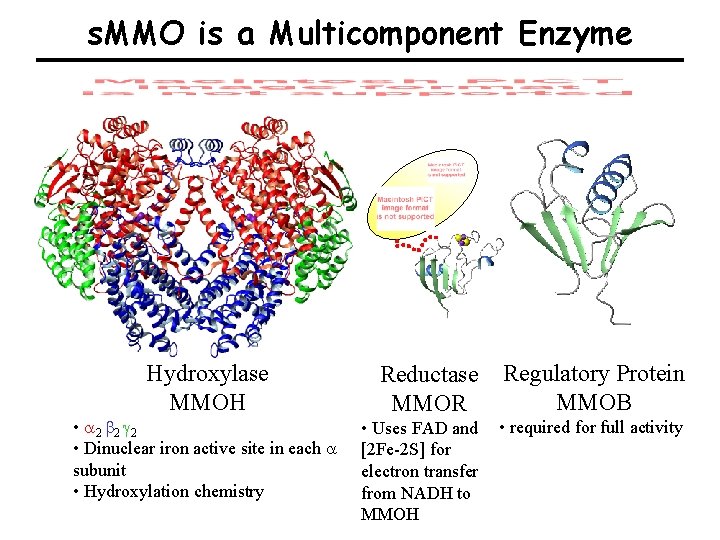
s. MMO is a Multicomponent Enzyme Hydroxylase MMOH • a 2 b 2 g 2 • Dinuclear iron active site in each a subunit • Hydroxylation chemistry Reductase MMOR Regulatory Protein MMOB • Uses FAD and [2 Fe-2 S] for electron transfer from NADH to MMOH • required for full activity
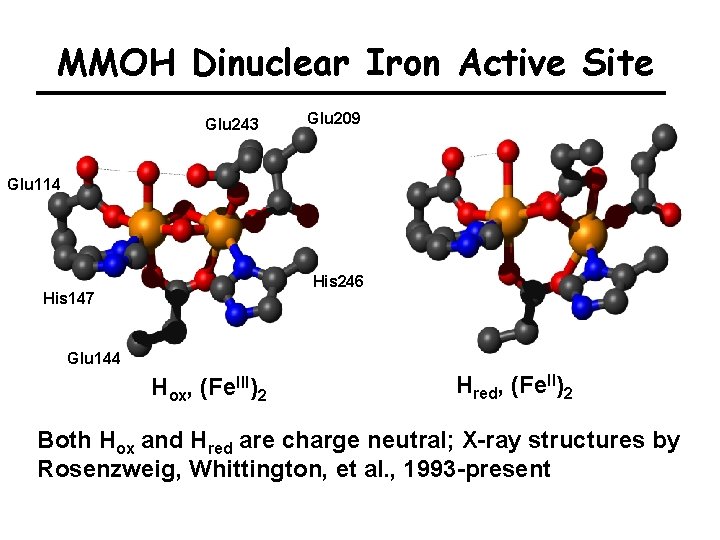
MMOH Dinuclear Iron Active Site Glu 243 Glu 209 Glu 114 His 246 His 147 Glu 144 Hox, (Fe. III)2 Hred, (Fe. II)2 Both Hox and Hred are charge neutral; X-ray structures by Rosenzweig, Whittington, et al. , 1993 -present
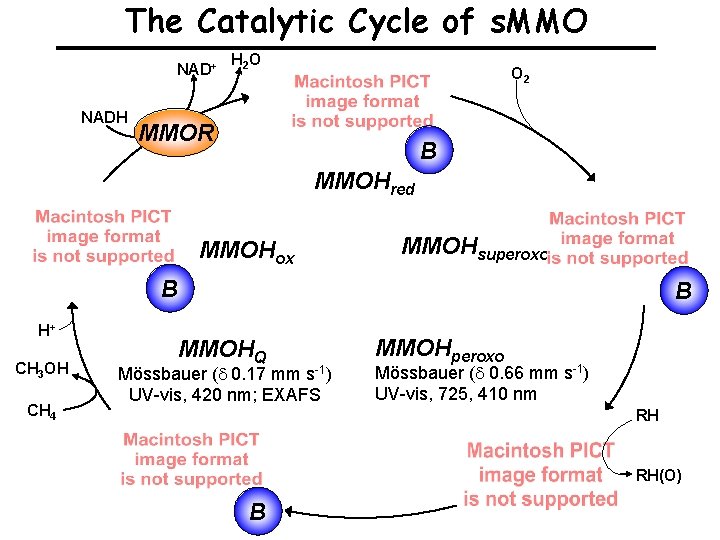
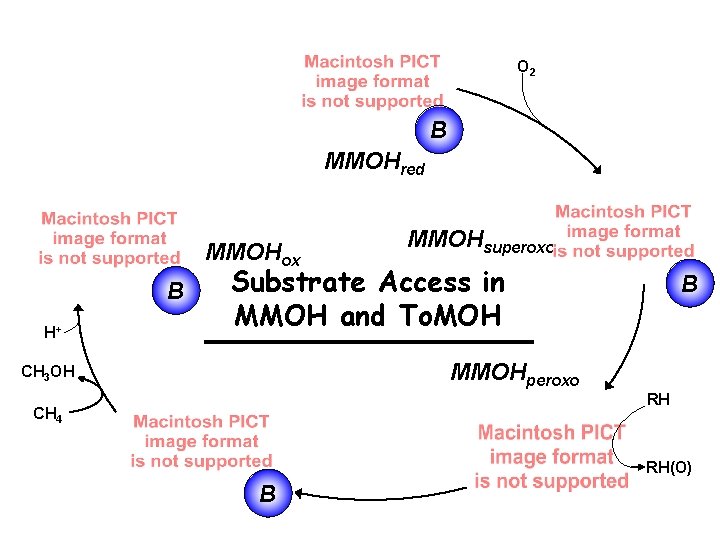
The Catalytic Cycle of s. MMO NAD+ NADH H 2 O O 2 MMOR B MMOHred MMOHox MMOHsuperoxo B H+ CH 3 OH CH 4 B MMOHQ Mössbauer (d 0. 17 mm s-1) UV-vis, 420 nm; EXAFS MMOHperoxo Mössbauer (d 0. 66 mm s-1) UV-vis, 725, 410 nm RH RH(O) B

Reactions of CH 3 X Substrates with Q H+ CH 3 OH CH 4 MMOHQ Mössbauer (d 0. 17 mm s-1) UV-vis, 420 nm; EXAFS B


Reaction of Q with Methane by Double-Mixing SF k. H/k. D = 26 T = 20 ºC [H]red = 16. 8 µM [CH 4] = 0. 50 m. M kobs = 14. 1(1) s-1 CH 4 CD 4
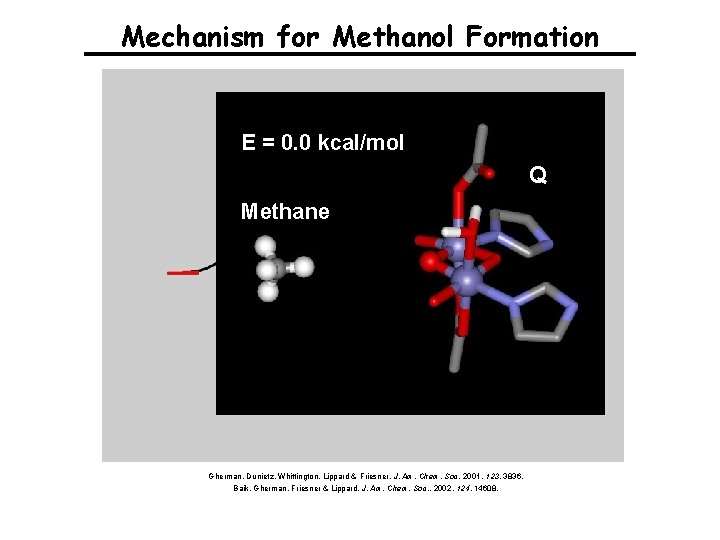
Mechanism for Methanol Formation E = 0. 0 kcal/mol Q Methane Gherman, Dunietz, Whittington, Lippard & Friesner, J. Am. Chem. Soc. 2001, 123, 3836. Baik, Gherman, Friesner & Lippard, J. Am. Chem. Soc. , 2002, 124, 14608.
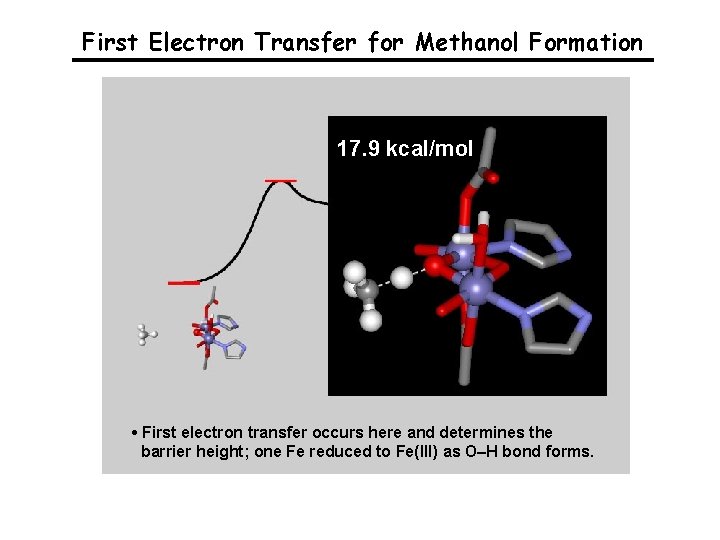
First Electron Transfer for Methanol Formation 17. 9 kcal/mol • First electron transfer occurs here and determines the barrier height; one Fe reduced to Fe(III) as O–H bond forms.
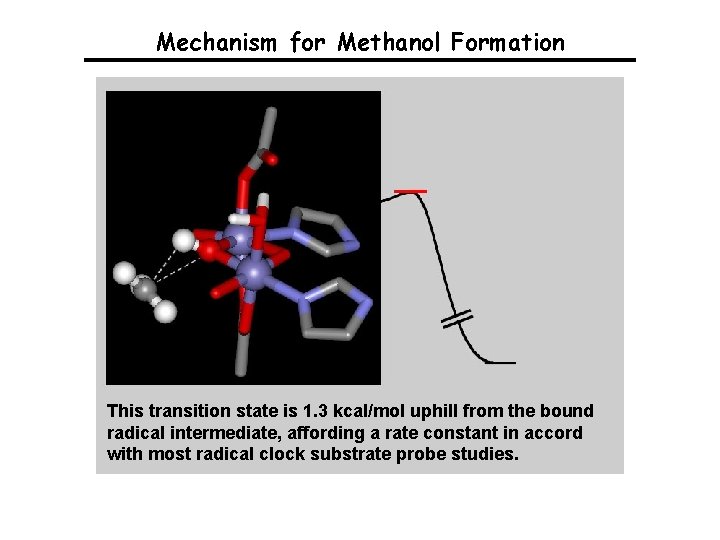
Mechanism for Methanol Formation This transition state is 1. 3 kcal/mol uphill from the bound radical intermediate, affording a rate constant in accord with most radical clock substrate probe studies.
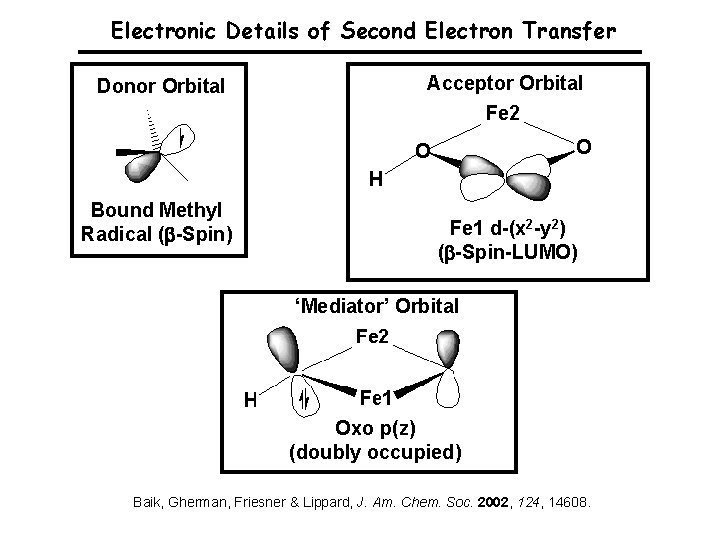
Electronic Details of Second Electron Transfer Donor Orbital Bound Methyl Radical ( -Spin) Acceptor Orbital Fe 1 d-(x 2 -y 2) ( -Spin-LUMO) ‘Mediator’ Orbital Oxo p(z) (doubly occupied) Baik, Gherman, Friesner & Lippard, J. Am. Chem. Soc. 2002, 124, 14608.
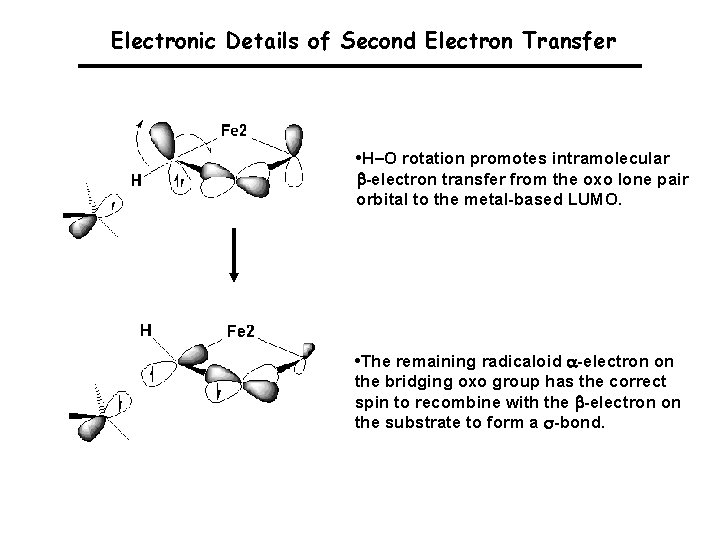
Electronic Details of Second Electron Transfer • H–O rotation promotes intramolecular -electron transfer from the oxo lone pair orbital to the metal-based LUMO. • The remaining radicaloid -electron on the bridging oxo group has the correct spin to recombine with the -electron on the substrate to form a s-bond.
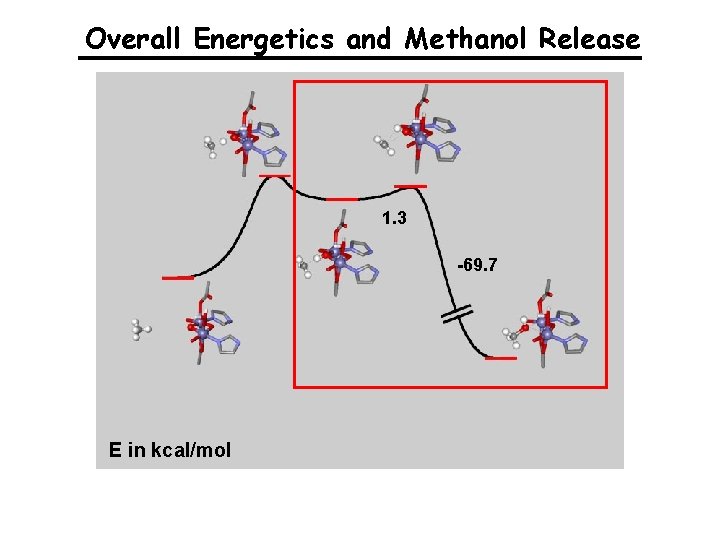
Overall Energetics and Methanol Release 1. 3 -69. 7 E in kcal/mol

Reactions of Q with Substrates Reveal Complexities C 2 H 6 CH 4 C 2 D 6 CD 4 Puzzles: This result indicates that, for ethane, the rate-determining step in not C–H bond activation. Yet kobs is the same! Answers: For CH 4, H atom abstraction is rate determining; for C 2 H 6, binding is rate determining. The bond in C 2 H 6 is weaker, lowering of the C–H bond activation energy by ~5. 6 kcal/mol, from both experiment and theory.

kobs vs Nitromethane Concentration for Q Decay Solid circles, CH 3 NO 2 Open circles CD 3 NO 2 p. H = 7, 20 C ; KIE, 8. 1 Direct evidence for bound substrate in a Q reaction is facilitated by the high solubility of nitromethane. Ambundo, E. A. ; Friesner, R. A. ; Lippard, S. J. J. Am. Chem. Soc. 2002, 124, 8770 -8771.
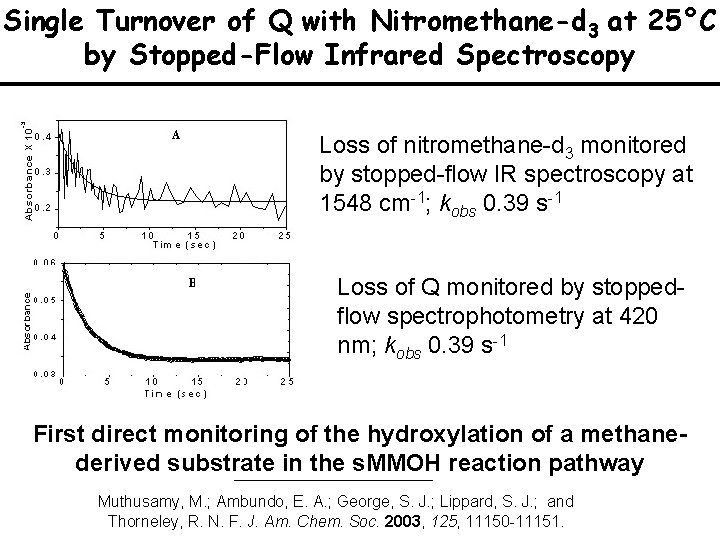
Single Turnover of Q with Nitromethane-d 3 at 25°C by Stopped-Flow Infrared Spectroscopy Loss of nitromethane-d 3 monitored by stopped-flow IR spectroscopy at 1548 cm-1; kobs 0. 39 s-1 Loss of Q monitored by stoppedflow spectrophotometry at 420 nm; kobs 0. 39 s-1 First direct monitoring of the hydroxylation of a methanederived substrate in the s. MMOH reaction pathway Muthusamy, M. ; Ambundo, E. A. ; George, S. J. ; Lippard, S. J. ; and Thorneley, R. N. F. J. Am. Chem. Soc. 2003, 125, 11150 -11151.

KIE for Reactions of Q with CH 3 X Substratesa CLASS I SUBSTRATES H atom abstraction rate-determining: CH 4, CH 3 CN, CH 3 NO 2 CLASS II SUBSTRATES Binding rate-determining: C 2 H 6, CH 3 OH Ambundo, E. A. ; Friesner, R. A. ; Lippard, S. J. J. Am. Chem. Soc. 2002, 124, 8770 -8771.
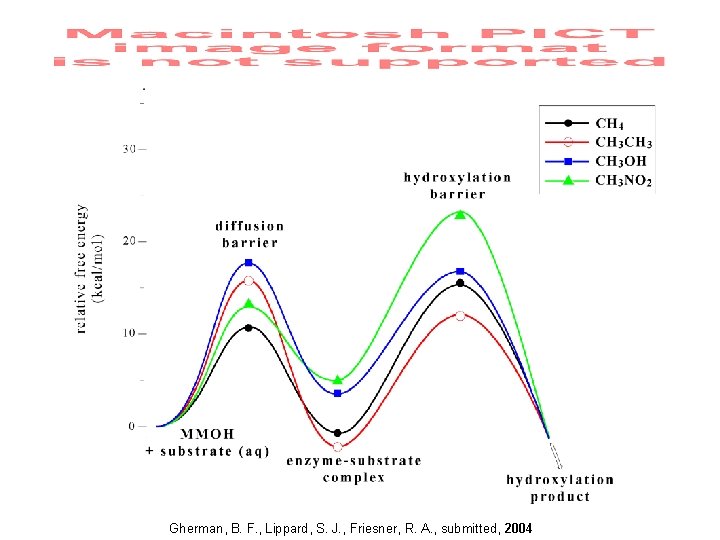
Gherman, B. F. , Lippard, S. J. , Friesner, R. A. , submitted, 2004
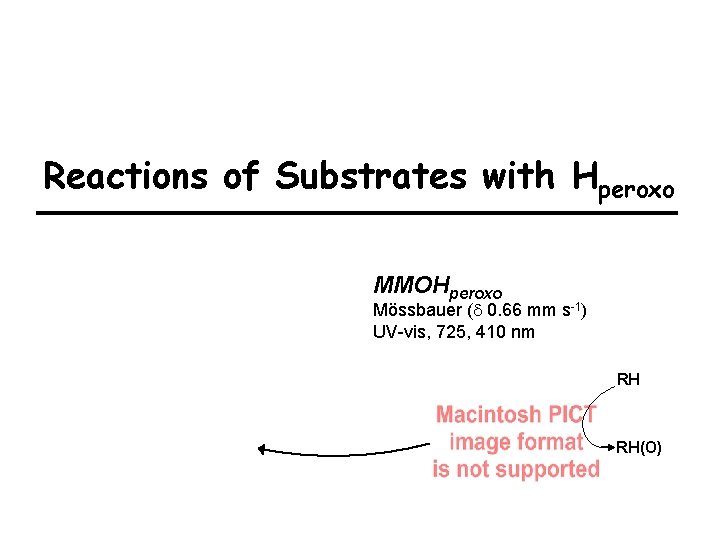
Reactions of Substrates with Hperoxo MMOHperoxo Mössbauer (d 0. 66 mm s-1) UV-vis, 725, 410 nm RH RH(O)

Preliminary Evidence for Hperoxo Reacting with Substrates filled circles - propylene open circles – methane Hperoxo appears to react with propylene Low solubility of substrates limits experiment Could propylene accelerate the conversion of Hperoxo to Q? Valentine, A. M. ; Stahl, S. S. ; Lippard, S. J. J. Am. Chem. Soc. 1999, 121, 3876 -3887.

Hperoxo and Q Reactions with Ethyl Vinyl Ether l 705 or 720 nm l 420 nm Conditions: T = 20 ºC, [H]red = 51. 5 m. M, [B] = 103 m. M Ether concentration in excess and variable, 3 - 70 m. M The product of propylene reaction with Hperoxo is propylene oxide
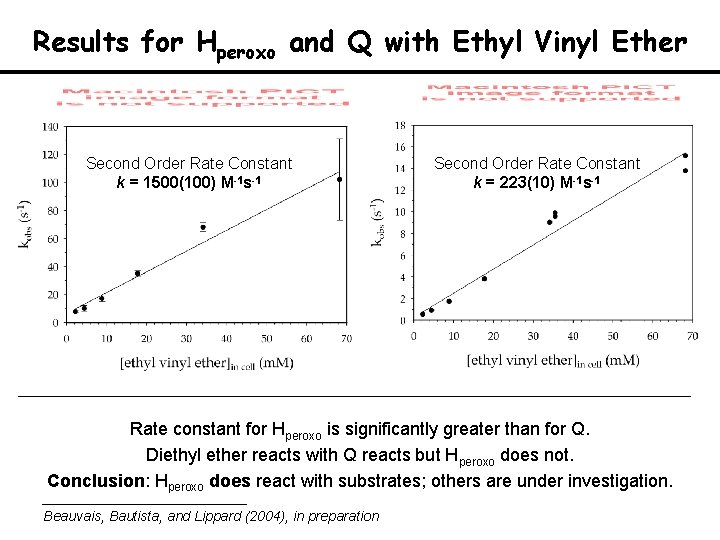
Results for Hperoxo and Q with Ethyl Vinyl Ether Second Order Rate Constant k = 1500(100) M-1 s-1 Second Order Rate Constant k = 223(10) M-1 s-1 Rate constant for Hperoxo is significantly greater than for Q. Diethyl ether reacts with Q reacts but Hperoxo does not. Conclusion: Hperoxo does react with substrates; others are under investigation. Beauvais, Bautista, and Lippard (2004), in preparation

Mechanisms for Epoxidation of Ethyl Vinyl Ether By Hperoxo: By Q: Hperoxo may react by 2 -electron transfer and Q by single-electron transfer. In addition, there might be distinct binding and activation steps.
O 2 B MMOHred MMOHox B H+ MMOHsuperoxo Substrate Access in MMOH and To. MOH B MMOHperoxo CH 3 OH RH CH 4 RH(O) B
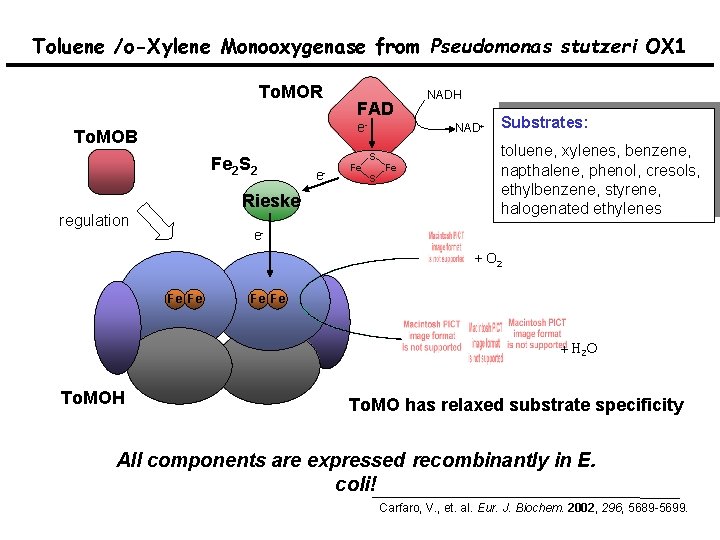
Toluene /o-Xylene Monooxygenase from Pseudomonas stutzeri OX 1 To. MOR FAD e- To. MOB Fe 2 S 2 Rieske regulation e- Fe NADH NAD+ S S Fe Substrates: toluene, xylenes, benzene, napthalene, phenol, cresols, ethylbenzene, styrene, halogenated ethylenes e+ O 2 Fe Fe + H 2 O To. MOH To. MO has relaxed substrate specificity All components are expressed recombinantly in E. coli! Carfaro, V. , et. al. Eur. J. Biochem. 2002, 296, 5689 -5699.
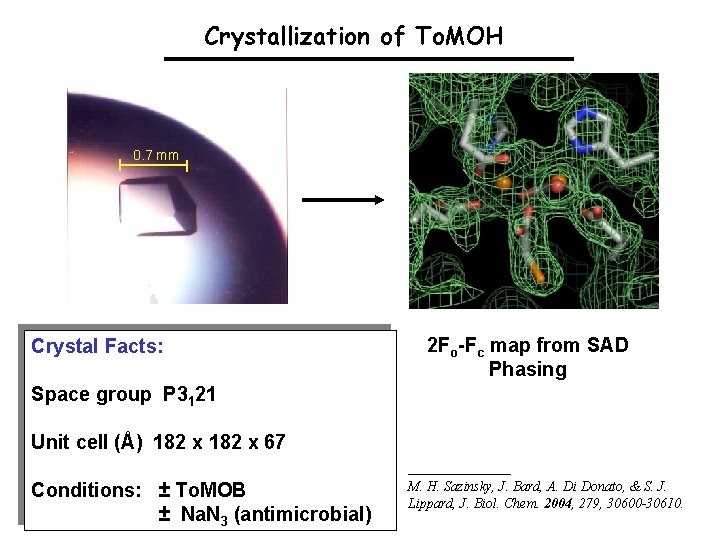
Crystallization of To. MOH 0. 7 mm Crystal Facts: 2 Fo-Fc map from SAD Phasing Space group P 3121 Unit cell (Å) 182 x 67 Conditions: ± To. MOB ± Na. N 3 (antimicrobial) M. H. Sazinsky, J. Bard, A. Di Donato, & S. J. Lippard, J. Biol. Chem. 2004, 279, 30600 -30610.
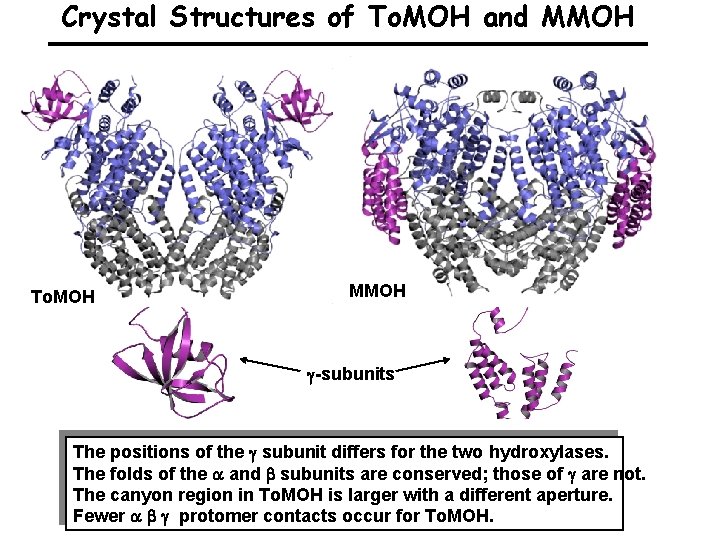
Crystal Structures of To. MOH and MMOH To. MOH MMOH -subunits The positions of the subunit differs for the two hydroxylases. The folds of the and subunits are conserved; those of are not. The canyon region in To. MOH is larger with a different aperture. Fewer protomer contacts occur for To. MOH.
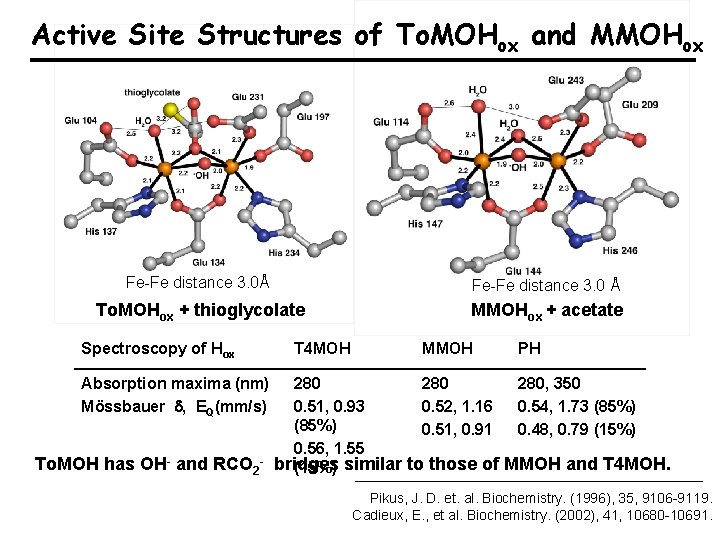
Active Site Structures of To. MOHox and MMOHox Fe-Fe distance 3. 0Å Fe-Fe distance 3. 0 Å To. MOHox + thioglycolate MMOHox + acetate Spectroscopy of Hox Absorption maxima (nm) Mössbauer , EQ(mm/s) To. MOH has OH- and RCO 2 - T 4 MOH MMOH PH 280 280, 350 0. 51, 0. 93 0. 52, 1. 16 0. 54, 1. 73 (85%) 0. 51, 0. 91 0. 48, 0. 79 (15%) 0. 56, 1. 55 bridges (15%) similar to those of MMOH and T 4 MOH. Pikus, J. D. et. al. Biochemistry. (1996), 35, 9106 -9119. Cadieux, E. , et al. Biochemistry. (2002), 41, 10680 -10691.
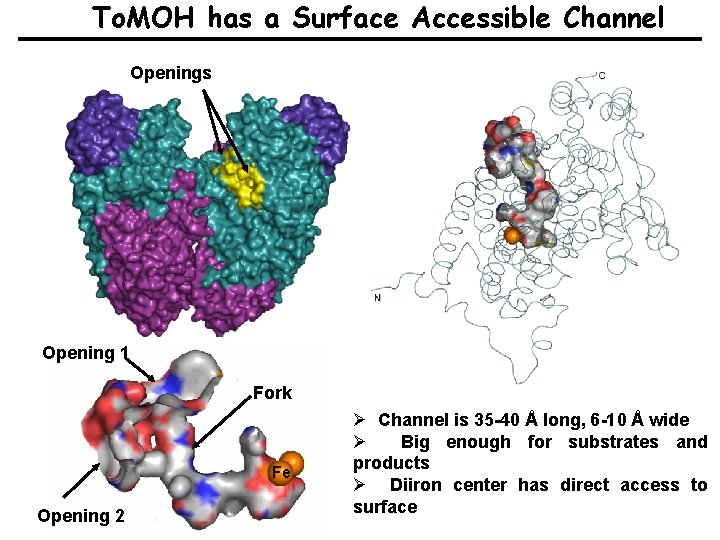
To. MOH has a Surface Accessible Channel Openings Opening 1 Fork Fe Opening 2 Ø Channel is 35 -40 Å long, 6 -10 Å wide Ø Big enough for substrates and products Ø Diiron center has direct access to surface
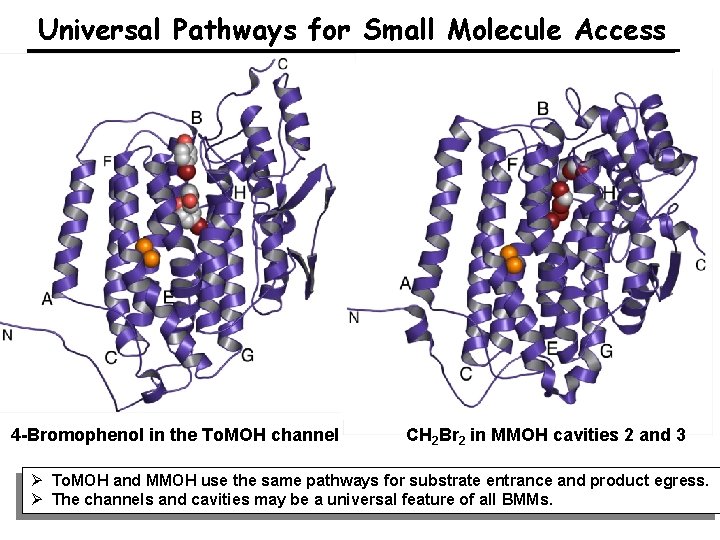
Universal Pathways for Small Molecule Access 4 -Bromophenol in the To. MOH channel CH 2 Br 2 in MMOH cavities 2 and 3 Ø To. MOH and MMOH use the same pathways for substrate entrance and product egress. Ø The channels and cavities may be a universal feature of all BMMs.
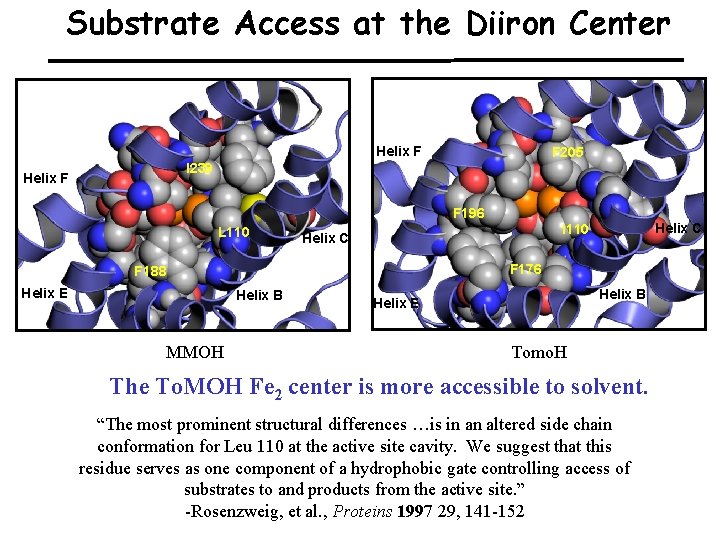
Substrate Access at the Diiron Center Helix F F 205 I 239 Helix F F 196 L 110 F 176 F 188 Helix E Helix B MMOH Helix C I 110 Helix C Helix B Helix E Tomo. H The To. MOH Fe 2 center is more accessible to solvent. “The most prominent structural differences …is in an altered side chain conformation for Leu 110 at the active site cavity. We suggest that this residue serves as one component of a hydrophobic gate controlling access of substrates to and products from the active site. ” -Rosenzweig, et al. , Proteins 1997 29, 141 -152
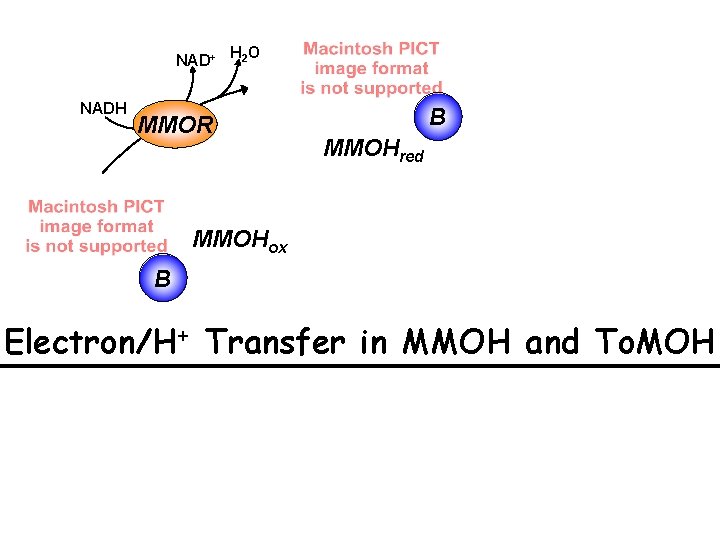
NAD+ NADH H 2 O MMOR B MMOHred MMOHox B Electron/H+ Transfer in MMOH and To. MOH
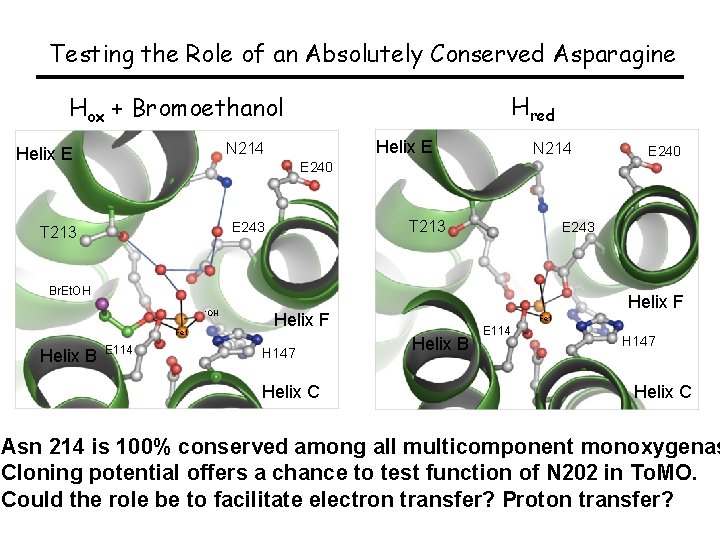
Testing the Role of an Absolutely Conserved Asparagine Hred Hox + Bromoethanol Helix E N 214 T 213 E 243 Helix E N 214 E 240 T 213 E 243 Br. Et. OH -OH Fe 1 Helix B E 114 Helix F H 147 Helix C E 240 Fe 1 Helix B E 114 H 147 Helix C Asn 214 is 100% conserved among all multicomponent monoxygenas Cloning potential offers a chance to test function of N 202 in To. MO. Could the role be to facilitate electron transfer? Proton transfer?

Steady-State Turnover of WT and N 202 A To. MOH (E. R. Cadieux and S. J. Lippard, Unpublished results (2004)) WT Specific Activity (nmol/min/ mg) WT 19% N 202 A 25% N 202 A Single turnover results for To. MOHred + O 2 + To. MOD + phenol reveal no difference between wild type and N 202 A mutant protein activities. Working hypothesis: E. T. or H + pathway involves N 202 in the To. MO system.

- Slides: 64