Principles of Bioinorganic Chemistry 2003 Metallochaperones Metal Folding

Principles of Bioinorganic Chemistry - 2003

Metallochaperones; Metal Folding PRINCIPLES: • Metallochaperones guide and protect metals to natural sites • Chaperone and target receptor protein structurally homologou • Metal-mediated protein structure changes affect transcription • Metal-mediated protein structure changes affect translation • Metal-induced protein structure changes also activate enzymes • Metal-induced bending of DNA affects function • Metal ionic radii and M–L water bridging are used to advantage ILLUSTRATIONS: • Copper insertion into metalloenzymes • Zinc finger proteins control transcription • Ca 2+, a second messenger and sentinel at the synapse • Cisplatin, an anticancer drug
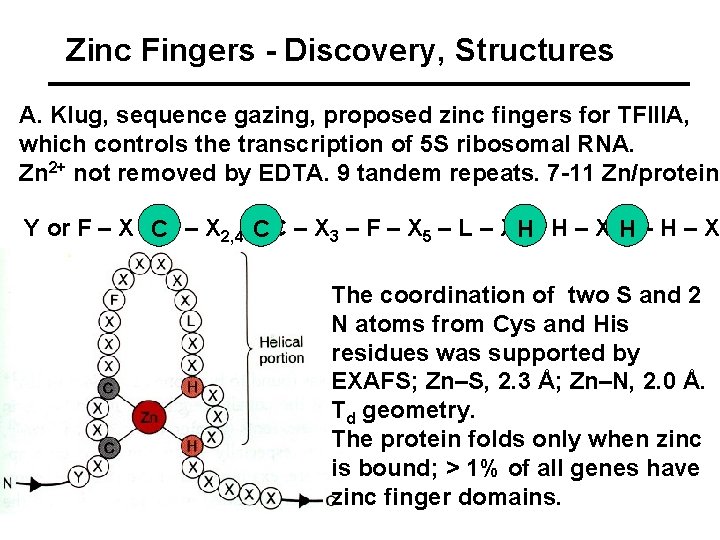
Zinc Fingers - Discovery, Structures A. Klug, sequence gazing, proposed zinc fingers for TFIIIA, which controls the transcription of 5 S ribosomal RNA. Zn 2+ not removed by EDTA. 9 tandem repeats. 7 -11 Zn/protein. Y or F – X –CC – X 2, 4 –CC – X 3 – F – X 5 – L – X 2 H– H – X 3, 4 H– H – X The coordination of two S and 2 N atoms from Cys and His residues was supported by EXAFS; Zn–S, 2. 3 Å; Zn–N, 2. 0 Å. Td geometry. The protein folds only when zinc is bound; > 1% of all genes have zinc finger domains.
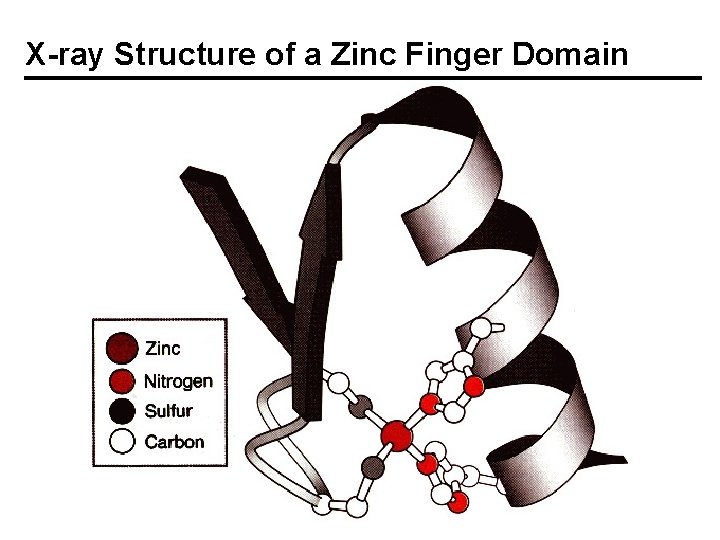
X-ray Structure of a Zinc Finger Domain
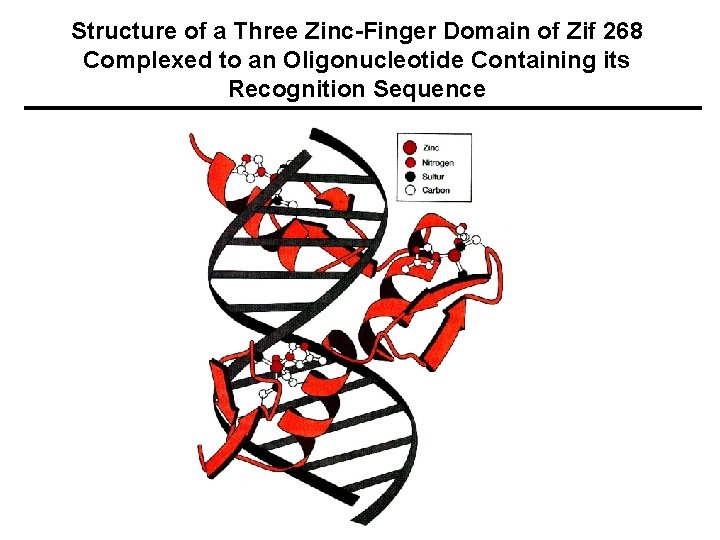
Structure of a Three Zinc-Finger Domain of Zif 268 Complexed to an Oligonucleotide Containing its Recognition Sequence
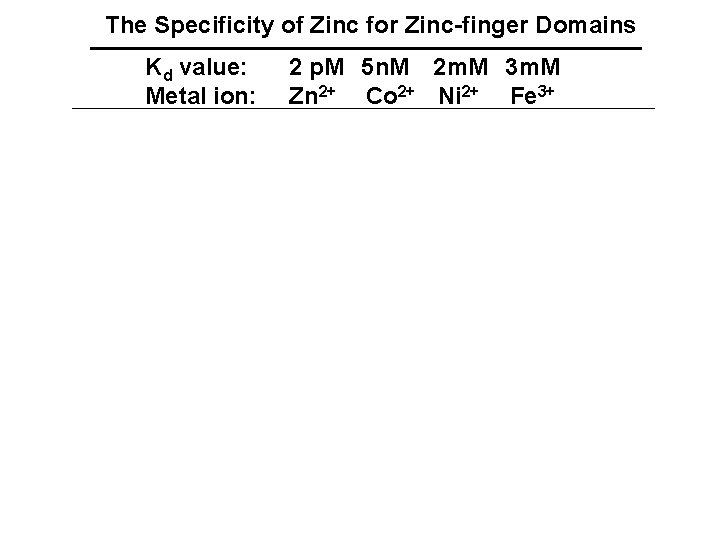
The Specificity of Zinc for Zinc-finger Domains Kd value: Metal ion: 2 p. M 5 n. M 2 m. M 3 m. M Zn 2+ Co 2+ Ni 2+ Fe 3+

Metal Folding of Biopolymers PRINCIPLES: • Metal-mediated protein structure changes affect transcription • Metal-mediated protein structure changes affect translation • Metal-induced protein structure changes also activate enzymes • Metal-induced DNA structure changes effect anticancer activity ILLUSTRATIONS: • Ca 2+, a second messenger and sentinel at the synapse • Cisplatin, an anticancer drug

Roles of Calcium in Protein Folding and Activation Calcium at the synapse: binds to the C 2 domains of synaptotagmin and helps to control neurological signal transduction. Synaptotagmin is the sensor for Ca 2+ influx. Calcium also binds to calmodulin. As Ca 2+ levels in the cell increase, calmodulin senses these changes and binds to its target proteins with Kd values of 10 - 100 n. M or less. The following are amino acid sequences from activated proteins:

Synaptotagmin is a calcium sensor Synaptotagmin III with Mg 2+ C 2 A-C 2 B crystal structure Brunger and coworkers 1999
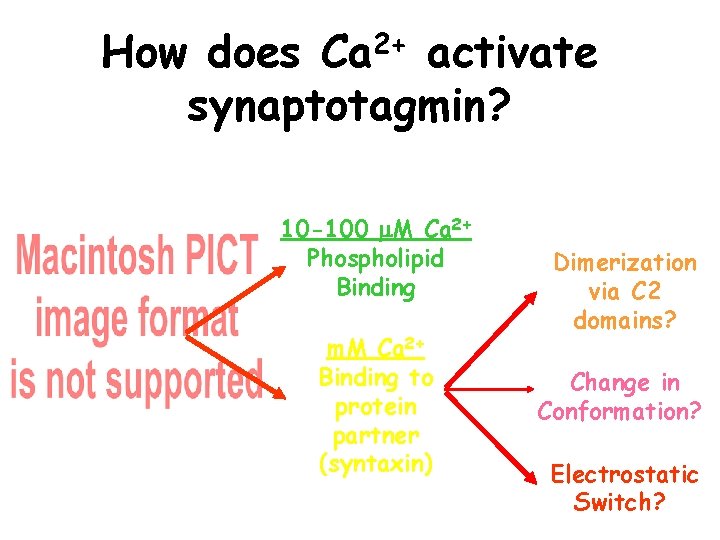
How does Ca 2+ activate synaptotagmin? 10 -100 M Ca 2+ Phospholipid Binding m. M Ca 2+ Binding to protein partner (syntaxin) Dimerization via C 2 domains? Change in Conformation? Electrostatic Switch?
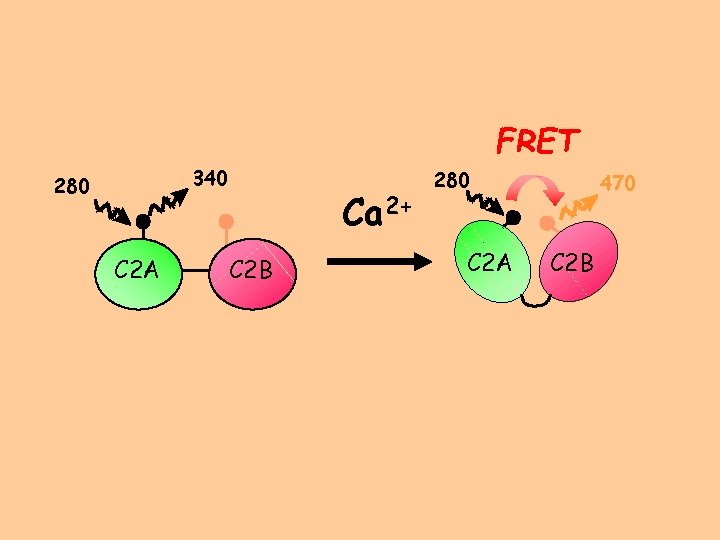
FRET 340 280 C 2 A Ca 2+ C 2 B 280 C 2 A 470 C 2 B

Addition of Calcium Results in an Increase in Energy Transfer apo m. M Ca 2+ Garcia, R. A. ; Forde, C. A. ; Godwin, H. A. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 5883 -5888.

Other metal ions that can promote fusion also confer this conformation change, but to a lesser extent than does Ca 2+ Garcia, R. A. ; Forde, C. A. ; Godwin, H. A. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 5883 -5888.
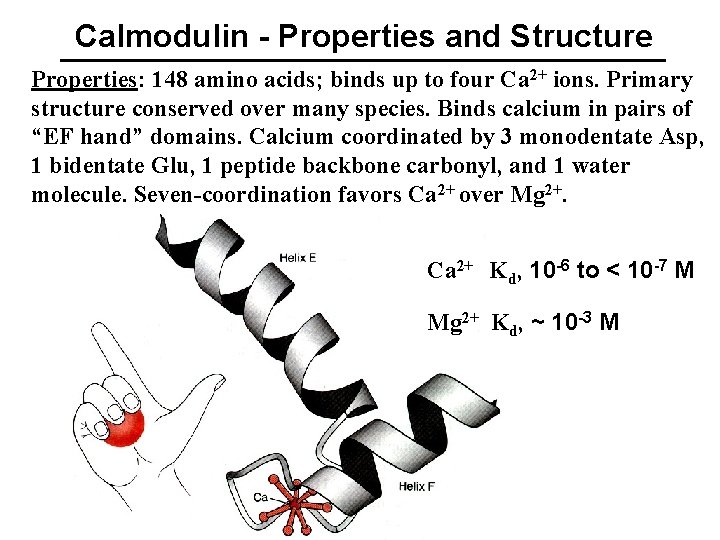
Calmodulin - Properties and Structure Properties: 148 amino acids; binds up to four Ca 2+ ions. Primary structure conserved over many species. Binds calcium in pairs of “EF hand” domains. Calcium coordinated by 3 monodentate Asp, 1 bidentate Glu, 1 peptide backbone carbonyl, and 1 water molecule. Seven-coordination favors Ca 2+ over Mg 2+. Ca 2+ Kd, 10 -6 to < 10 -7 M Mg 2+ Kd, ~ 10 -3 M
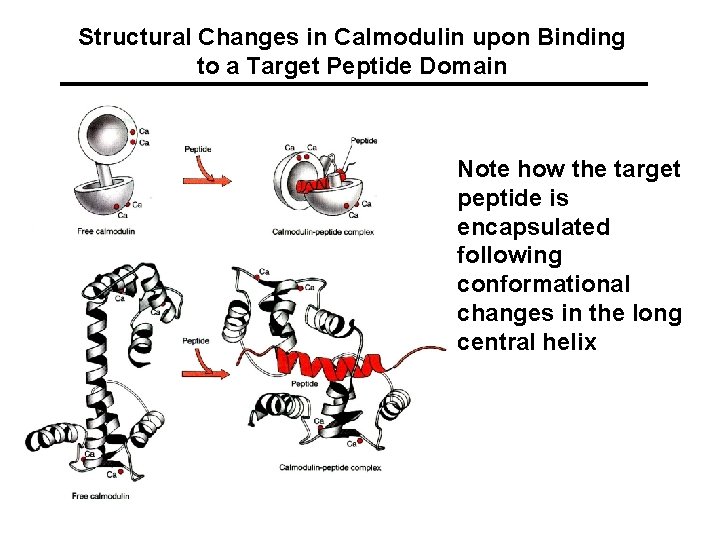
Structural Changes in Calmodulin upon Binding to a Target Peptide Domain Note how the target peptide is encapsulated following conformational changes in the long central helix
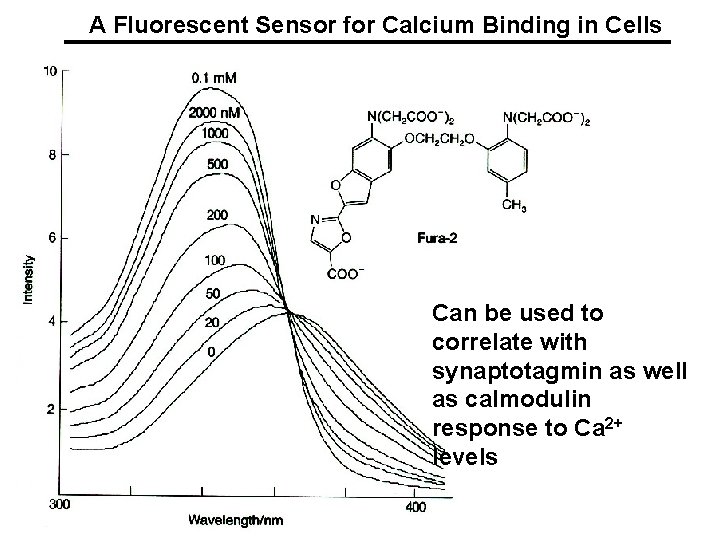
A Fluorescent Sensor for Calcium Binding in Cells Can be used to correlate with synaptotagmin as well as calmodulin response to Ca 2+ levels

Summary - Points to Remember • In eukaryotes iron homeostasis is achieved by regulating the concentrations of the transferrin receptor and apoferritin at the translational level. IRP’s are the conformational switch and utilize an Fe 4 S 4 cluster to alter protein structure. • A mercury resistance operon in bacteria detoxifies R 2 Hg by lyase and reduction to Hg(0) which volatilizes harmlessly out of the cell. Mer. R is the sensor which undergoes a conformational change upon Hg binding and activates the genes at the level of transcription. This topic was not covered in class, but please read Chapter 6, Section 6. 3.

Summary - Points to Remember, cont’d • Zinc fingers form upon metal-binding to the apo peptide domains and activate transcription of thousands of genes. Evidence is accumulating that zinc-binding domains also act as metalloregulators in response to zinc levels in the cell. • Synaptotagmin C 2 domains are the calcium sensors at the synapse responsible for Ca-dependent signal transduction in helping to achieve synaptic plasticity. • Calmodulin alters its structure upon calcium influx into cells and activates many genes at the transcriptional level. Calmodulin allows calcium to serve as a second messenger in response to signals at the cell surface, affording a signal transduction pathway.

Syphilis Bacterial infection




Factors Associated with Antitumor Drug Development A. Serendipity: > 7000: 1 in most screens B. Perseverance: ~ 13 years between laboratory and pharmacy C. Cost: > $750 M D. Moral questions: gene therapy E. The aversion to metal-based pharmaceuticals
Cisplatin: From DNA Damage to Cancer Therapy Idea Creativity Understanding & Invention Serendipity Intelligence Discovery


% Survival Cisplatin: An Extraordinary Anticancer Drug w/ cisplatin w/o cisplatin Cisplatin cis-diamminedichloroplatinum(II) Years after Orchidectomy source: I. Damjanov, Scientific American, 1995 • Cisplatin is highly effective against testicular cancer. It is also used to treat head, neck, ovarian, and several other types of human tumors.

- Slides: 27