Principles of Antenatal Screening for Downs Syndrome Cecily

























- Slides: 54

Principles of Antenatal Screening for Down’s Syndrome Cecily Cromby Wolfson Institute of Preventive Medicine Barts and The London, Queen Mary’s School of Medicine and Dentistry

Definition of Screening is the systematic application of a test or inquiry, to identify individuals at sufficient risk of a specific disorder to benefit from further investigation or direct preventive action, among persons who have not sought medical attention on account of symptoms of that disorder. NJ Wald, J Med Screen, 1: 76, 1994

Definition of Screening The purpose of antenatal screening for Down’s syndrome is to offer women a test that can identify those with a high risk of having an affected pregnancy so that they can be offered a diagnostic test and, if a diagnosis of Down’s syndrome is made can have a termination of pregnancy if they wish.

Screening terminology Detection Rate (DR) Proportion of affected individuals with screen positive results. Also known as sensitivity. False Positive Rate (FPR) Proportion of unaffected individuals with screen positive results. Specificity is the complement of FPR, the proportion of unaffected individuals with screen negative results. Odds of being affected given a positive result (OAPR) Ratio of affected positives : unaffected positives Sometimes expressed as the positive predictive value (PPV): affected positives / all positives

Screening terminology Screen positive Screen negative Totals Down’s syndrome A True +ve Unaffected Totals B False +ve A+B C False –ve A+C D True -ve B+D C+D A+B+C+D

Screening terminology Screen positive Screen negative Totals Down’s syndrome A True +ve Unaffected Totals B False +ve A+B C False –ve A+C D True -ve B+D C+D DR = A A+C FPR = B B+D A+B+C+D

Screening terminology Median The median is the middle value if all the values are laid out in a straight line in numerical value. For screening purposes it is a better estimate of the ‘average’ than the mean as it is less influenced by values at either extreme of the distribution.

Screening terminology Median 5 11 19 23 24 27 29 35 41 50 89 Median level of AFP at 16 weeks gestation = 27 iu/m. L

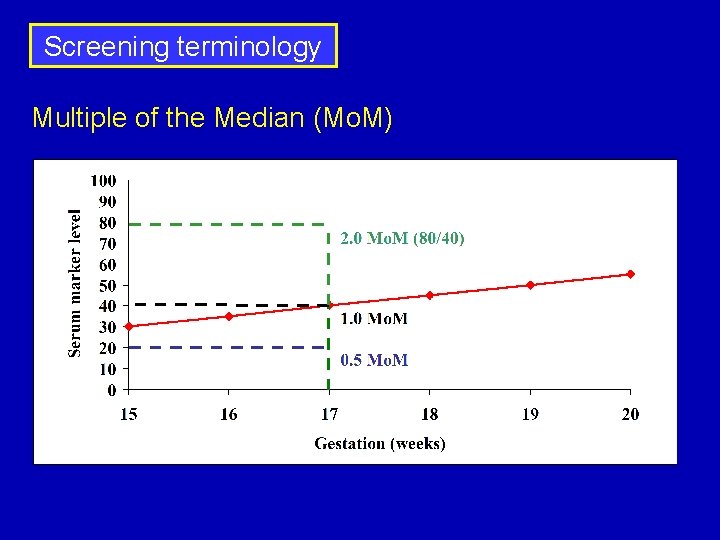
Screening terminology Multiple of the Median (Mo. M) Mo. M = Measured Value Median Value Mo. M = Observed Value Expected Value

Screening terminology Multiple of the Median (Mo. M)

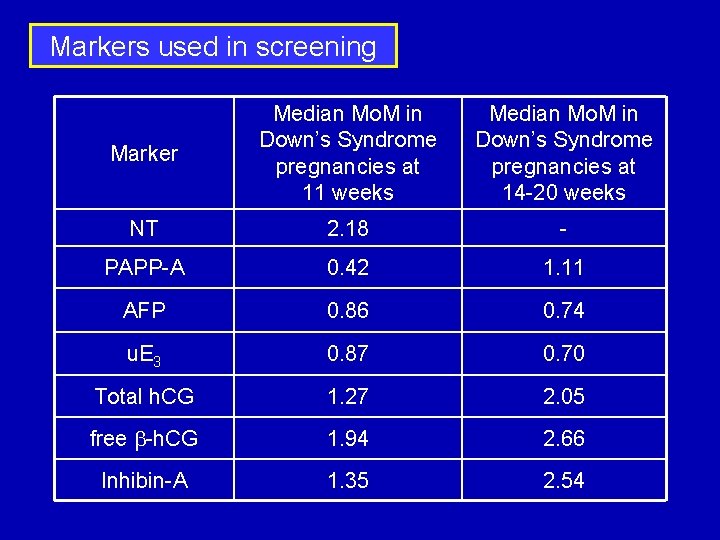
Markers used in screening Marker Median Mo. M in Down’s Syndrome pregnancies at 11 weeks Median Mo. M in Down’s Syndrome pregnancies at 14 -20 weeks NT 2. 18 - PAPP-A 0. 42 1. 11 AFP 0. 86 0. 74 u. E 3 0. 87 0. 70 Total h. CG 1. 27 2. 05 free b-h. CG 1. 94 2. 66 Inhibin-A 1. 35 2. 54

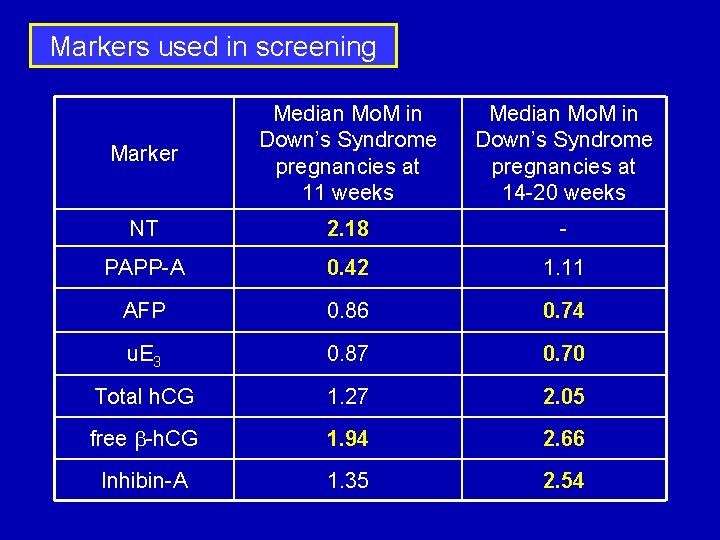
Markers used in screening Marker Median Mo. M in Down’s Syndrome pregnancies at 11 weeks Median Mo. M in Down’s Syndrome pregnancies at 14 -20 weeks NT 2. 18 - PAPP-A 0. 42 1. 11 AFP 0. 86 0. 74 u. E 3 0. 87 0. 70 Total h. CG 1. 27 2. 05 free b-h. CG 1. 94 2. 66 Inhibin-A 1. 35 2. 54

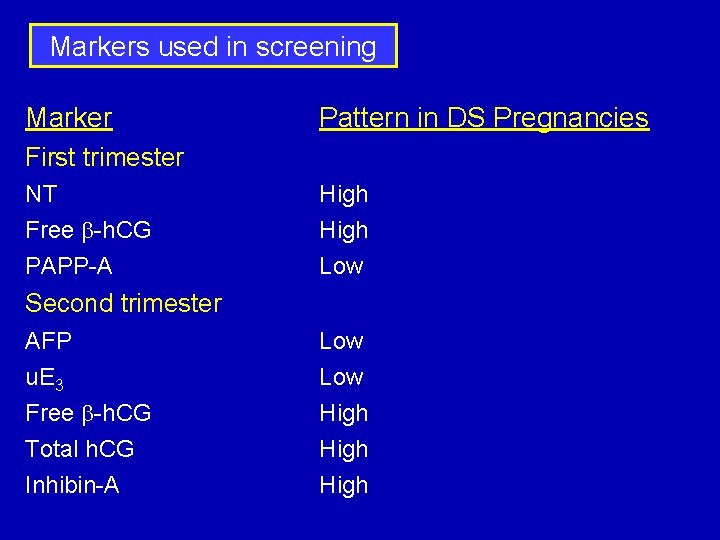
Markers used in screening Marker Pattern in DS Pregnancies First trimester NT Free b-h. CG PAPP-A High Low Second trimester AFP u. E 3 Free b-h. CG Total h. CG Inhibin-A Low High

Markers used in screening Marker Origin First trimester NT Free b-h. CG PAPP-A Placenta Second trimester AFP u. E 3 Free b-h. CG Total h. CG Inhibin-A Fetus & Placenta

Markers used in screening Marker Variation with GA First trimester NT Free b-h. CG PAPP-A Increases Decreases Increases Second trimester AFP u. E 3 Free b-h. CG Total h. CG Inhibin-A Increases Decreases until 17 wks, then increases

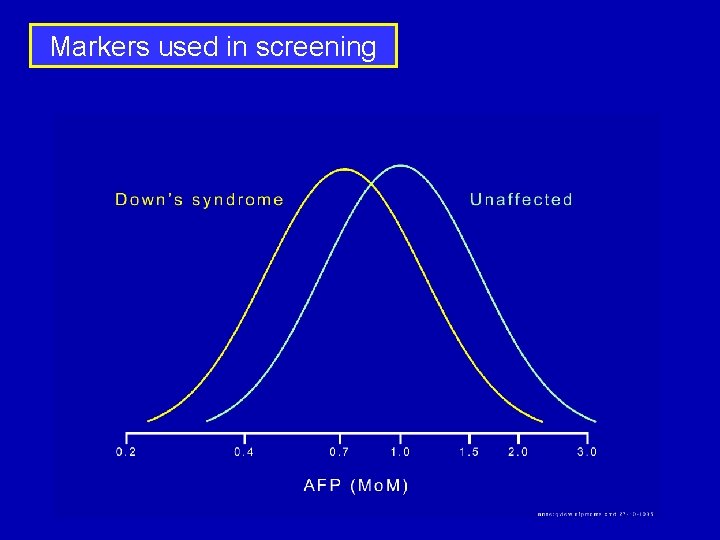
Markers used in screening

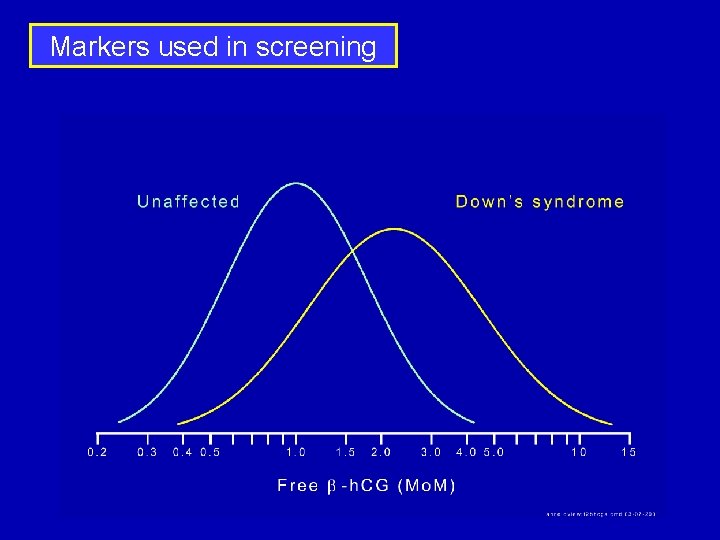
Markers used in screening

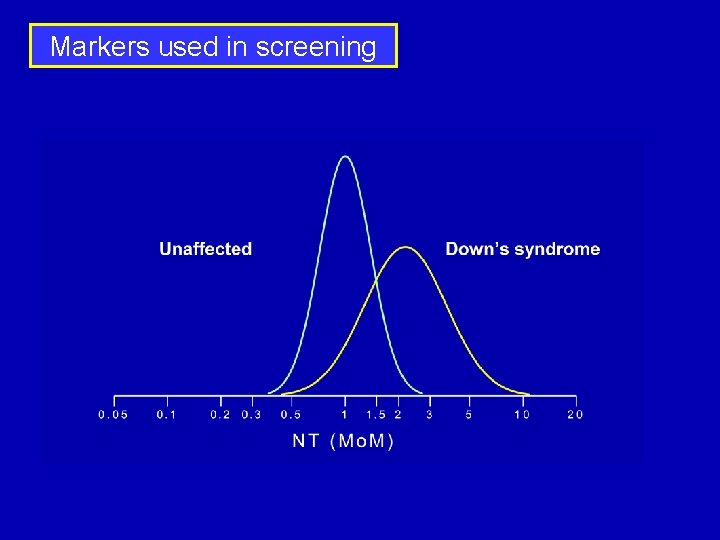
Markers used in screening

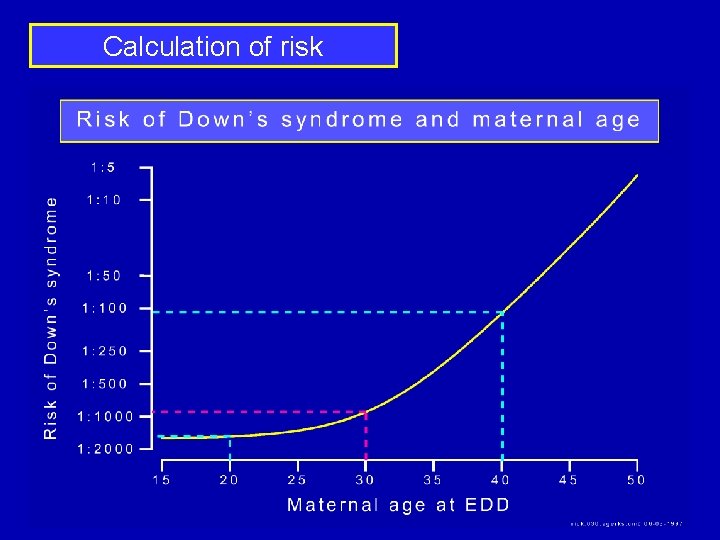
Calculation of risk

Calculation of risk

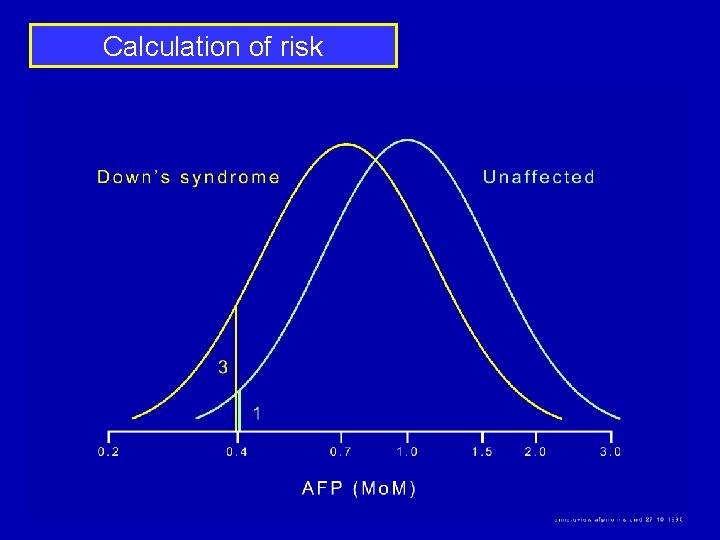
Calculation of risk 30 -year-old: Age specific risk of Down’s syndrome is 1 in 900 AFP level of 0. 4 Mo. M Likelihood Ratio of 3: increases risk 3 -fold 3 x 1 in 900 = 1 in 300

Calculation of risk

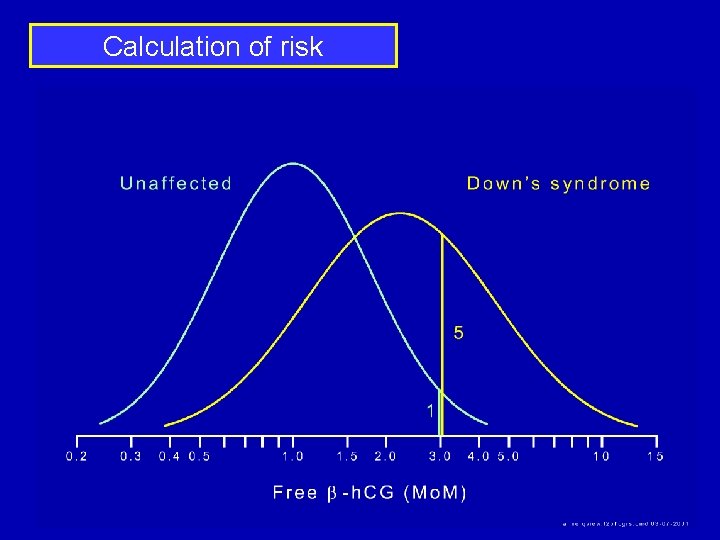
Calculation of risk Age and AFP risk: 1 in 300 h. CG level of 3 Mo. M Likelihood Ratio of 5: increases risk 5 -fold 5 x 1 in 300 = 1 in 60

Calculation of risk

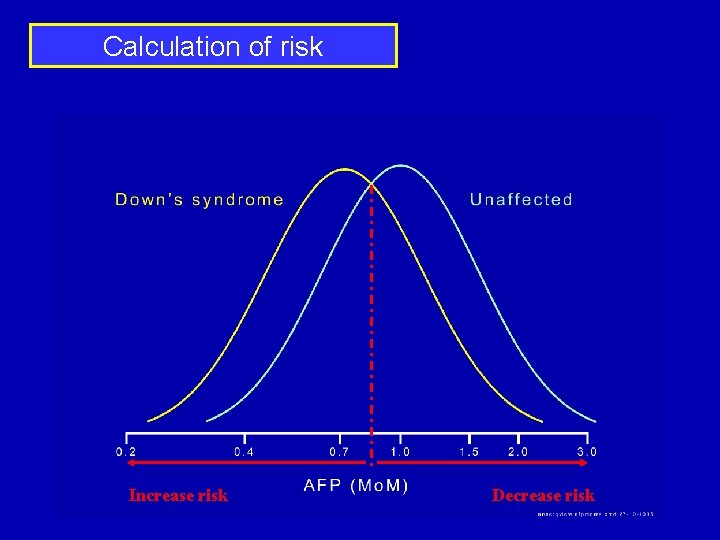
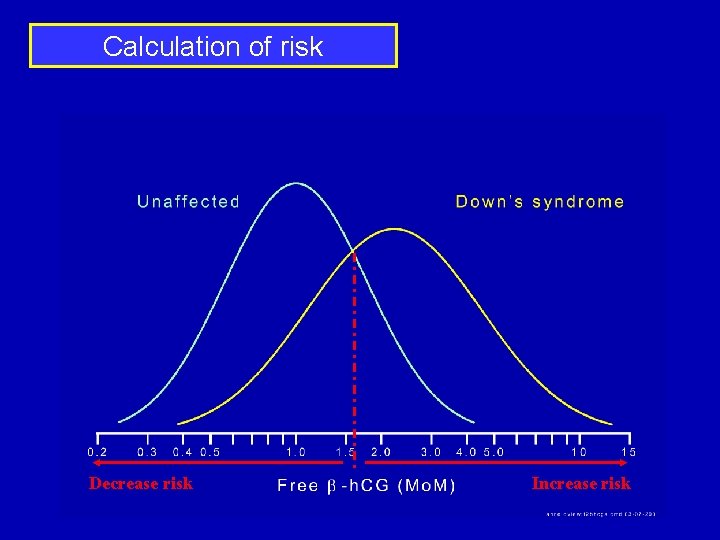
Calculation of risk Increase risk Decrease risk

Calculation of risk Decrease risk Increase risk

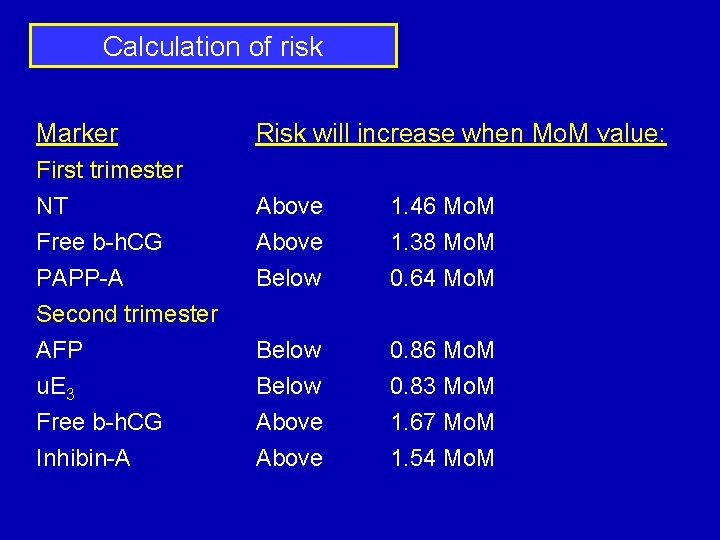
Calculation of risk Marker First trimester NT Free b-h. CG PAPP-A Second trimester AFP u. E 3 Free b-h. CG Inhibin-A Risk will increase when Mo. M value: Above Below 1. 46 Mo. M 1. 38 Mo. M 0. 64 Mo. M Below Above 0. 86 Mo. M 0. 83 Mo. M 1. 67 Mo. M 1. 54 Mo. M

Calculation of risk

Calculation of risk Factors affecting interpretation of results • Ultrasound dating • Previous Down’s or NTD pregnancy • Weight • Twins • Insulin dependent diabetes • Ethnic group • Smoking • IVF • Vaginal bleeding • Amniocentesis

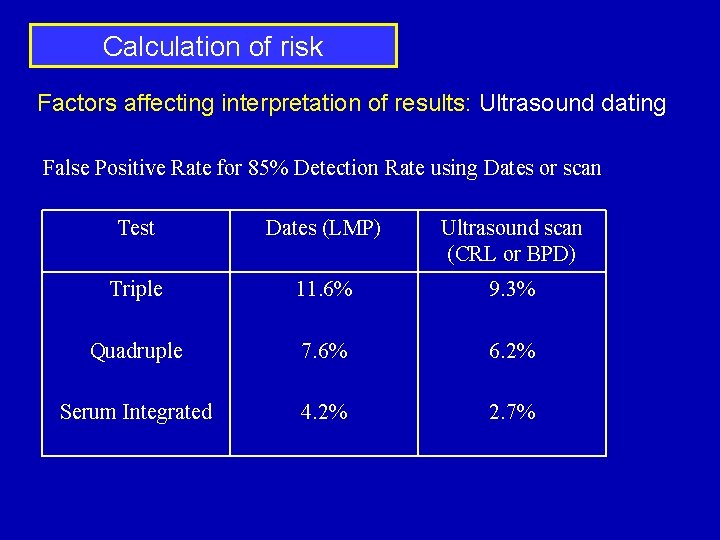
Calculation of risk Factors affecting interpretation of results: Ultrasound dating False Positive Rate for 85% Detection Rate using Dates or scan Test Dates (LMP) Ultrasound scan (CRL or BPD) Triple 11. 6% 9. 3% Quadruple 7. 6% 6. 2% Serum Integrated 4. 2% 2. 7%

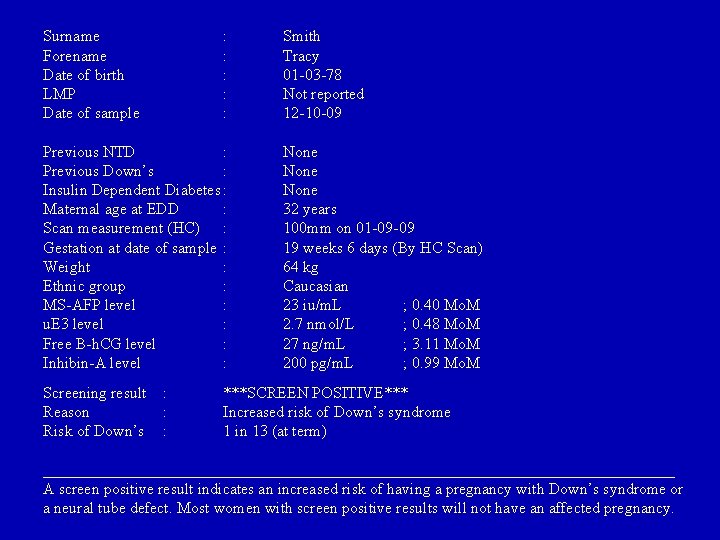
Surname Forename Date of birth LMP Date of sample : : : Previous NTD : Previous Down’s : Insulin Dependent Diabetes : Maternal age at EDD : Scan measurement (HC) : Gestation at date of sample : Weight : Ethnic group : MS-AFP level : u. E 3 level : Free B-h. CG level : Inhibin-A level : Screening result Reason Risk of Down’s : : : Smith Tracy 01 -03 -78 Not reported 12 -10 -09 None 32 years 100 mm on 01 -09 -09 19 weeks 6 days (By HC Scan) 64 kg Caucasian 23 iu/m. L ; 0. 40 Mo. M 2. 7 nmol/L ; 0. 48 Mo. M 27 ng/m. L ; 3. 11 Mo. M 200 pg/m. L ; 0. 99 Mo. M ***SCREEN POSITIVE*** Increased risk of Down’s syndrome 1 in 13 (at term) ________________________________________ A screen positive result indicates an increased risk of having a pregnancy with Down’s syndrome or a neural tube defect. Most women with screen positive results will not have an affected pregnancy.

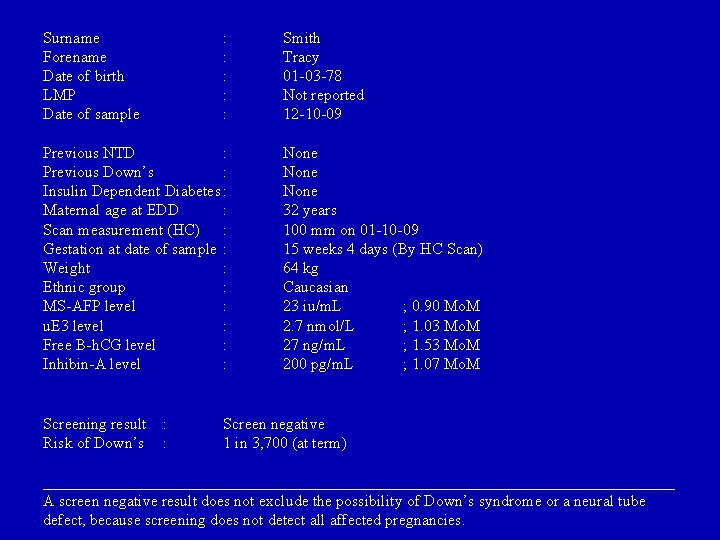
Surname Forename Date of birth LMP Date of sample : : : Previous NTD : Previous Down’s : Insulin Dependent Diabetes : Maternal age at EDD : Scan measurement (HC) : Gestation at date of sample : Weight : Ethnic group : MS-AFP level : u. E 3 level : Free B-h. CG level : Inhibin-A level : Screening result Risk of Down’s : : Smith Tracy 01 -03 -78 Not reported 12 -10 -09 None 32 years 100 mm on 01 -10 -09 15 weeks 4 days (By HC Scan) 64 kg Caucasian 23 iu/m. L ; 0. 90 Mo. M 2. 7 nmol/L ; 1. 03 Mo. M 27 ng/m. L ; 1. 53 Mo. M 200 pg/m. L ; 1. 07 Mo. M Screen negative 1 in 3, 700 (at term) ________________________________________ A screen negative result does not exclude the possibility of Down’s syndrome or a neural tube defect, because screening does not detect all affected pregnancies.

Calculation of risk Factors affecting interpretation of results: Previous DS or NTD Estimated that there is a recurrence risk of 1 in 290 (3. 45 per 1, 000) This is added to the unaffected risk at term

Calculation of risk Factors affecting interpretation of results: Weight The levels of the serum markers are, on average, higher in lighter women and lower in heavier women The markers produced by the fetus and the placenta are more concentrated in the blood of smaller women and more diluted in the blood of larger women On average, for a 20 kg increase in maternal weight: - Serum AFP decreases by about 17% - u. E 3 decreases by about 7% - h. CG decreases by about 16%

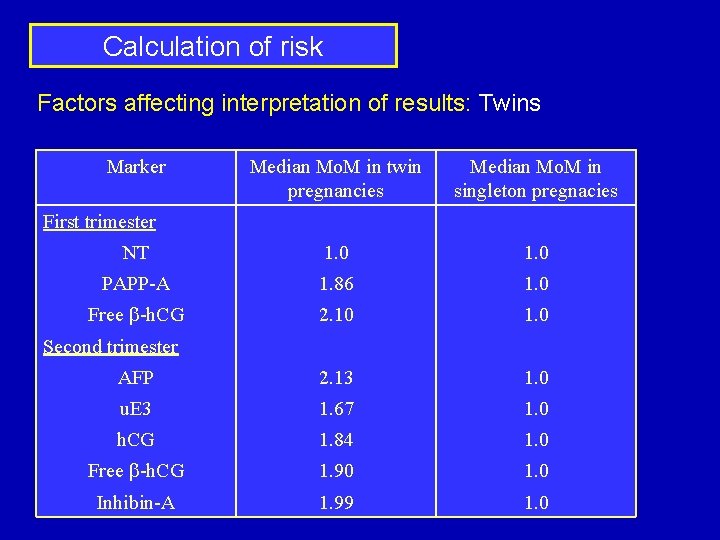
Calculation of risk Factors affecting interpretation of results: Twins Marker Median Mo. M in twin pregnancies Median Mo. M in singleton pregnacies NT 1. 0 PAPP-A 1. 86 1. 0 Free b-h. CG 2. 10 1. 0 AFP 2. 13 1. 0 u. E 3 1. 67 1. 0 h. CG 1. 84 1. 0 Free b-h. CG 1. 90 1. 0 Inhibin-A 1. 99 1. 0 First trimester Second trimester

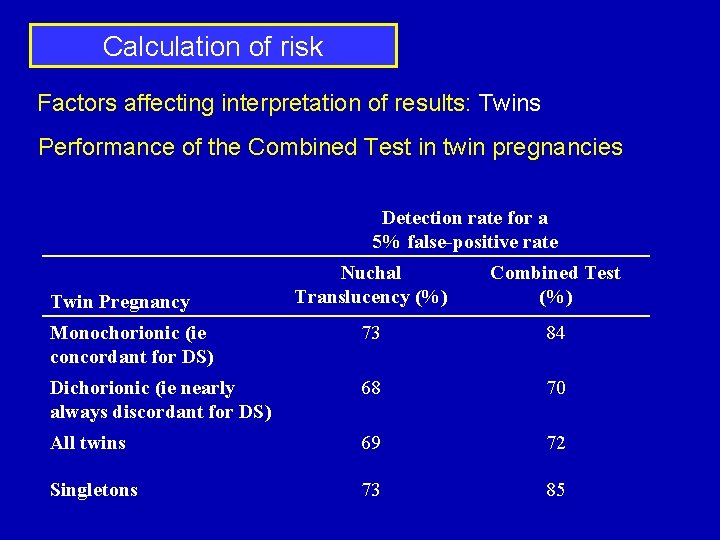
Calculation of risk Factors affecting interpretation of results: Twins Performance of the Combined Test in twin pregnancies Detection rate for a 5% false-positive rate Nuchal Translucency (%) Combined Test (%) Monochorionic (ie concordant for DS) 73 84 Dichorionic (ie nearly always discordant for DS) 68 70 All twins 69 72 Singletons 73 85 Twin Pregnancy

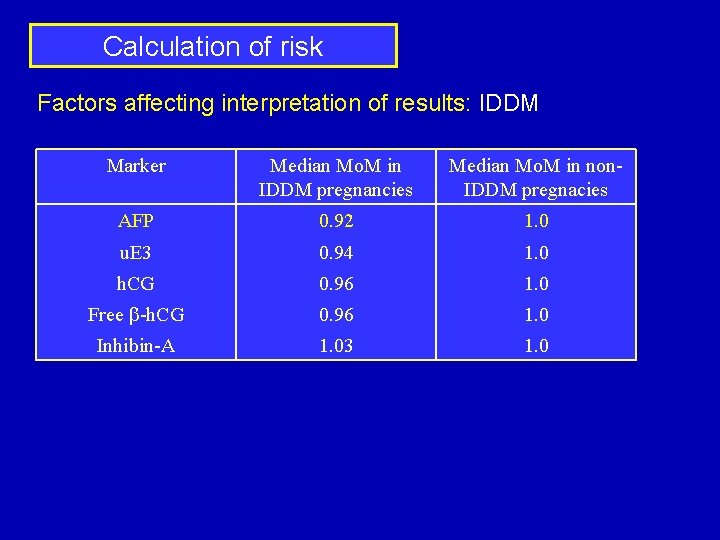
Calculation of risk Factors affecting interpretation of results: IDDM Marker Median Mo. M in IDDM pregnancies Median Mo. M in non. IDDM pregnacies AFP 0. 92 1. 0 u. E 3 0. 94 1. 0 h. CG 0. 96 1. 0 Free b-h. CG 0. 96 1. 0 Inhibin-A 1. 03 1. 0

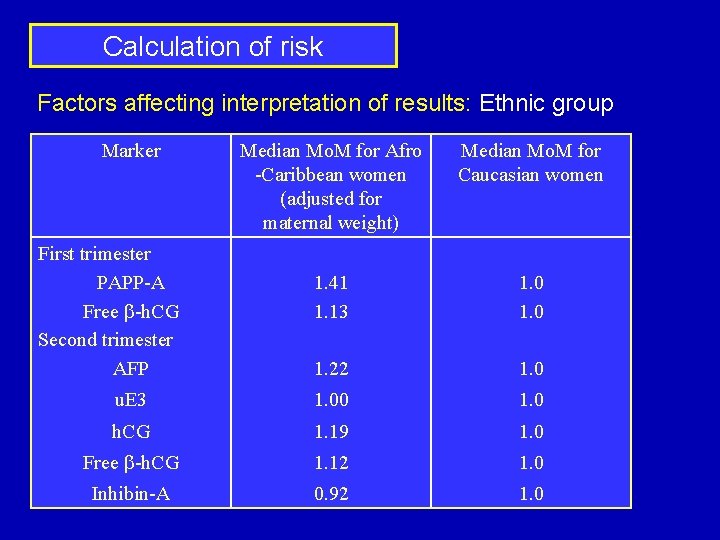
Calculation of risk Factors affecting interpretation of results: Ethnic group Marker Median Mo. M for Afro -Caribbean women (adjusted for maternal weight) Median Mo. M for Caucasian women 1. 41 1. 13 1. 0 1. 22 1. 0 u. E 3 1. 00 1. 0 h. CG 1. 19 1. 0 Free b-h. CG 1. 12 1. 0 Inhibin-A 0. 92 1. 0 First trimester PAPP-A Free b-h. CG Second trimester AFP

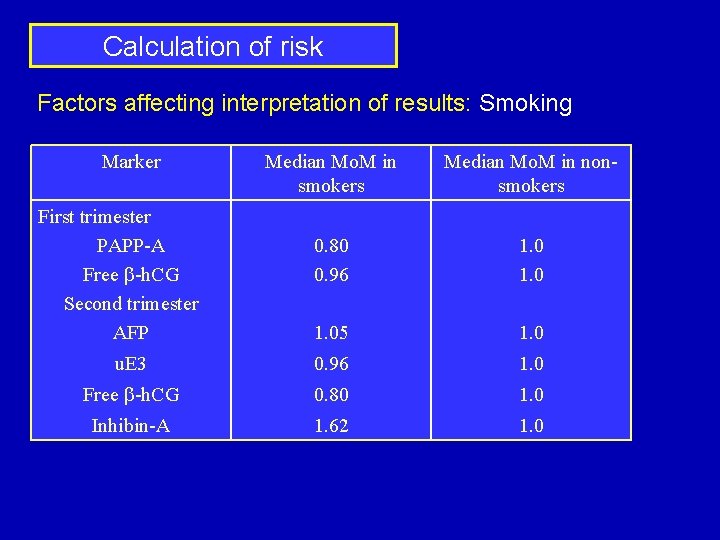
Calculation of risk Factors affecting interpretation of results: Smoking Marker Median Mo. M in smokers Median Mo. M in nonsmokers 0. 80 0. 96 1. 05 1. 0 u. E 3 0. 96 1. 0 Free b-h. CG 0. 80 1. 0 Inhibin-A 1. 62 1. 0 First trimester PAPP-A Free b-h. CG Second trimester AFP

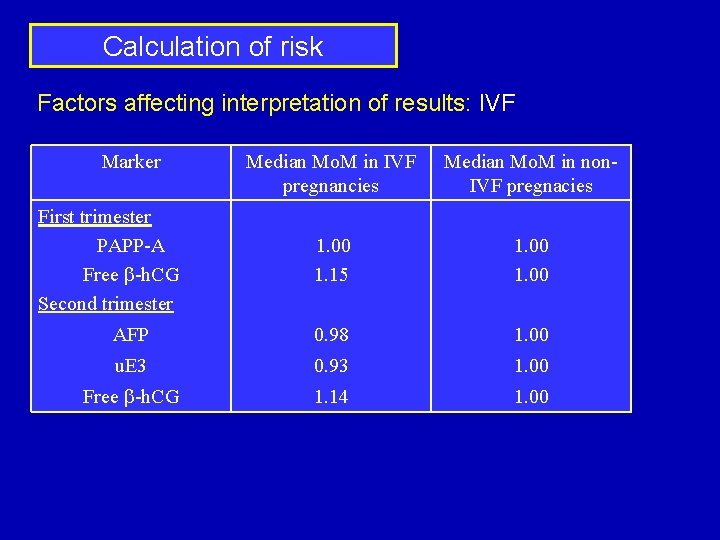
Calculation of risk Factors affecting interpretation of results: IVF Marker Median Mo. M in IVF pregnancies Median Mo. M in non. IVF pregnacies 1. 00 1. 15 1. 00 AFP 0. 98 1. 00 u. E 3 0. 93 1. 00 Free b-h. CG 1. 14 1. 00 First trimester PAPP-A Free b-h. CG Second trimester

Calculation of risk Factors affecting interpretation of results: Vaginal bleeding & amniocentesis

Calculation of risk Adjusting for factors affecting the serum markers Divide the observed Mo. M value by the expected Mo. M value for the particular factor warranting adjustment Example: Insulin dependent diabetes – Divide the observed AFP Mo. M value by the median AFP Mo. M value in IDDM pregnancies (expected Mo. M) – divide observed Mo. M by 0. 92 – Unadjusted Mo. M value of 0. 92 were observed, the IDDM adjusted Mo. M would be 1. 0 Mo. M – Unadjusted Mo. M value of 1. 0 were observed, the IDDM adjusted Mo. M would be 1. 09 Mo. M

Calculation of risk Adjusting for factors affecting the serum markers Divide the observed Mo. M value by the expected Mo. M value for the particular factor warranting adjustment Example: IVF – Divide the observed h. CG Mo. M value by the median h. CG Mo. M value in IVF pregnancies (expected Mo. M) – divide observed Mo. M by 1. 14 – Unadjusted h. CG Mo. M value of 1. 14 were observed, the IVF adjusted Mo. M would be 1. 0 Mo. M – Unadjusted h. CG Mo. M value of 1. 0 were observed, the IVF adjusted Mo. M would be 0. 88 Mo. M

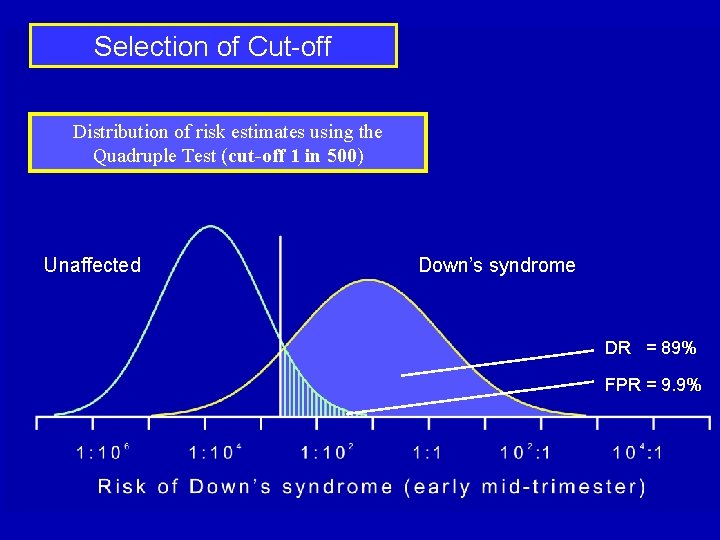
Selection of Cut-off Distribution of risk estimates using the Quadruple Test (cut-off 1 in 500) Unaffected Down’s syndrome DR = 89% FPR = 9. 9%

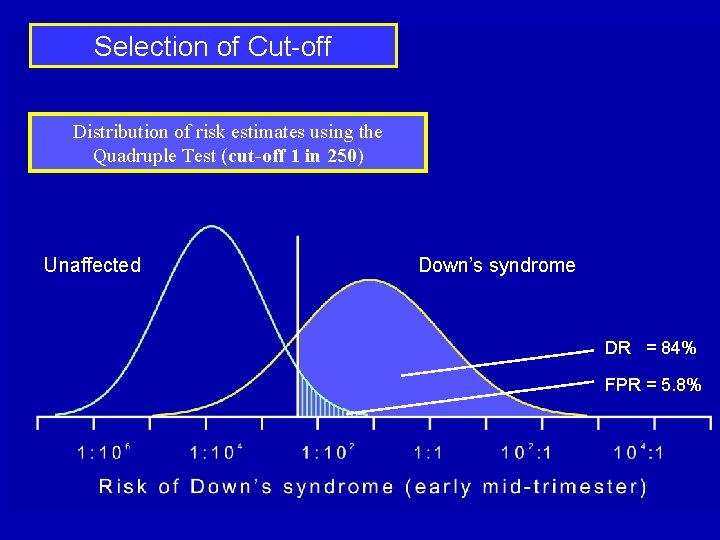
Selection of Cut-off Distribution of risk estimates using the Quadruple Test (cut-off 1 in 250) Unaffected Down’s syndrome DR = 84% FPR = 5. 8%

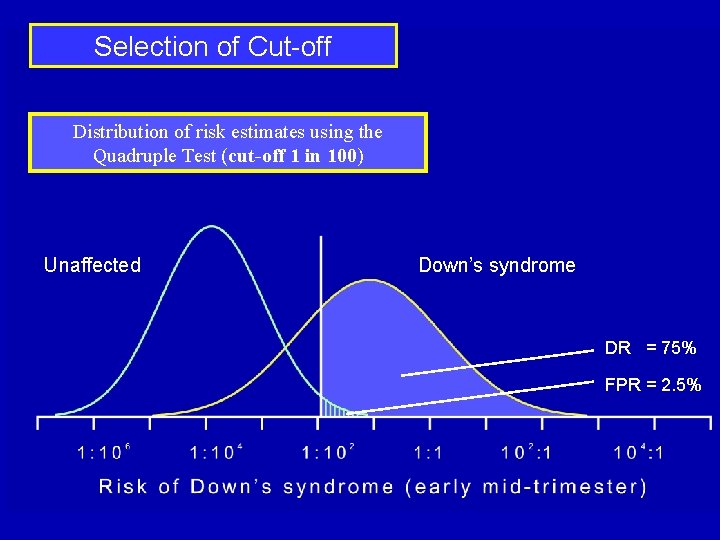
Selection of Cut-off Distribution of risk estimates using the Quadruple Test (cut-off 1 in 100) Unaffected Down’s syndrome DR = 75% FPR = 2. 5%

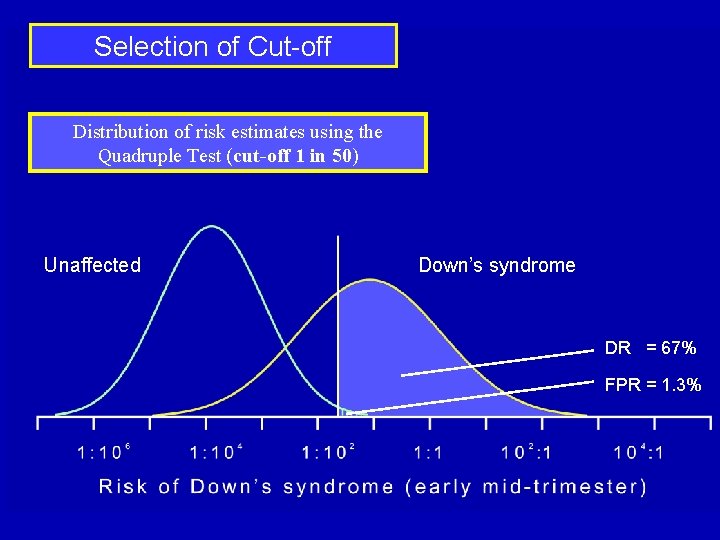
Selection of Cut-off Distribution of risk estimates using the Quadruple Test (cut-off 1 in 50) Unaffected Down’s syndrome DR = 67% FPR = 1. 3%

Reporting risk and performance at first trimester, second trimester or term? Historically first trimester screening programmes have reported risk and performance at time of screening (12 wks) and second trimester screening programmes have reported risk and performance at term.

Reporting risk and performance at first trimester, second trimester or term? Why does it matter what stage risk and performance are reported at? Natural fetal loss bias: Fetal loss between 1 st trimester and term is approx 43% Fetal loss between 2 nd trimester and term is approx 23% Marker related fetal loss bias: Down’s syndrome fetuses with, for example, very large NT measurements or very high h. CG levels are more likely to miscarry than those with average marker levels

Reporting risk and performance at first trimester, second trimester or term? Why does it matter what stage risk and performance are reported at? We can roughly adjust for natural fetal loss when calculating risk and performance, but it is very difficult to adjust for marker related fetal loss as data are scarce.

Reporting risk and performance at first trimester, second trimester or term? At what stage should we report risk and performance? As very few pregnancies with Down’s syndrome now reach term, we have little available data on term births. Recent large studies (SURUSS and FASTER) have provided us with information on the screening performance of the tests at second trimester, which will have taken account of the natural and marker related fetal loss from first to second trimester, but not to term. The most reliable risk and performance estimates we can therefore give are at second trimester. When screening in the first trimester, risk and performance should be adjusted to give second trimester estimates to provide consistency between programmes and to allow comparison.

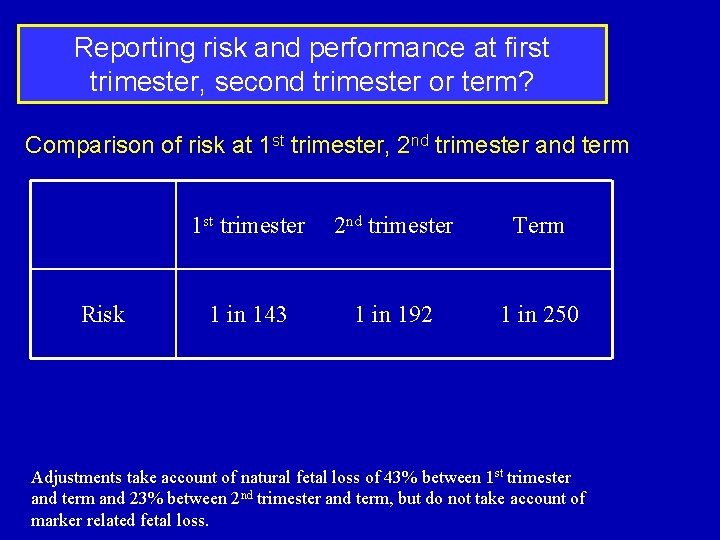
Reporting risk and performance at first trimester, second trimester or term? Comparison of risk at 1 st trimester, 2 nd trimester and term Risk 1 st trimester 2 nd trimester Term 1 in 143 1 in 192 1 in 250 Adjustments take account of natural fetal loss of 43% between 1 st trimester and term and 23% between 2 nd trimester and term, but do not take account of marker related fetal loss.

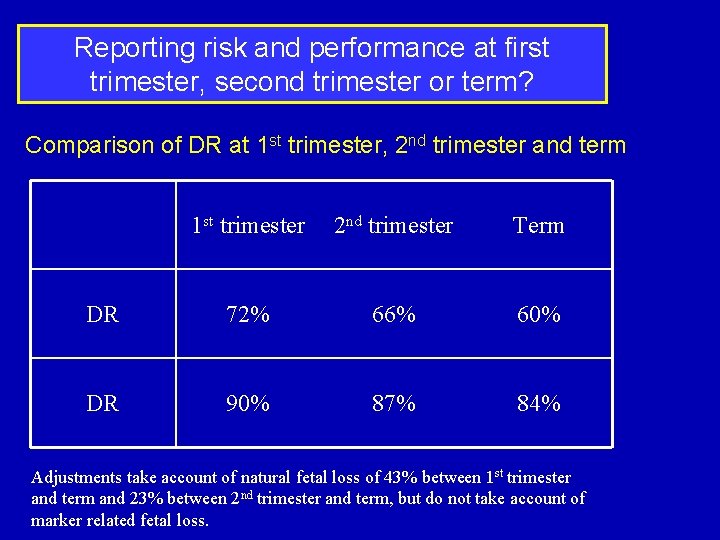
Reporting risk and performance at first trimester, second trimester or term? Comparison of DR at 1 st trimester, 2 nd trimester and term 1 st trimester 2 nd trimester Term DR 72% 66% 60% DR 90% 87% 84% Adjustments take account of natural fetal loss of 43% between 1 st trimester and term and 23% between 2 nd trimester and term, but do not take account of marker related fetal loss.

Timing of the tests First trimester Down’s syndrome screening possible between 10, 0 and 13, 6 Second trimester Down’s syndrome screening possible between 14, 0 and 22, 6 Open neural tube defect screening possible between 15, 0 and 22, 6