Principles Applications of BTEX Bioremediation Pedro J J

Principles & Applications of BTEX Bioremediation Pedro J. J. Alvarez, Ph. D. , P. E. , DEE University of Iowa


Prospectus § § What are BTEX and why care about them? What is needed to biodegrade them? How to exploit biodegradation for site cleanup? What are the more serious technical and political challenges related to BTEX bioremediation? § What is epistemology and how can it help us address some of these challenges?
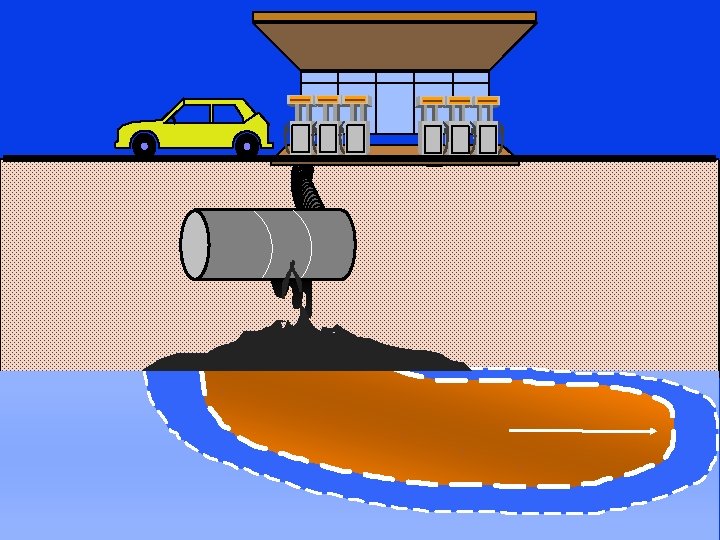

“Water, water everywhere, nor any drop to drink” The Rime of the Ancient Mariner, Samuel Taylor Coleridge

Contaminants of Concern: BTEX CH 2 CH 3 Benzene CH 3 Toluene CH 3 Ethylbenzene CH 3 o-Xylene m-Xylene CH 3 p-Xylene Importance: • Relatively high solubility = High migration potential • Toxicity: Benzene can cause leukemia at 5 µg/l • Volatile, hydrophobic, biodegradable

Requirements for Biodegradation 1. Existence of organism(s) with required catabolic potential. Xenobiotic will be degraded to an appreciable extent only if the organism has enzymes that catalyze its conversion to a product that is an intermediate or a substrate for common metabolic pathways. The greater the differences in structure between the xenobiotic and the constituents of living organisms (or the less common the xenobiotic building blocks are in living matter), the less likelihood of extensive transformation or the slower the transformation.

Requirements for Biodegradation (contd) 2. Presence of organism(s) in the environment. BTEX degraders are commonly found, but differences in relative abundance of dissimilar phenotypes may lead to apparent discrepancies in the biodegradability of a given BTEX compound at different sites. Depending on the relative abundance of different strains, B could degrade earlier than T at one site, but the opposite may be observed at other sites.
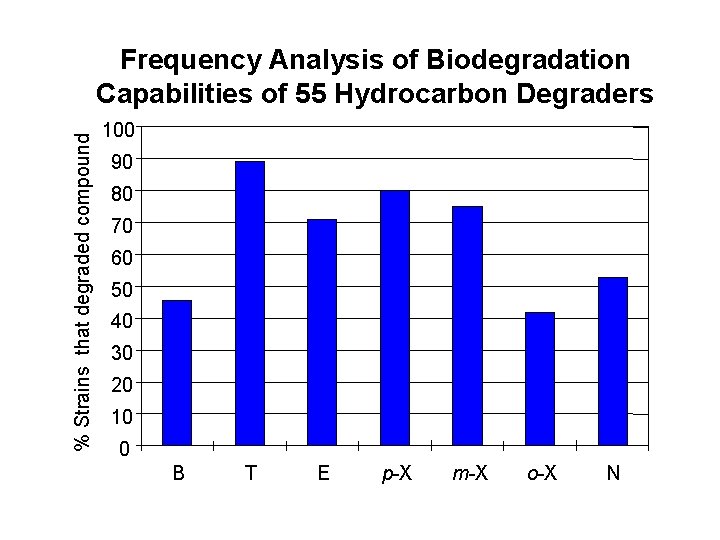
% Strains that degraded compound Frequency Analysis of Biodegradation Capabilities of 55 Hydrocarbon Degraders 100 90 80 70 60 50 40 30 20 10 0 B T E p-X m-X o-X N

Requirements for Biodegradation (contd) 3. Compound must be accessible to organism: a) Physicochemical aspects (bioavailability). Desorption, dissolution, diffusion, and mass transport b) Biochemical aspects. Membrane permeability (important for intracellular enzymes), uptake regulation.
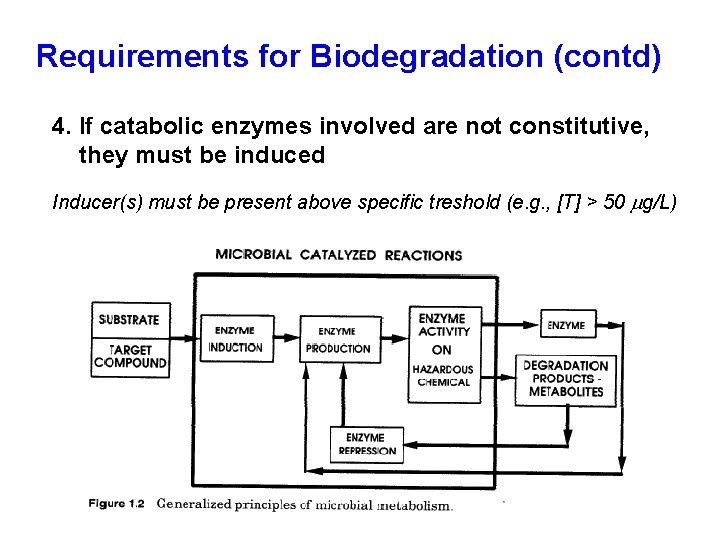
Requirements for Biodegradation (contd) 4. If catabolic enzymes involved are not constitutive, they must be induced Inducer(s) must be present above specific treshold (e. g. , [T] > 50 mg/L)
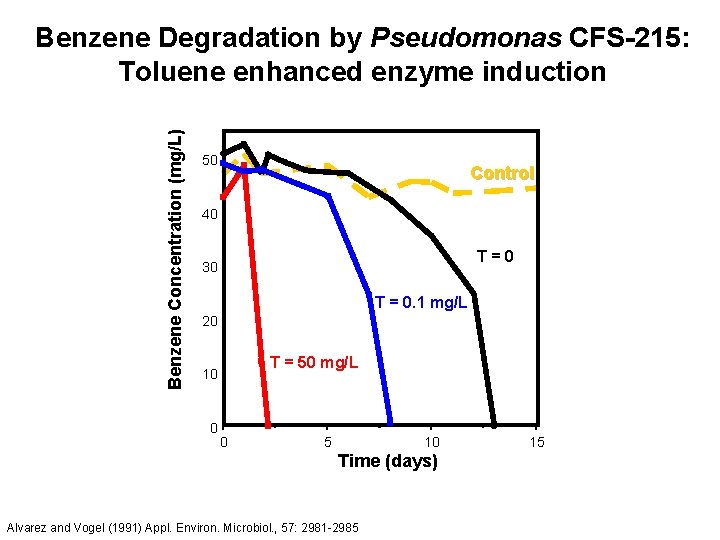
Benzene Concentration (mg/L) Benzene Degradation by Pseudomonas CFS-215: Toluene enhanced enzyme induction 50 Control 40 T=0 30 T = 0. 1 mg/L 20 T = 50 mg/L 10 0 0 5 10 Time (days) Alvarez and Vogel (1991) Appl. Environ. Microbiol. , 57: 2981 -2985 15
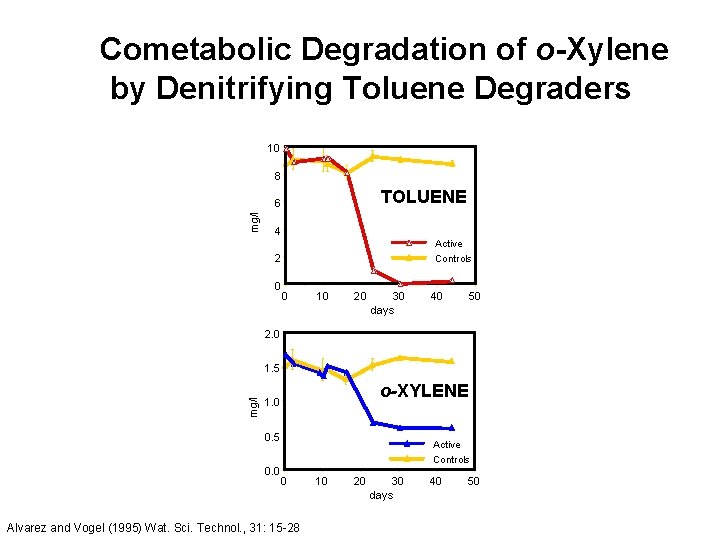
Cometabolic Degradation of o-Xylene by Denitrifying Toluene Degraders 10 8 TOLUENE mg/l 6 4 Active 2 0 Controls 0 10 20 30 days 40 50 2. 0 mg/l 1. 5 o-XYLENE 1. 0 0. 5 0. 0 Active Controls 0 Alvarez and Vogel (1995) Wat. Sci. Technol. , 31: 15 -28 10 20 30 days 40 50

Requirements for Biodegradation (contd) 5. Environment conducive to growth of desirable phenotypes and functioning of their enzymes: a) Presence of “recognizable” substrate(s) that can serve as energy and carbon source(s) (e. g. , the BTEX) and limiting nutrients (N and P, trace metals, etc. ). b) Moisture (80% of soil field capacity, or 15% H 2 O on a weight basis, is optimum for vadose zone remediation. Need at least 40% of field capacity). c) Availability of e- acceptors (e. g. , O 2 for oxidative reactions) or edonors (e. g. , H 2 for reductive transformations). The e- acceptor establishes metabolism mode and specific reactions.
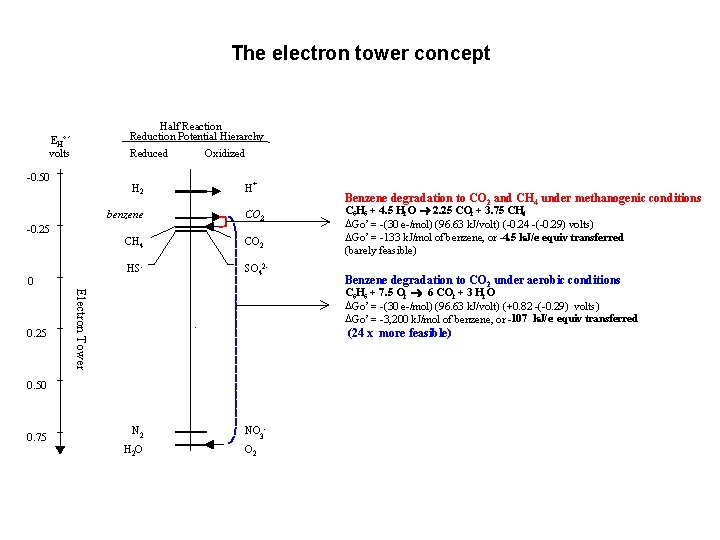
The electron tower concept Half Reaction Reduction Potential Hierarchy EH°´ volts Reduced -0. 50 H 2 -0. 25 + H benzene CO 2 CH 4 CO 2 HS- SO 42 - 0 C 6 H 6 + 4. 5 H 2 O ® 2. 25 CO 2 + 3. 75 CH 4 DGo’ = -(30 e-/mol) (96. 63 k. J/volt) (-0. 24 -(-0. 29) volts) DGo’ = -133 k. J/mol of benzene, or -4. 5 k. J/e- equiv transferred (barely feasible) Benzene degradation to CO 2 under aerobic conditions (24 x more feasible) 0. 50 0. 75 Benzene degradation to CO 2 and CH 4 under methanogenic conditions C 6 H 6 + 7. 5 O 2 ® 6 CO 2 + 3 H 2 O DGo’ = -(30 e-/mol) (96. 63 k. J/volt) (+0. 82 -(-0. 29) volts) DGo’ = -3, 200 k. J/mol of benzene, or -107 k. J/e- equiv transferred Electron Tower 0. 25 Oxidized N 2 H 2 O NO 3 O 2

Aerobic BTEX Degradation l BTEX are hydrocarbons (highly reduced) so their Oxidation to CO 2 is highly feasible thermodynamically (fuel) l Aerobic BTEX biodegradation is fast (O 2 diffusion is often rate-limiting) l Aerobic BTEX degraders are ubiquitous (e. g. , Pseudomonas) l Need oxygenase enzymes (i. e. , enzymes that “activate” O 2 and add it to carbon atoms in the BTEX molecule) l The ring must be dihydroxylated before ring fission. Once the ring is opened, the resulting fatty acids are readily metabolized further to CO 2.

Anaerobic BTEX Degradation l Rates are much slower because anaerobic electron acceptors NO 3 -, Fe+3, SO 4 -2, and CO 2) are not as strong oxidants as O 2. (e. g. , l Benzene, the most toxic of the BTEX, is recalcitrant under anaerobic conditions (i. e. , it degrades very slowly – after TEX, or not at all) l Anaerobic degradation mechanisms are not fully understood. Benzoyl-Co. A is a common intermediate, and it is reduced prior to ring fission by hydrolysis. The oxygen in the evolved CO 2 is from water. l Anaerobic BTEX degradation processes (e. g. , denitrifying, ironreducing, sulfidogenic, and methanogenic) are important natural attenuation mechanisms.
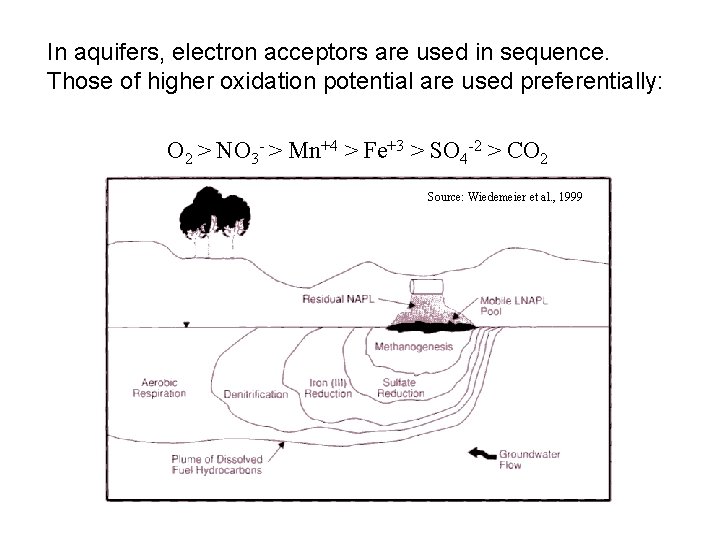
In aquifers, electron acceptors are used in sequence. Those of higher oxidation potential are used preferentially: O 2 > NO 3 - > Mn+4 > Fe+3 > SO 4 -2 > CO 2 Source: Wiedemeier et al. , 1999

Requirements for Biodegradation (contd) 5. Favorable environment (continued): d) Adequate temperature (rates double for ∆T = +10°C). e) Adequate p. H (6 -9). f) Absence/control of toxic substances (e. g. , precipitation of heavy metals, dilution of toxic conc. ). g) Absence of easily degradable, non-target substrates that could be preferentially metabolized (ethanol? ). 6. Time. Without engineered enhancement, benzene half-lives on the order of 100 days are common in aquifers. Want degradation rate > migration rate

What is Bioremediation? Ø It is a managed or spontaneous process in which biological, especially microbiological, catalysis acts on pollutants, thereby remedying or eliminating environmental contamination present in water, wastewater, sludge, soil, aquifer material, or gas streams. (a. k. a. biorestoration). l Ex Situ (Above ground) l In Situ (In its original place, below ground) Engineered Systems (biostimulation vs. bioaugmentation) Natural Attenuation (intrinsic/passive)

Why Use Bioremediation? · Can be faster and cheaper (at least 10 x less expensive than removal & incineration, or pump and treat) · Minimum land environmental disturbance (e. g. , generation of lesser volume of remediation wastes) · Can attack hard-to-withdraw hydrophobic pollutants · Done on site, eliminates transportation cost & liability · Environmentally sound (natural pathways) · Does not dewater the aquifer

When is engineered bioremediation feasible? Feasibility depends on: 2 Feasible log k; (per day) 1 with Enhancement 0 1) Kh distribution of nutrients and e- acceptors (Kh > 10 -5 m/s) Feasible 2) Adsorption bioavailability (depends on Kow and foc, problem for PAHs) -1 Not feasible -2 3) Potential degradation rate (half life < 10 days) -3 6 5 4 3 - log Kh (cm/s) 2 1

Bioventing l Used to bioremediate BTEX trapped above water table l Vacuum pumps pull air through unsaturated soil l Need to infiltrate water (with nutrients) to prevent desiccation Source: Mac. Donald and Rittmanm (1993) ES&T, 27(10) 1974 -1979

Water Circulation Systems l Used to bioremediate BTEX in saturated zone (Raymond) l Contaminated water is extracted, treated (air-stripping, activated carbon, or biodegradation), and recycled. l Some is amended with nutrients and reinjected (pulsing is better). l Clogging near injection well screens and infiltration galleries can be a problem (bacterial growth, mineral precipitation) but pulsing reduces clogging (may need occasional Cl 2, H 2 O 2)
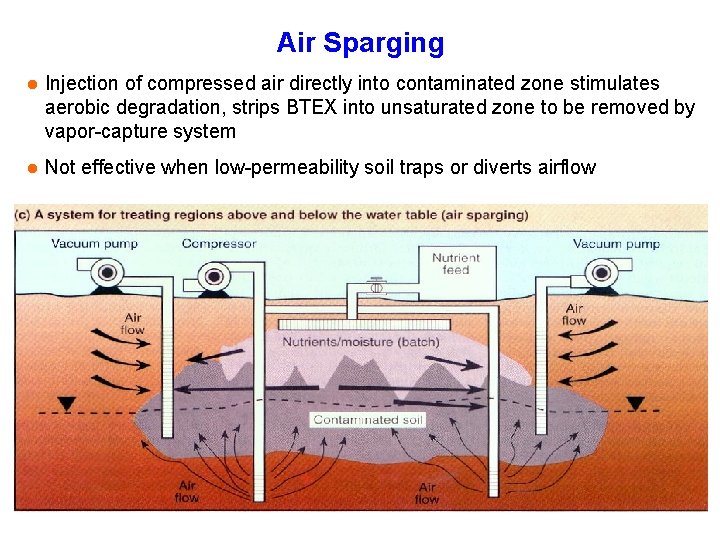
Air Sparging l Injection of compressed air directly into contaminated zone stimulates aerobic degradation, strips BTEX into unsaturated zone to be removed by vapor-capture system l Not effective when low-permeability soil traps or diverts airflow
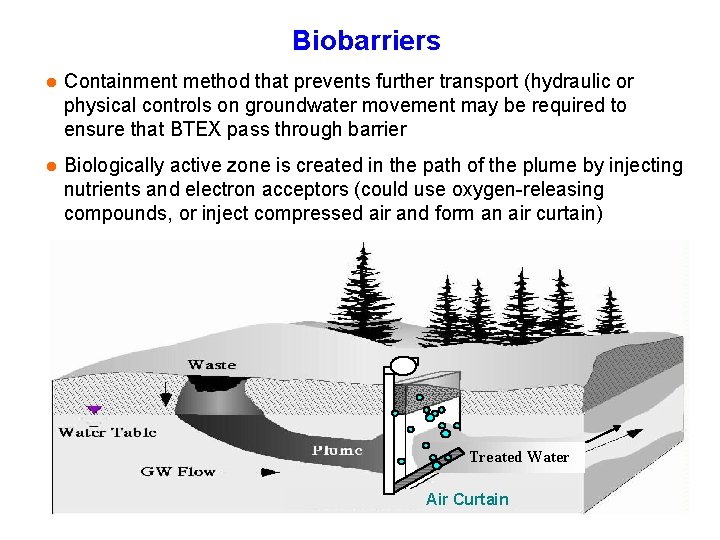
Biobarriers l Containment method that prevents further transport (hydraulic or physical controls on groundwater movement may be required to ensure that BTEX pass through barrier l Biologically active zone is created in the path of the plume by injecting nutrients and electron acceptors (could use oxygen-releasing compounds, or inject compressed air and form an air curtain) Treated Water Air Curtain
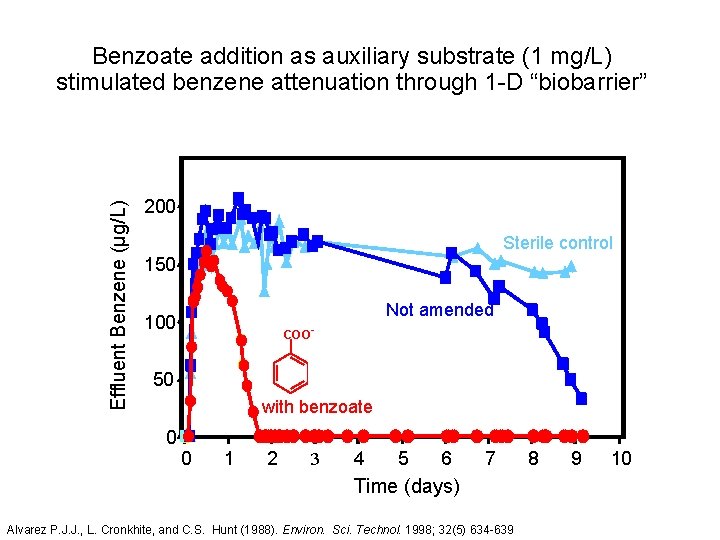
Effluent Benzene (µg/L) Benzoate addition as auxiliary substrate (1 mg/L) stimulated benzene attenuation through 1 -D “biobarrier” 200 Sterile control 150 Not amended 100 COO- 50 with benzoate 0 0 1 2 3 4 5 6 7 Time (days) Alvarez P. J. J. , L. Cronkhite, and C. S. Hunt (1988). Environ. Sci. Technol. 1998; 32(5) 634 -639 8 9 10

Bioremediation Market Ø According to the Organization for Economic Cooperation & Development), the global market potential for environmental biotechnology doubled in the past 10 years to $75 billions in the year 2000 Ø In USA, we have 400, 000 highly contaminated sites, and NRC estimates the cleanup cost to be on the order of $1, 000 billions Ø In USA, the current bioremediation market is only about $0. 5 billions

Bioremediation experienced many up- and downturns l 1950’s: Microbial infallibility hypothesis (Gayle, 1952) l 1970’s: Regulatory pressure stimulates development. Adding bacteria to contaminated sites becomes common practice. Failure to meet expectations (e. g. , DDT accumulation) prompts a major downturn. l 1980’s: It becomes clear that fundamental processes need to be understood before a successful technology can be designed. This realization, along with the fear of liability and Superfund, stimulates the blending of science and engineering to tackle environmental problems. l 1990’s: Many bioremediation and hybrid technologies are developed. However, decision makers insist on pump and treat, and Superfund is depleted. Poor cleanup record and high costs stimulate paradigm shift towards natural attenuation and RBCA.
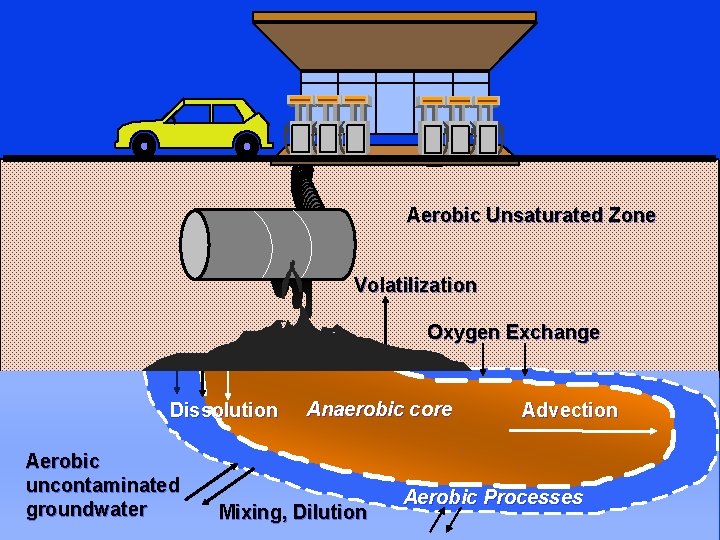
Aerobic Unsaturated Zone Volatilization Oxygen Exchange Dissolution Aerobic uncontaminated groundwater Anaerobic core Mixing, Dilution Advection Aerobic Processes

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea

Atenuação Natural PE Fluxo da água subterrânea
Plume Source

What is Monitored Natural Attenuation? ØMNA is the combination of natural biological, chemical and physical processes that act without human intervention to reduce the mass, toxicity, mobility, volume, or concentration of the contaminants (e. g. , biodegradation, dispersion, dilution, sorption, and volatilization). ØSuccess depends on adequate site characterization, a long-term monitoring plan consistent with the level of knowledge regarding subsurface conditions at the site, control of the contaminant source, and a reasonable time frame to achieve the objectives. ØMNA should not be a default technology or presumptive remedy. The burden of proof (e. g. , loss of contaminants at field scale, and geochemical foot-prints) should be on proponent, and evidence of its effectiveness should emphasize biodegradation.
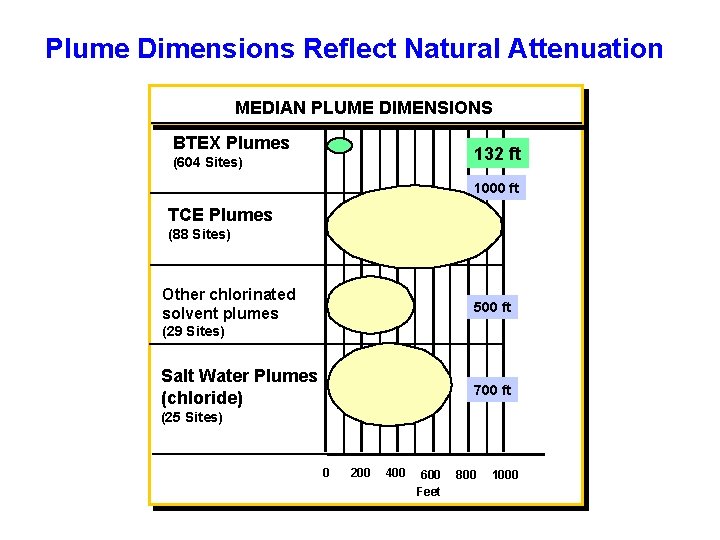
Plume Dimensions Reflect Natural Attenuation MEDIAN PLUME DIMENSIONS BTEX Plumes 132 ft (604 Sites) 1000 ft TCE Plumes (88 Sites) Other chlorinated solvent plumes 500 ft (29 Sites) Salt Water Plumes (chloride) 700 ft (25 Sites) 0 200 400 600 Feet 800 1000

Concentration “safe”
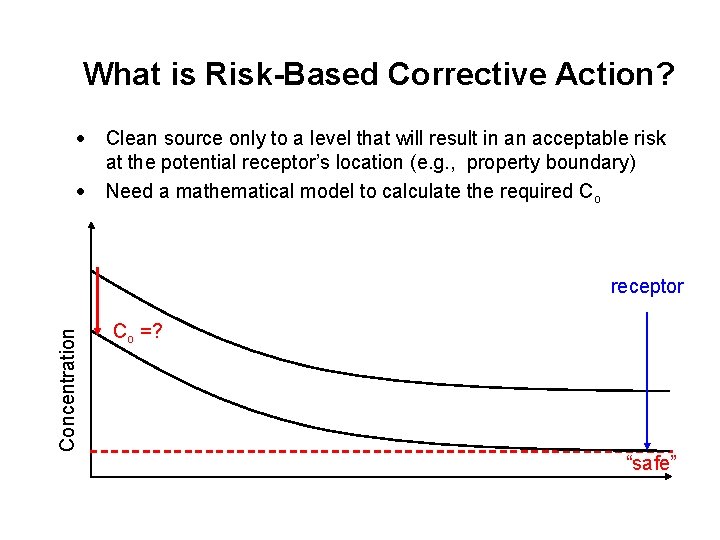
What is Risk-Based Corrective Action? · · Clean source only to a level that will result in an acceptable risk at the potential receptor’s location (e. g. , property boundary) Need a mathematical model to calculate the required Co Concentration receptor Co =? “safe”

Analytical Solution of the Advection-Dispersion-Sorption Equation with First-Order Decay, for Constant Rectangular Source (Domenico, 1987) 1/ 2 é ( y -Y / 2) ùüïìï é (Z ) ù é (-Z ) ùüï æ C o ö é x - vt (1+ 4 l a x / v) ùìï é ( y +Y / 2)ù ê ú C(x, t) = ç ÷erfcê erf erf úí ê ê 1/ 2 1 / 2 ýí 1/ 2 ú 1 / 2 úý è 8 ø ëê êë 2(a y x) úûïþïî ëê 2 (a z x) ûú 2(a x vt) ûúïî êë 2 (a y x) úû ëê 2(a z x) ûúïþ ìïæ x öé æ 4 l a ö 1 / 2ù üï x ÷÷ê 1 - ç 1+ ÷ ú ý exp[- k s (t - x / v)] exp íçç v ø ûú ïþ ïîè 2 a x øëê è Ø Models are useful analytical tools, and can be used to demonstrate that natural attenuation is occurring Ø Limited predictive capability (order-of-magnitude accuracy): groundwater flow and microbial behavior rarely follow simplifying assumptions.
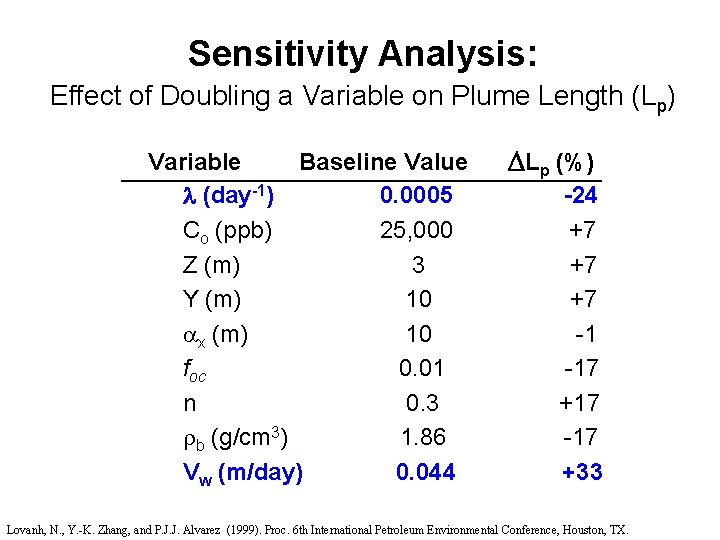
Sensitivity Analysis: Effect of Doubling a Variable on Plume Length (Lp) Variable Baseline Value (day-1) 0. 0005 Co (ppb) 25, 000 Z (m) 3 Y (m) 10 x (m) 10 foc 0. 01 n 0. 3 b (g/cm 3) 1. 86 Vw (m/day) 0. 044 Lp (%) -24 +7 +7 +7 -1 -17 +17 -17 +33 Lovanh, N. , Y. -K. Zhang, and P. J. J. Alvarez (1999). Proc. 6 th International Petroleum Environmental Conference, Houston, TX.
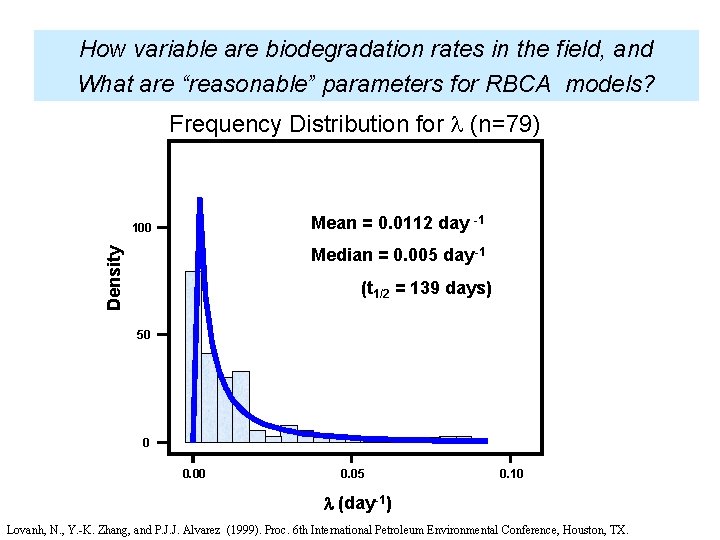
How variable are biodegradation rates in the field, and What are “reasonable” parameters for RBCA models? Frequency Distribution for (n=79) Mean = 0. 0112 day -1 100 Density Median = 0. 005 day-1 (t 1/2 = 139 days) 50 0 0. 05 0. 10 (day-1) Lovanh, N. , Y. -K. Zhang, and P. J. J. Alvarez (1999). Proc. 6 th International Petroleum Environmental Conference, Houston, TX.

Current Status of Bioremediation l We have made significant advances towards understanding the biochemical and genetic basis for biodegradation. However, bioremediation is still an underutilized technology. l Bioremediation is not universally understood, or trusted by those who must approve it. To take full advantage of its potential, we need to communicate better the capabilities and limitations of bioremediation, and answer: n What is being done in the subsurface, Why, How, and Who is doing what? n How fast is it being done, and can we control it and make it go faster? n When can we meet cleanup standards in a cost-effective manner? n Can we reasonably predict that we want to happen, will happen?

EPISTEMOLOGY OF BIOREMEDIATION episteme = knowledge Theory of the method and basis we use to acquire knowledge, including the possibility and opportunity to advance fundamental understanding, sphere of action, and the philosophy of the scientific disciplines that we rely upon. Reductionism: l System analysis through separation of its components (eliminates complexity to enhance interpretation). l Based on the premise that a system can be known by studying its components, and that an idea can be understood if we understand its concepts separately. l Used increasingly in bioremediation research to investigate mechanisms. Holism: l The totality of a system is greater than the sum of its parts (synergism & antagonism)
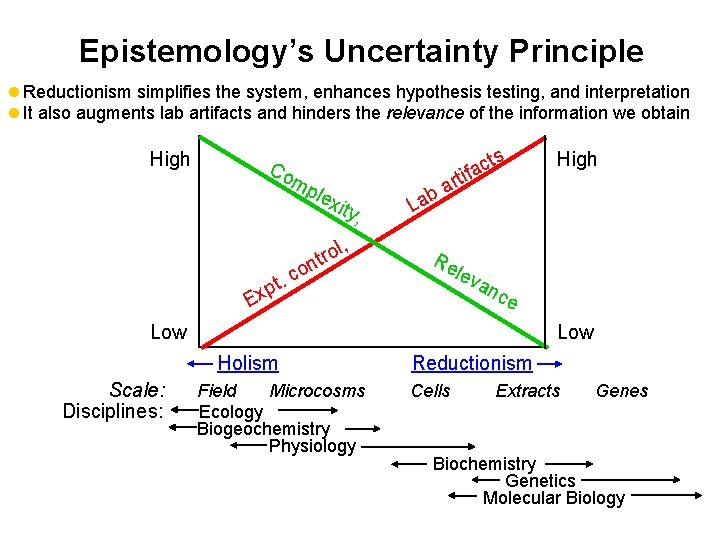
Epistemology’s Uncertainty Principle l Reductionism simplifies the system, enhances hypothesis testing, and interpretation l It also augments lab artifacts and hinders the relevance of the information we obtain High Co mp lex ity, l, Ex pt. o ntr co b La ts c a rtif a Re lev an ce Low Holism Scale: Disciplines: High Field Microcosms Ecology Biogeochemistry Physiology Reductionism Cells Extracts Genes Biochemistry Genetics Molecular Biology

Implications l Quantitative extrapolation from the lab to the field is taboo. (interpolate but do not extrapolate) l Rely more on holistic disciplines (e. g. , ecology, biogeochemistry) and iterate more between the field and the lab, between basic and applied research. l Multidisciplinary Research (interstices) l Aurea mediocridad (San Ignacio de Loyola)

Pay attention to detail. You never know who is watching your work, and where your next promotion or demotion will come from. · Bioremediation is seldom a straight line to an imagined goal (many branching decision points requiring flexibility and versatility) · Remedial technologies are rapidly evolving. Be committed to life-long learning, and be aware that imagination and creativity could more important than knowledge

Conclusions ØIndigenous microorganisms can often destroy BTEX and other common groundwater contaminants, making bioremediation (often) technically feasible. ØThe pendulum recently swung towards natural attenuation. This can save money but take much longer to achieve cleanup and appear as if officials are walking away from contaminated sites. Early public involvement is critical to minimize such controversy.

Lets Take a Break!

TYPES OF MICROBES USED A. Indigenous Microorganisms Used in most applications (99%) Pseudomonas have wide catabolic capacity May need to enhance proliferation/enzyme induction B. Acclimated Strains Preselected naturally occurring bacteria Generally not needed for BTEX Often fail to function in situ; common reasons: - Conc. of target compound too low to support growth - Other substances and organisms inhibit growth - Microbe uses other food than target contaminant - Target compound not accessible to microbe C. Genetically Engineered Microbes (GEMs) Could combine desirable traits from different microbes: - Ability to withstand stress & degrade recalcitrant compounds - Not needed for BTEX, many technical & political constraints



Análisis de varianza de las interacciones BTEXN l Las capacidades de degradación fueron mas amplias cuando los BTEXN fueron alimentados como mezcla que separadamente (particularmente cuando el T estaba presente) l Las interacciones negativas (e. g. , inhibición competitiva, toxicidad) fueron estadísticamente significativas cuando se alimentó 1 mg/L a cada una. l Por estadística de Kappa se encontró una correlación significativa entre las habilidades para degradar T y E, p-X y m-X, y p-X y o-X. La falla de degradar B fue correlacionada con la inhabilidad para degradar o-X.

Specific degradation rate d. C/dt/X Monod’s Equation k k 2 k. CX d. C =KS + C dt KS Contaminant Concentration, C

Why First-Order Degradation Rates? Monod’s Equation, When C << KS d. C= - k XC - k X » C + KS dt KS C d. C= - C dt k. X = K S (not constant)
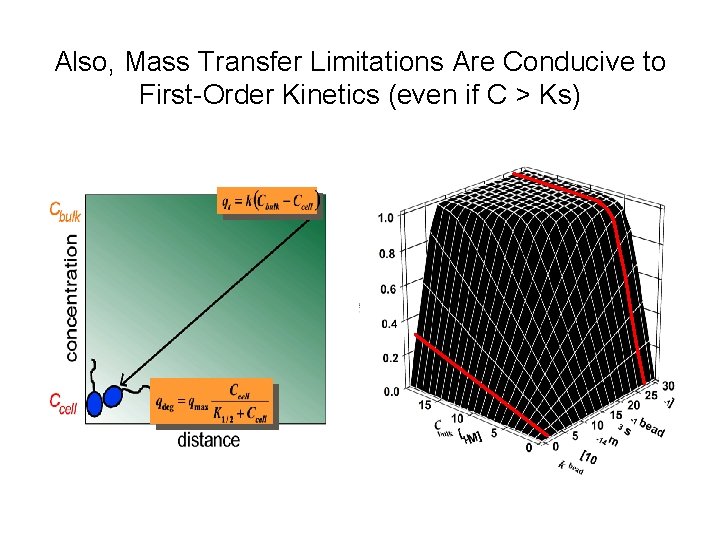
Also, Mass Transfer Limitations Are Conducive to First-Order Kinetics (even if C > Ks)

Alta concentración microbiana = Taza más rápida Simulaciones empleadas: k = 0. 28 g-T/g-células/día KS = 8. 6 mg-T/L Y = 0. 6 g- células/g-T TOLUENO (mg/L) 80 60 107 células/m. L 40 102 células/m. L 20 0 0 30 60 90 120 Tiempo (Días) 150
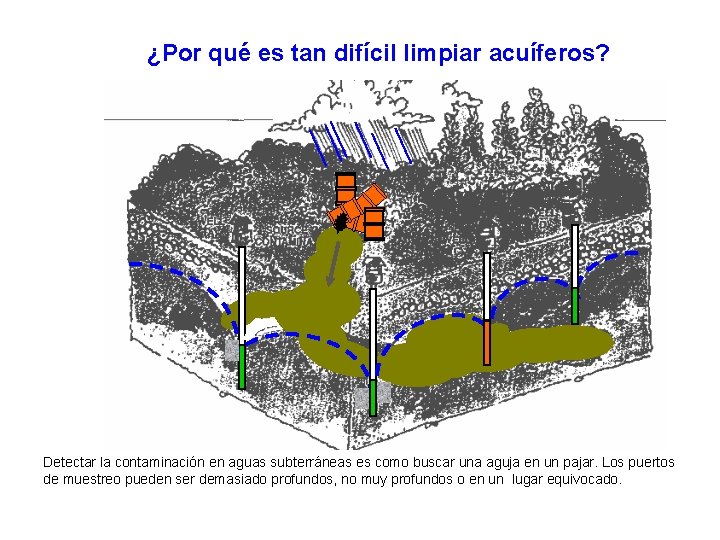
¿Por qué es tan difícil limpiar acuíferos? Detectar la contaminación en aguas subterráneas es como buscar una aguja en un pajar. Los puertos de muestreo pueden ser demasiado profundos, no muy profundos o en un lugar equivocado.
- Slides: 74