Principle and Applications of Complexometric titration Bhakti Ramesh

Principle and Applications of Complexometric titration Bhakti Ramesh Bhadra MSc I Environmental Sciences Roll no. 1 Sem 2 K. J Somaiya Clg. Of Sci. and Comm.

Table of Contents � Introduction � Ligands � Complexing Agents � Masking and Demasking � Complex titration with EDTA � Indicators � Regions of EDTA Titration � Factors affecting the Stability of Complexes � Factors important in determining the magnitude of break in titration curve � Applications � EDTA Applications � References

Introduction �A titration based on the formation of a coordination complex is known as a complexometric titration. �Complex formation titrations are used to titrate cations via complex formation reagents. �The complex can form only when… 1. The central atom (a metal ion (or cation) in a complex) accepts an electron pair from one or more ligands (ligand = electron-pair donating species). 2. The ligand possesses at least one electron pair to donate. 3. The bonding (coordinate covalent bonding) occurs.

Ligands � Molecules/ Ions which can donate atleast one pair of electrons. � Types of LigandsØ Unidentate- ammonia, hydroxide ion Ø Bidentate- ethylenediamine, oxalate ion Ø Polydentate- EDTA � Chelation is a process in which a polydentate ligand bonds to a metal ion, forming a ring. The complex produced by this process is called a chelate, and the polydentate ligand is referred to as a chelating agent.
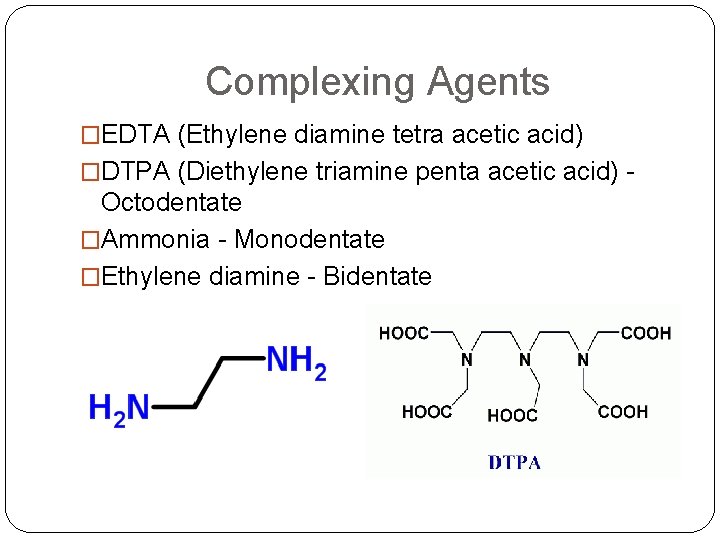
Complexing Agents �EDTA (Ethylene diamine tetra acetic acid) �DTPA (Diethylene triamine penta acetic acid) - Octodentate �Ammonia - Monodentate �Ethylene diamine - Bidentate

Masking and Demasking �Masking is without the removal of a particular substance it is so transformed that it doesn’t take part in chemical reaction. Ø Effective masking agent is Cyanide Ø Cd, Zn, Cu, Co, Ni Ø Not with alkaline earth metals, Manganese and lead �Demasking is process in which masked substance regains its ability to enter in reaction. Ø Zn and Cd complex can be demasked by formaldehyde
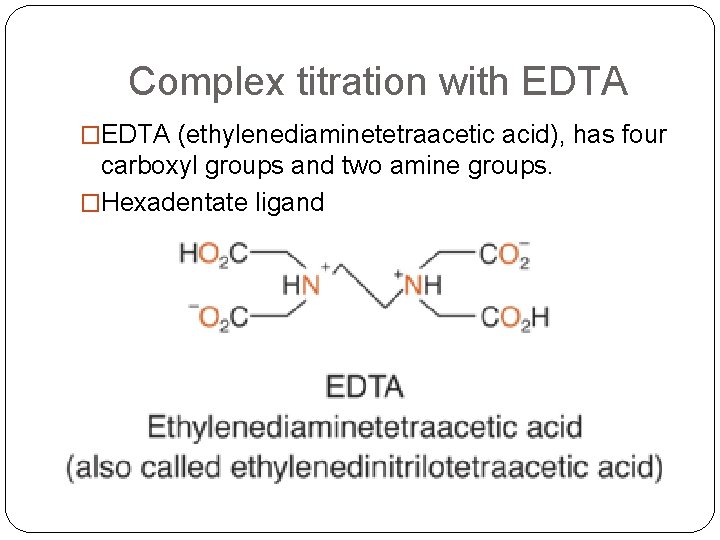
Complex titration with EDTA �EDTA (ethylenediaminetetraacetic acid), has four carboxyl groups and two amine groups. �Hexadentate ligand

Indicators Colorimetric indicators � Also known as metallochromic indicators. � The color change occurs when the metal ion is bound with the indicator. Metal ion + Indicator → Complex (Chelate) + Indicator Ca/ Mg + EBT → Ca + EDTA + EBT � This binding is p. H dependent. � Masking agents are often used for complexometric titrations, which allow for the removal of interferents. Metal Ion Indicators � Over 200 organic compounds form colored chelates with ions in a p. M range that is unique to the cation and the dye selected. � To be useful, the dye-metal chelates usually will be visible at 10 -6 -10 -7 M concentration. � The dye is selected such that the color change corresponds to the p. M at equivalence.
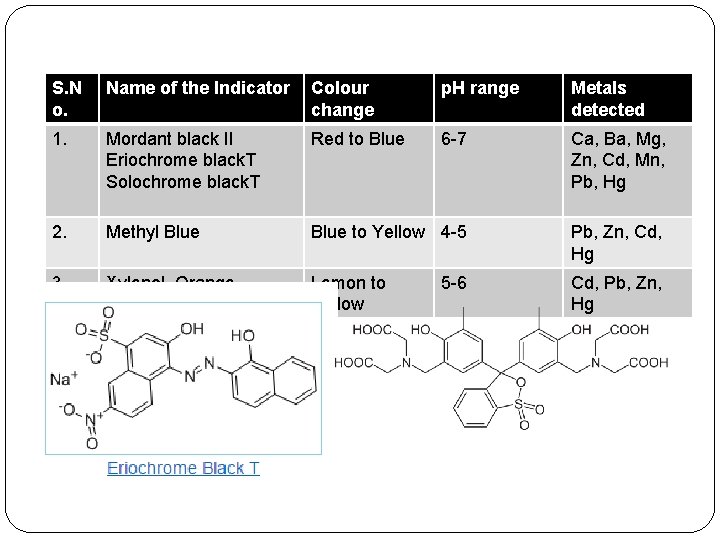
S. N o. Name of the Indicator Colour change p. H range Metals detected 1. Mordant black II Eriochrome black. T Solochrome black. T Red to Blue 6 -7 Ca, Ba, Mg, Zn, Cd, Mn, Pb, Hg 2. Methyl Blue to Yellow 4 -5 Pb, Zn, Cd, Hg 3. Xylenol Orange Lemon to Yellow Cd, Pb, Zn, Hg 5 -6
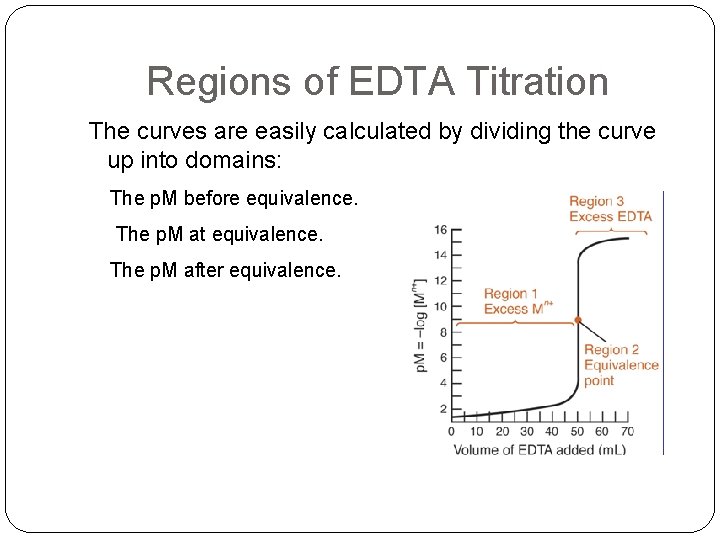
Regions of EDTA Titration The curves are easily calculated by dividing the curve up into domains: The p. M before equivalence. The p. M at equivalence. The p. M after equivalence.

Factors affecting the Stability of Complexes 1. Nature of Metal Ionsa) b) c) 2. Nature of Ligandsa) b) 3. Ionic size Ionic charge The availability suitable energy levels to accommodate the accepted electrons Greater the base strength of ligand, stabler is the complex Concentration of ligand p. H on complex formation. Control of p. H is important since the H+ ion plays an important role in chelation. 4. The presence of interfering ions such as CNCitrate, Tartrate, F-and other complex forming agents.

Factors important in determining the magnitude of break in titration curve 1. The stability of complex formed: ↑stability constant → ↑ charge in free metal conc. → ↑ clear the of complex @ eq. pt. end pt. 2. The number of steps involved in complex formation: Fewer the number of steps required in the formation of complex, greater would be the break in titration curve at equivalent point and clear would be the end point. ↓ the no. of steps → ↑ break in titration → ↑ clear the curve @ eq. pt. end pt. 3. Effect of p. H: ↓p. H → ↓ Stability of the complex

Applications v. Back titration �can be performed for the determintion of several metal ions can not be titrated directly but form stable EDTA complexes. �The procedure, a known amount of EDTA is added to the analyte sample solution and the excess is back titrated with a standard solution of “weak” metal ion, Mg 2+. �The weak metal ion will not displace the analyte from its EDTA complex. �Calgamite can be used as an indicator for the back titration of the excess EDTA with standard

v. Direct Titration �Many metals can be determined by direct titrations with EDTA. �Weak metal complexes such as Ca 2+ and Mg 2+ should be titrated in basic solution using EBT, Calmagite, or Arsenazo I as the indicator. v. Displacement Titration �Mg. Y 2 - or Zn. Y 2 - complex is added to the solution of unknown metal ion composition. �The unknown metal displaces the Mg 2+ or Zn 2+, which is then back titrated. �The technique only works when the unknown metal has tighter binding to EDTA than the Zn 2+ or Mg 2+.

EDTA Applications �Testing of Calcium content in urine sample �Determination of Cobalt in paint driers �Determination of Magnesium in fertilizers �Determination of Hardness of water

References � http: //www. srmuniv. ac. in/sites/default/files/downloads/ Complexometric_Titration. pdf 04/03/2017 � https: //www. slideshare. net/obydullah/complexometrictitrations 04/03/2017 � https: //www. researchgate. net/publication/242713763_ Theoretical_Basis_of_Analysis_Complexometric_Titra tions 04/03/2017 � http: //download. fa. itb. ac. id/filenya/Handout%20 Kuliah/ Inorganic%20 Pharmaceutical%20 Analysis%202008/E nglish%20 Version/05 a. %20 COMPLEXOMETRIC%20 TITRATION%20 Application. pdf 04/03/2017 � https: //www. coursehero. com/file/6936020/Applications -of-Complexometric-Titrations/ 04/03/2017
- Slides: 16