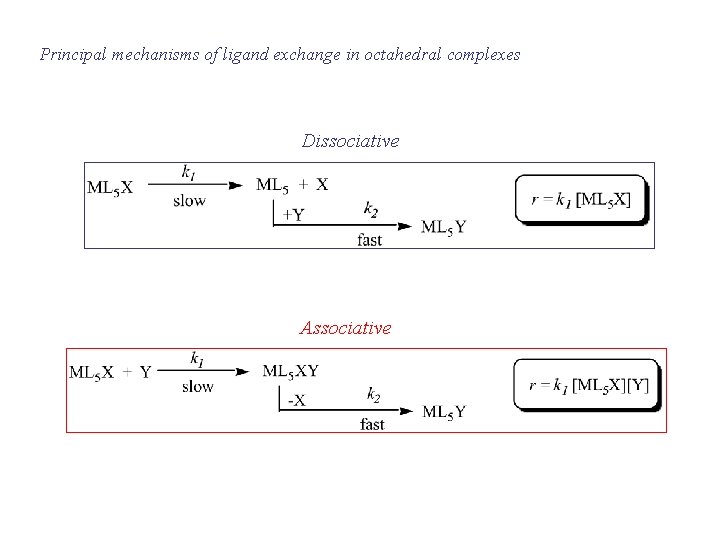
Principal mechanisms of ligand exchange in octahedral complexes


![Outer sphere mechanism [Fe(CN)6]4 - + [Ir. Cl 6]2[Co(NH 3)5 Cl]2+ + [Ru(NH 3)6]2+ Outer sphere mechanism [Fe(CN)6]4 - + [Ir. Cl 6]2[Co(NH 3)5 Cl]2+ + [Ru(NH 3)6]2+](https://slidetodoc.com/presentation_image_h/b901c8b10d66e373305d325ca86f6133/image-12.jpg)
![Inner sphere mechanism [Co(NH 3)5 Cl)]2+ + [Cr(H 2 O)6]2+ [Co(NH 3)5 Cl)]2+: : Inner sphere mechanism [Co(NH 3)5 Cl)]2+ + [Cr(H 2 O)6]2+ [Co(NH 3)5 Cl)]2+: :](https://slidetodoc.com/presentation_image_h/b901c8b10d66e373305d325ca86f6133/image-13.jpg)














- Slides: 47

Principal mechanisms of ligand exchange in octahedral complexes Dissociative Associative

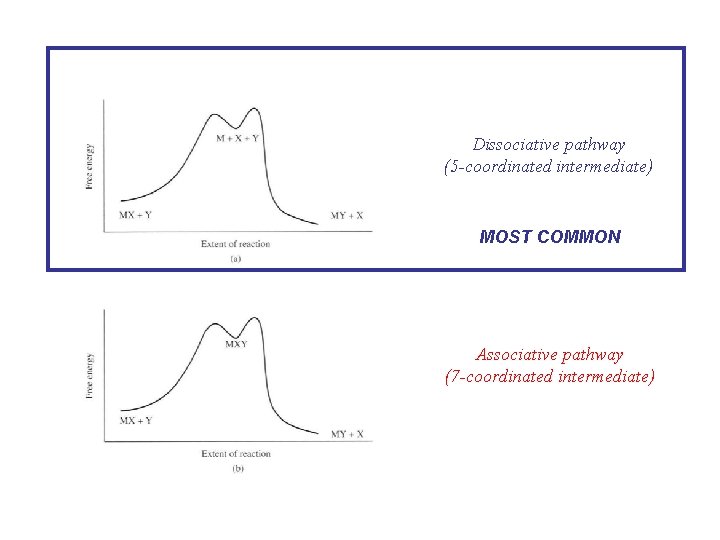
Dissociative pathway (5 -coordinated intermediate) MOST COMMON Associative pathway (7 -coordinated intermediate)

Experimental evidence for dissociative mechanisms Rate is independent of the nature of L

Experimental evidence for dissociative mechanisms Rate is dependent on the nature of L

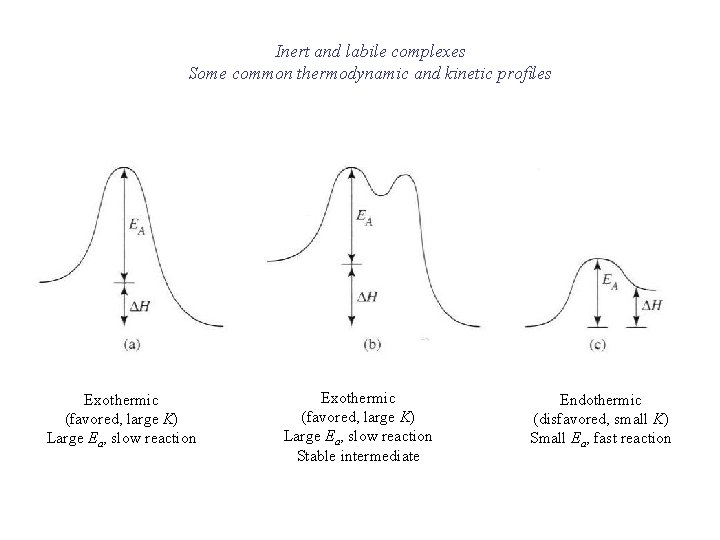
Inert and labile complexes Some common thermodynamic and kinetic profiles Exothermic (favored, large K) Large Ea, slow reaction Stable intermediate Endothermic (disfavored, small K) Small Ea, fast reaction

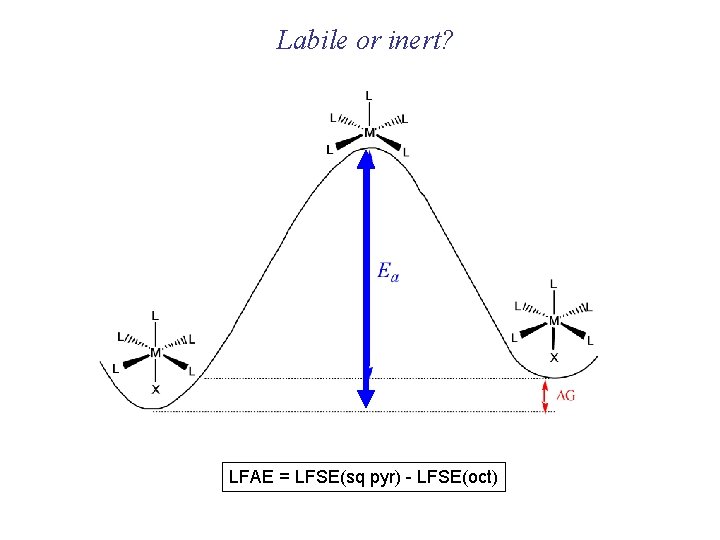
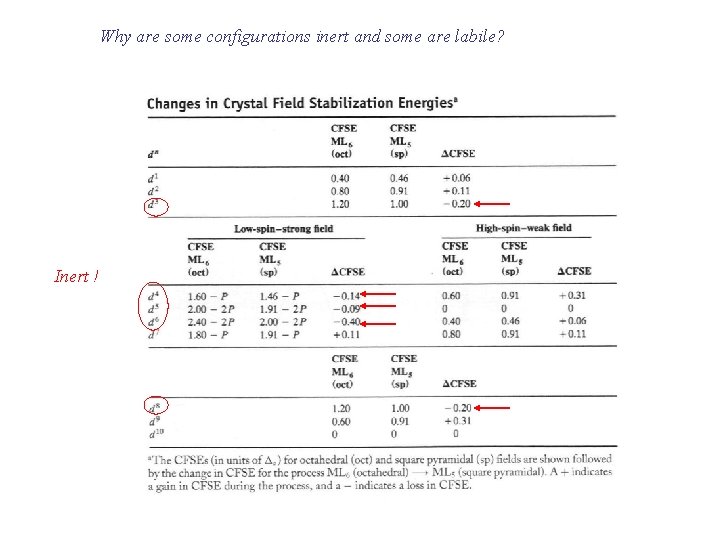
Labile or inert? LFAE = LFSE(sq pyr) - LFSE(oct)

Why are some configurations inert and some are labile? Inert !

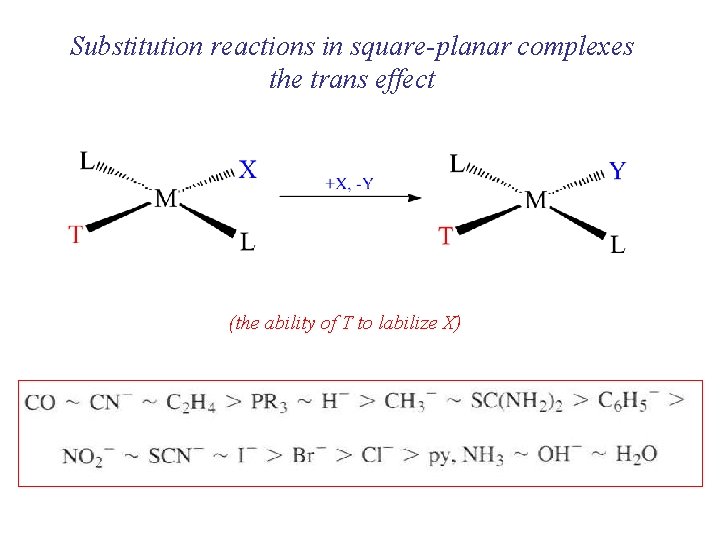
Substitution reactions in square-planar complexes the trans effect (the ability of T to labilize X)

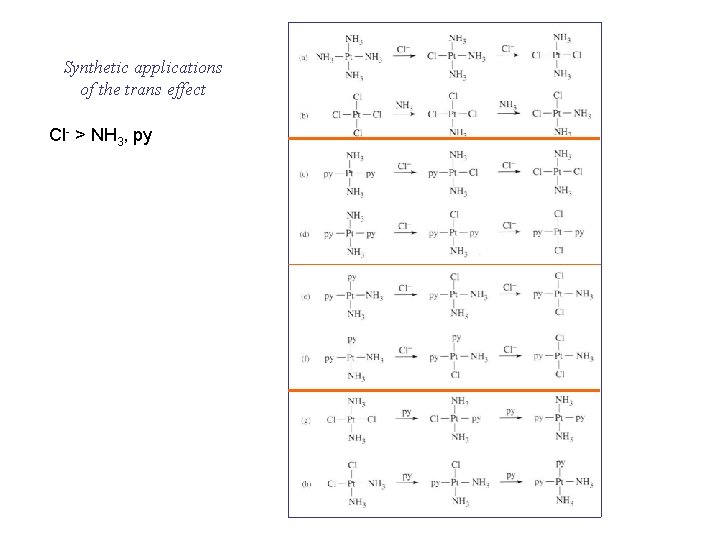
Synthetic applications of the trans effect Cl- > NH 3, py

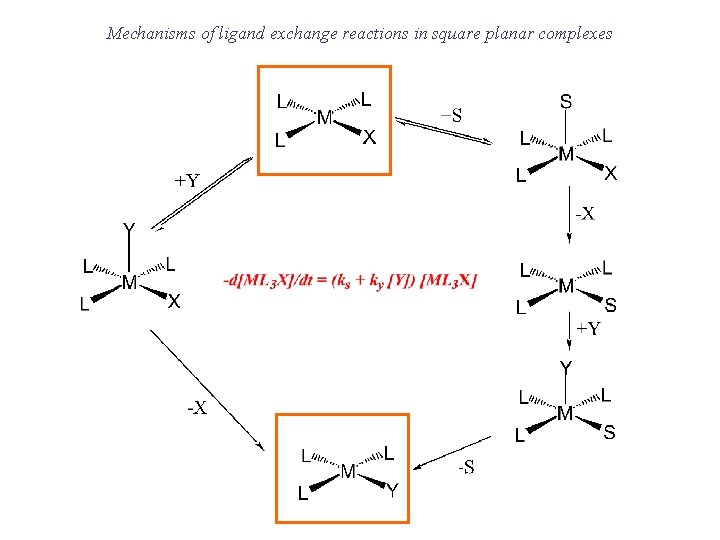
Mechanisms of ligand exchange reactions in square planar complexes

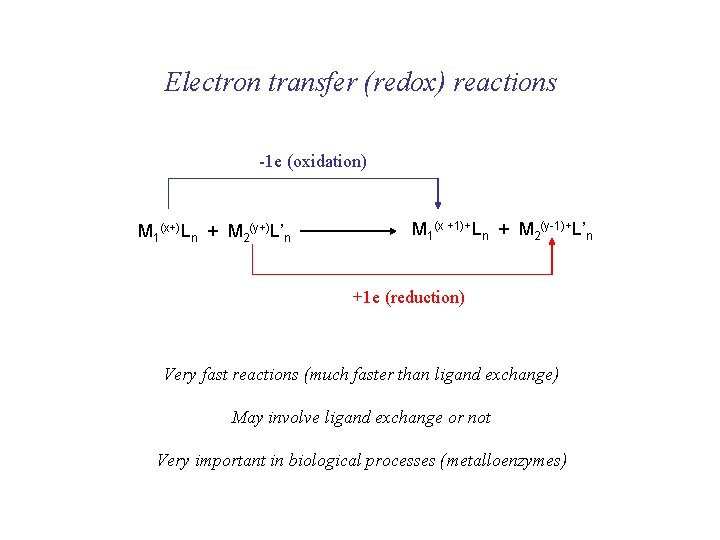
Electron transfer (redox) reactions -1 e (oxidation) M 1(x+)Ln + M 2(y+)L’n M 1(x +1)+Ln + M 2(y-1)+L’n +1 e (reduction) Very fast reactions (much faster than ligand exchange) May involve ligand exchange or not Very important in biological processes (metalloenzymes)
![Outer sphere mechanism FeCN64 Ir Cl 62CoNH 35 Cl2 RuNH 362 Outer sphere mechanism [Fe(CN)6]4 - + [Ir. Cl 6]2[Co(NH 3)5 Cl]2+ + [Ru(NH 3)6]2+](https://slidetodoc.com/presentation_image_h/b901c8b10d66e373305d325ca86f6133/image-12.jpg)
Outer sphere mechanism [Fe(CN)6]4 - + [Ir. Cl 6]2[Co(NH 3)5 Cl]2+ + [Ru(NH 3)6]2+ Reactions ca. 100 times faster than ligand exchange (coordination spheres remain the same) r = k [A][B] Tunneling mechanism [Fe(CN)6]3 - + [Ir. Cl 6]3[Co(NH 3)5 Cl]+ + [Ru(NH 3)6]3+
![Inner sphere mechanism CoNH 35 Cl2 CrH 2 O62 CoNH 35 Cl2 Inner sphere mechanism [Co(NH 3)5 Cl)]2+ + [Cr(H 2 O)6]2+ [Co(NH 3)5 Cl)]2+: :](https://slidetodoc.com/presentation_image_h/b901c8b10d66e373305d325ca86f6133/image-13.jpg)
Inner sphere mechanism [Co(NH 3)5 Cl)]2+ + [Cr(H 2 O)6]2+ [Co(NH 3)5 Cl)]2+: : : [Cr(H 2 O)6]2+ [Co. III(NH 3)5(m-Cl)Cr. II(H 2 O)6]4+ [Co. II(NH 3)5(m-Cl)Cr. III(H 2 O)6]4+ [Co. II(NH 3)5(H 2 O)]2+ + [Cr. III(H 2 O)5 Cl]2+ [Co(H 2 O)6]2+ + 5 NH 4+

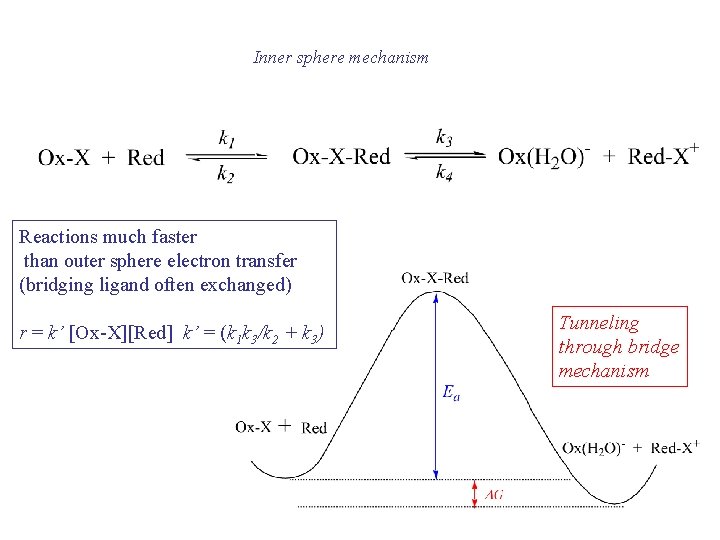
Inner sphere mechanism Reactions much faster than outer sphere electron transfer (bridging ligand often exchanged) r = k’ [Ox-X][Red] k’ = (k 1 k 3/k 2 + k 3) Tunneling through bridge mechanism

Brooklyn College Chem 76/76. 1/710 G Advanced Inorganic Chemistry (Spring 2008) Unit 6 Organometallic Chemistry Part 1 General Principles Suggested reading: Miessler/Tarr Chapters 13 and 14

Elements of organometallic chemistry Complexes containing M-C bonds Complexes with p-acceptor ligands Chemistry of lower oxidation states very important Soft-soft interactions very common Diamagnetic complexes dominant Catalytic applications


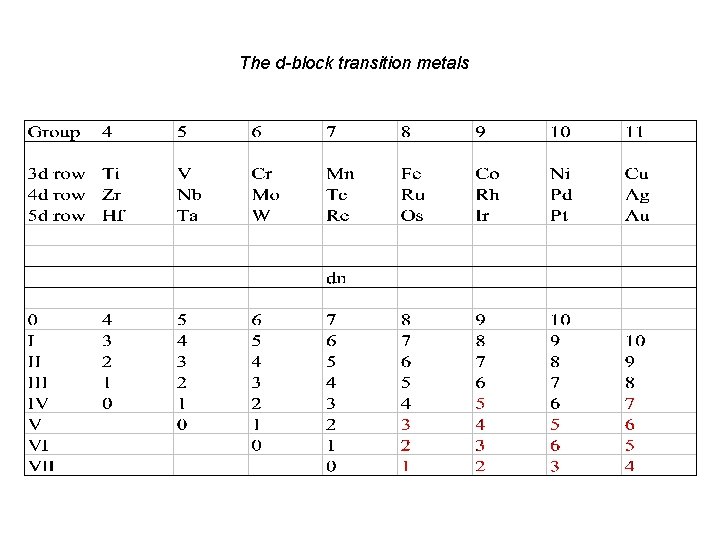
The d-block transition metals

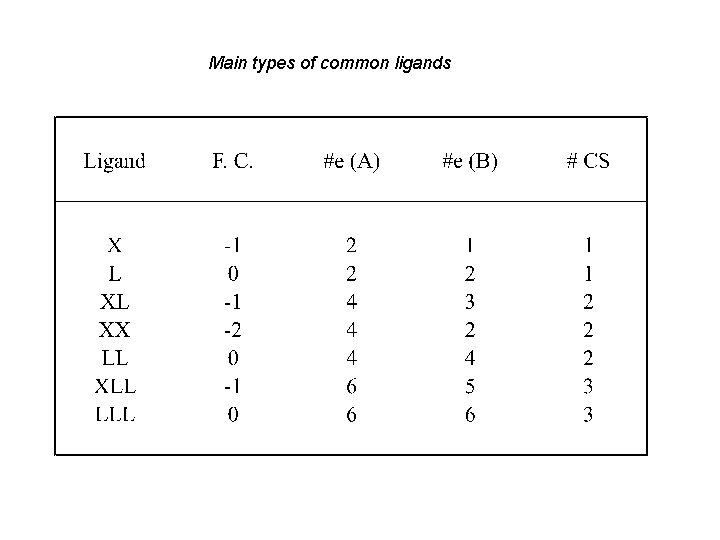
Main types of common ligands

A simple classification of the most important ligands X L L 2 X L 3

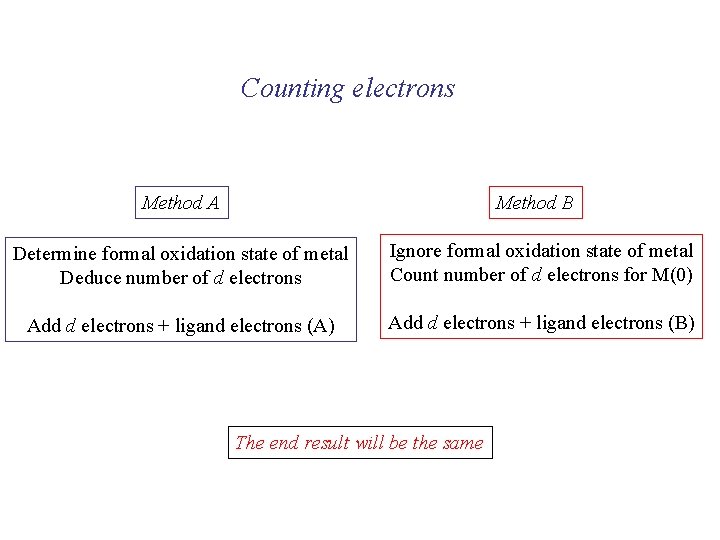
Counting electrons Method A Method B Determine formal oxidation state of metal Deduce number of d electrons Ignore formal oxidation state of metal Count number of d electrons for M(0) Add d electrons + ligand electrons (A) Add d electrons + ligand electrons (B) The end result will be the same


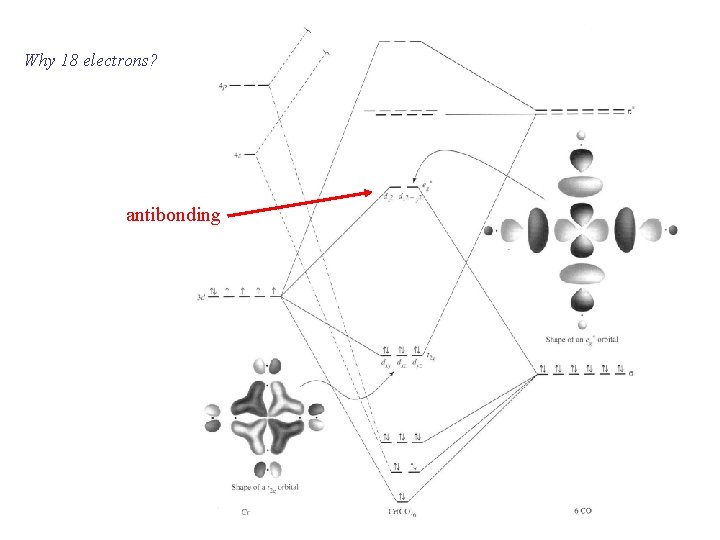
Why is this relevant? Stable mononuclear diamagnetic complexes generally contain 18 or 16 electrons The reactions of such complexes generally proceed through 18 - or 16 -electron intermediates Although many exceptions can be found, these are very useful practical rules for predicting structural and reactivity properties C. A. Tollman, Chem. Soc. Rev. 1972, 1, 337.

Why 18 electrons? antibonding

Organometallic complexes 18 -e most stable 16 -e stable (preferred for Rh(I), Ir(I), Pt(II), Pd(II)) <16 -e OK but usually very reactive > 18 -e possible but rare generally unstable

A closer look at some important ligands

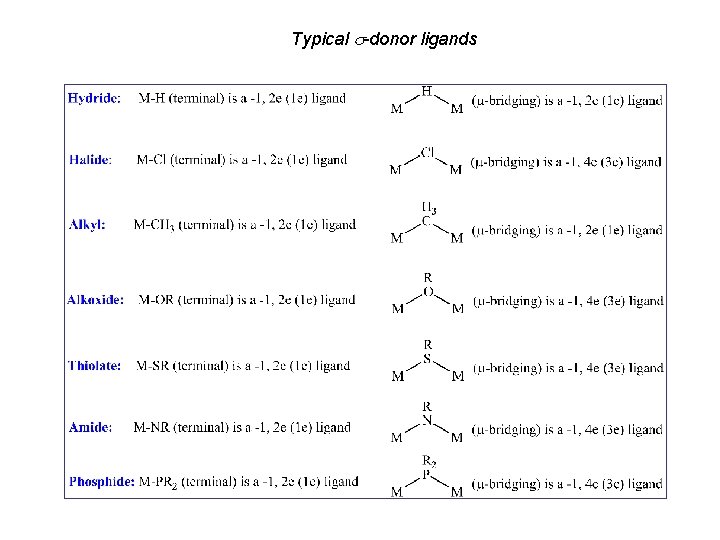
Typical -donor ligands

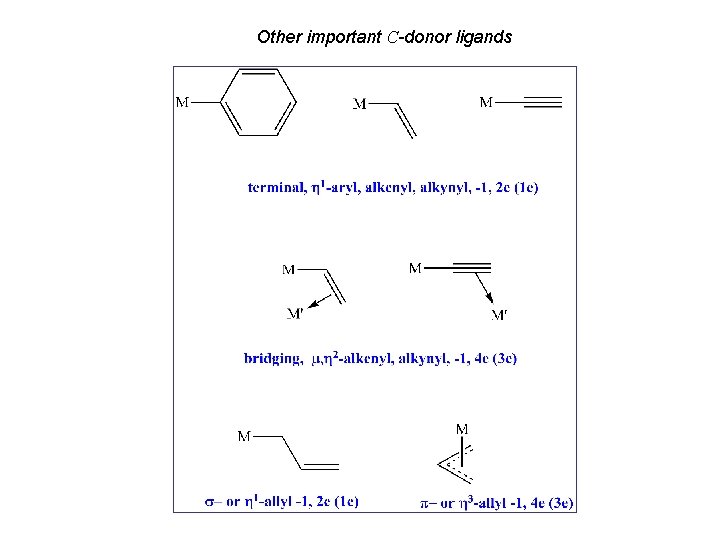
Other important C-donor ligands

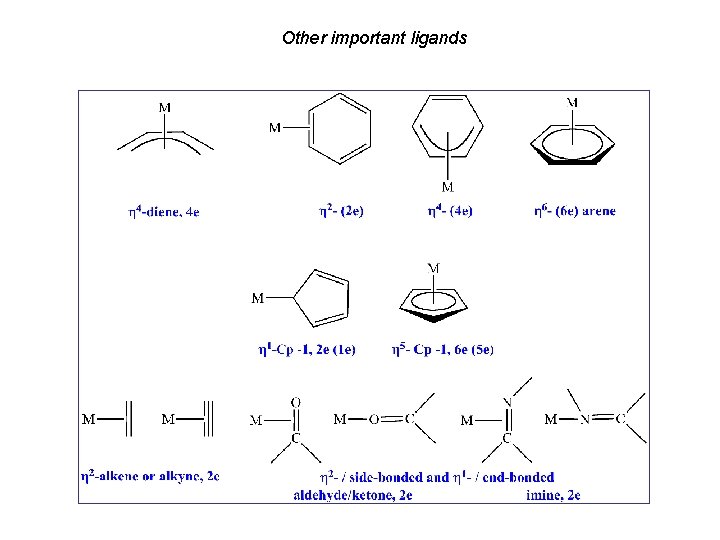
Other important ligands

Other important ligands

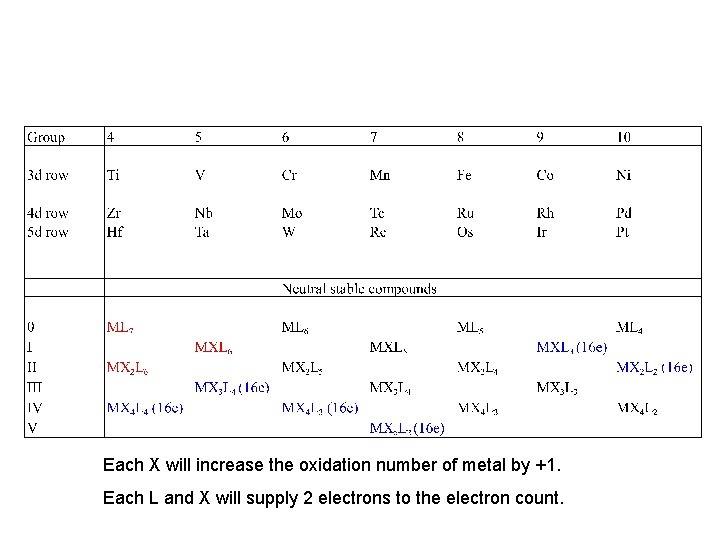
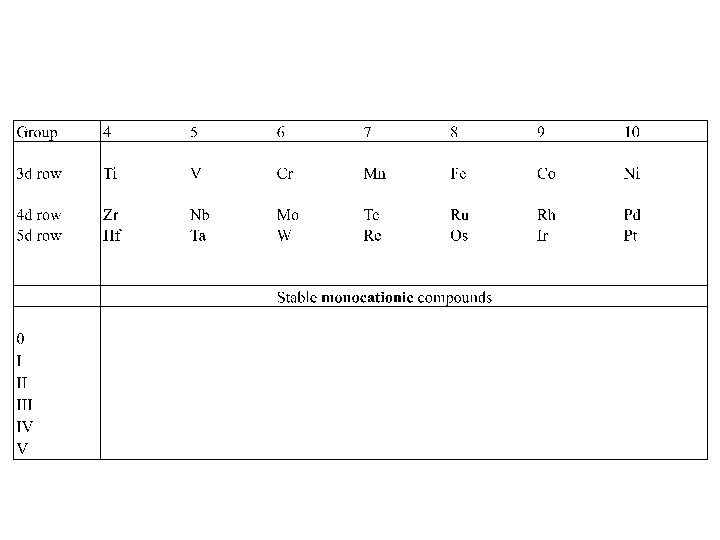
The M-L-X game

Each X will increase the oxidation number of metal by +1. Each L and X will supply 2 electrons to the electron count.


Now looking at compounds having a charge of +1 to obey 18 e rule. Elec count: 4 (M) +2 (NO) +12 (L 6) = 18 NO+ is isoelectronic to CO X increases O N by 1 Elec Count: 4 (M) + 4 (L 2) + 10 (L 5)

Actors and spectators Actor ligands are those that dissociate or undergo a chemical transformation (where the chemistry takes place!) Spectator ligands remain unchanged during chemical transformations They provide solubility, stability, electronic and steric influence (where ligand design is !)

Organometallic Chemistry 1. 2 Fundamental Reactions

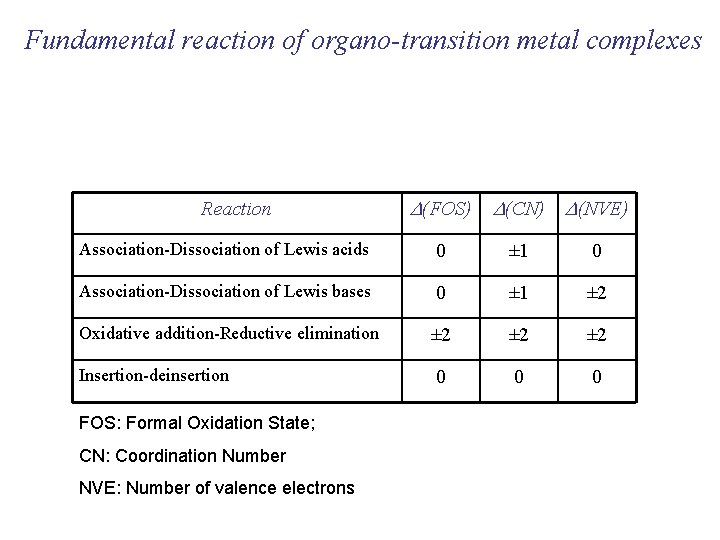
Fundamental reaction of organo-transition metal complexes Reaction D(FOS) D(CN) D(NVE) Association-Dissociation of Lewis acids 0 ± 1 0 Association-Dissociation of Lewis bases 0 ± 1 ± 2 Oxidative addition-Reductive elimination ± 2 ± 2 0 0 0 Insertion-deinsertion FOS: Formal Oxidation State; CN: Coordination Number NVE: Number of valence electrons

Association-Dissociation of Lewis acids D(FOS) = 0; D(CN) = ± 1; D(NVE) = 0 Lewis acids are electron acceptors, e. g. BF 3, Al. X 3, Zn. X 2 This shows that a metal complex may act as a Lewis base The resulting bonds are weak and these complexes are called adducts

Association-Dissociation of Lewis bases D(FOS) = 0; D(CN) = ± 1; D(NVE) = ± 2 A Lewis base is a neutral, 2 e ligand “L” (CO, PR 3, H 2 O, NH 3, C 2 H 4, …) in this case the metal is the Lewis acid Crucial step in many ligand exchange reactions For 18 -e complexes, only dissociation is possible For <18 -e complexes both dissociation and association are possible but the more unsaturated a complex is, the less it will tend to dissociate a ligand

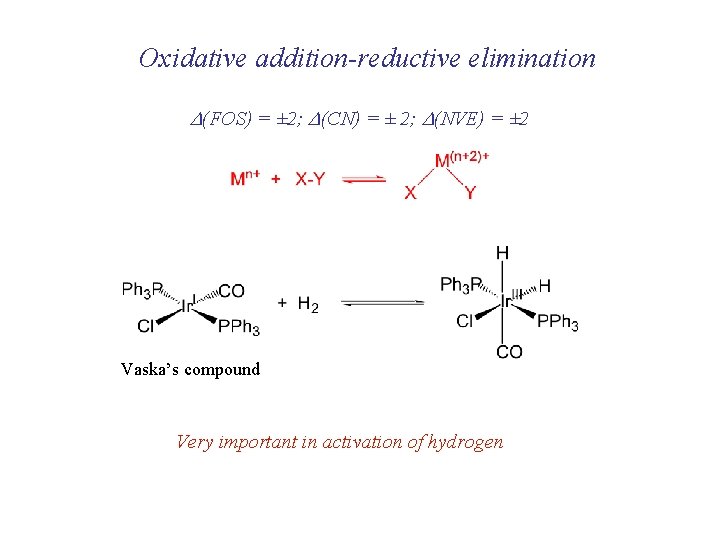
Oxidative addition-reductive elimination D(FOS) = ± 2; D(CN) = ± 2; D(NVE) = ± 2 Vaska’s compound Very important in activation of hydrogen

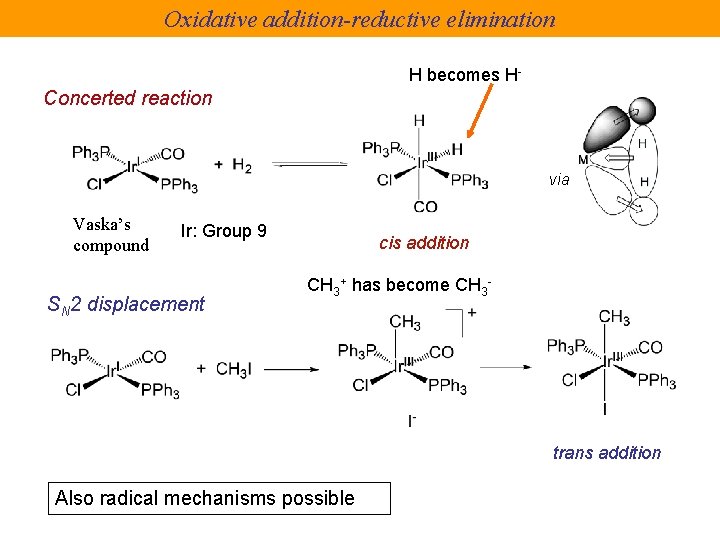
Oxidative addition-reductive elimination H becomes H- Concerted reaction via Vaska’s compound Ir: Group 9 SN 2 displacement cis addition CH 3+ has become CH 3 - trans addition Also radical mechanisms possible

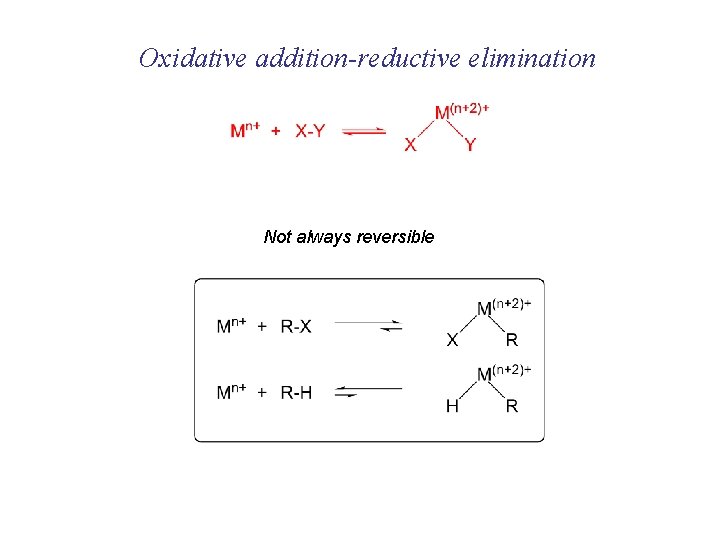
Oxidative addition-reductive elimination Not always reversible

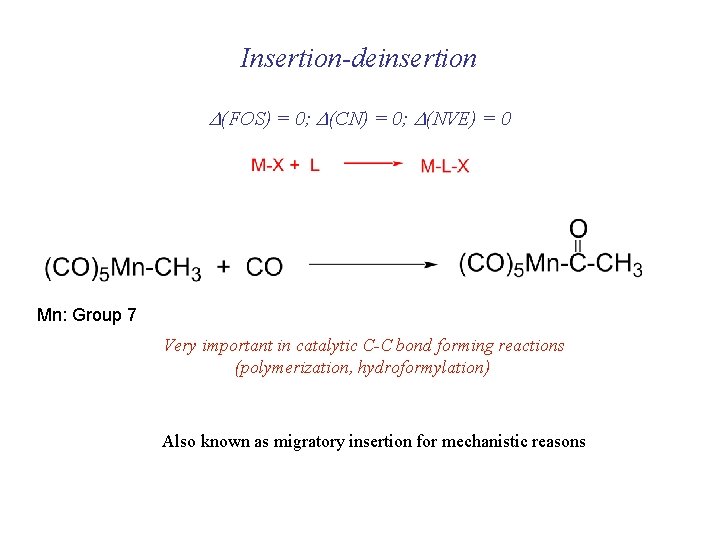
Insertion-deinsertion D(FOS) = 0; D(CN) = 0; D(NVE) = 0 Mn: Group 7 Very important in catalytic C-C bond forming reactions (polymerization, hydroformylation) Also known as migratory insertion for mechanistic reasons

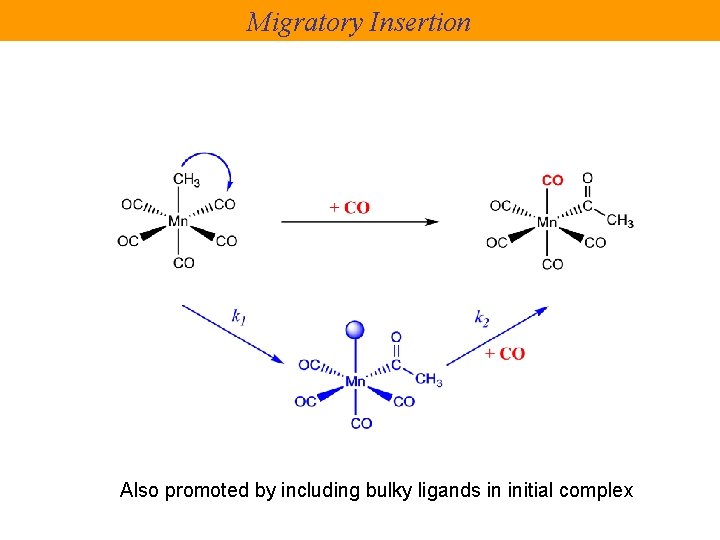
Migratory Insertion Also promoted by including bulky ligands in initial complex

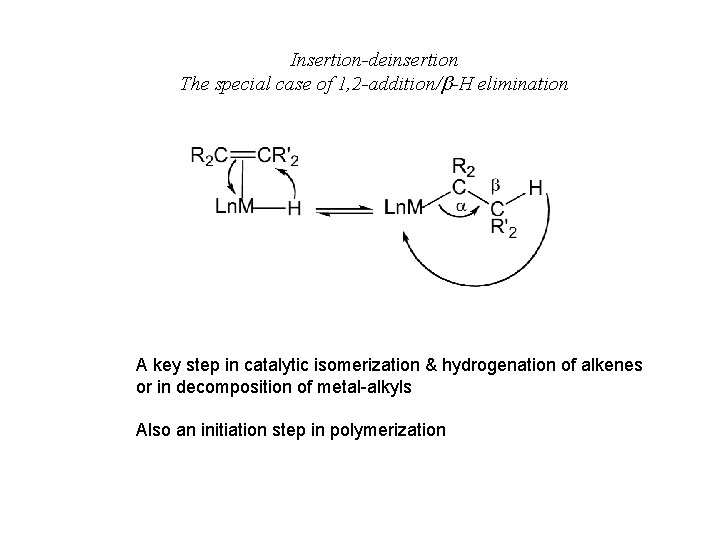
Insertion-deinsertion The special case of 1, 2 -addition/ -H elimination A key step in catalytic isomerization & hydrogenation of alkenes or in decomposition of metal-alkyls Also an initiation step in polymerization

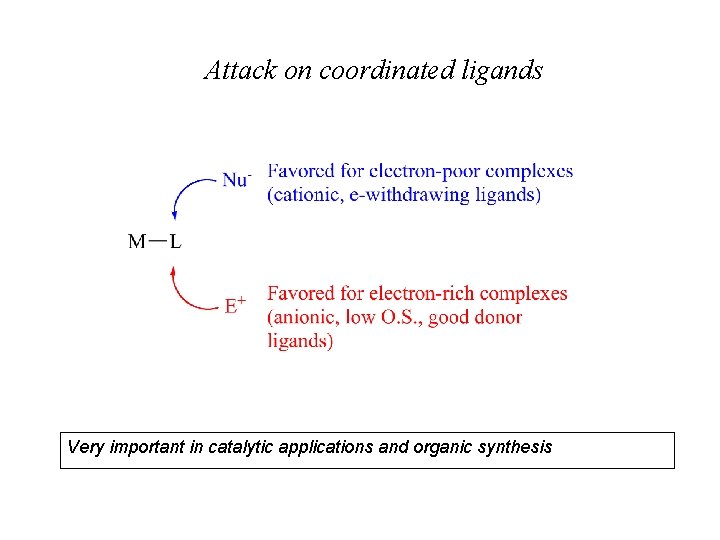
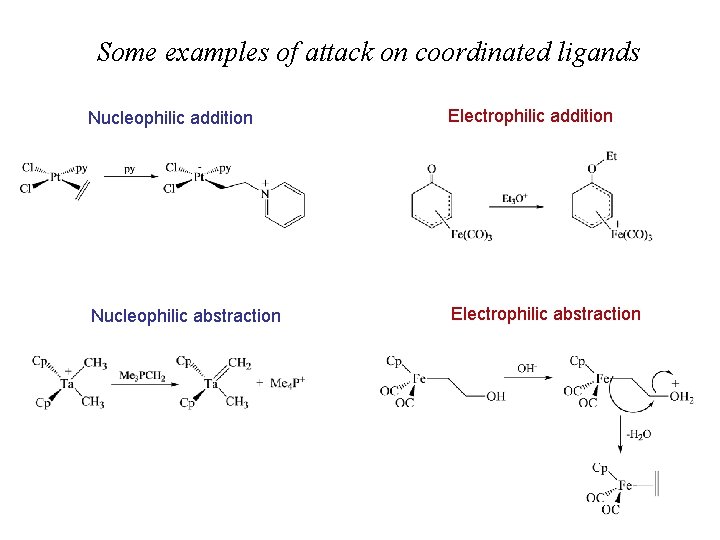
Attack on coordinated ligands Very important in catalytic applications and organic synthesis

Some examples of attack on coordinated ligands Nucleophilic addition Electrophilic addition Nucleophilic abstraction Electrophilic abstraction