Prevention and control of Hepatitis B In Central

Prevention and control of Hepatitis B In Central and Eastern Europe and Newly Independent States WHO/EURO

“Prevention and control of hepatitis B in CCEE and NIS” Siofok, Hungary, 1996, VHPB, WHO, CDC first opportunity to raise awareness on hepatitis B 4 to discuss universal immunization with decision makers. The aim was : 4 to summarize available data, 4 to identify needs to implement effective programmes 4 to underline main constraints 4

Situation in 1996 – 1 4 The WHO Regional Office estimated more than one million people acquire acute hepatitis B infection each year, most cases in NIS 4 Approximately 90 000 became chronic HBV carriers 4 In CCEE and NIS, levels of HBV endemicity were at intermediate or high endemic levels
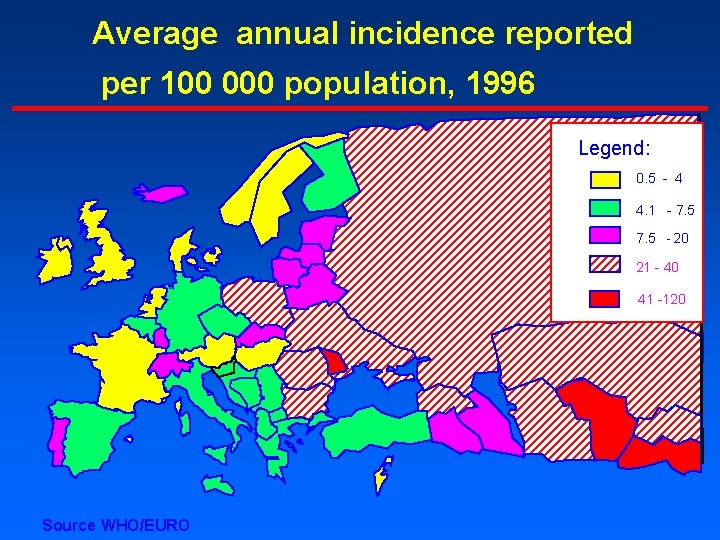
Average annual incidence reported per 100 000 population, 1996 Legend: 0. 5 - 4 4. 1 - 7. 5 - 20 21 - 40 41 -120 Source WHO/EURO

Epidemiology in Europe, 1996 4 The level of endemicity increased from north to south and from west to east, with carrier rates; – northwestern Europe < 0. 1% – midwestern Europe 0. 1 -0. 5% – southwestern Europe, 1 -5% – eastern Europe 2 -7% – central Asian Republics > 7%
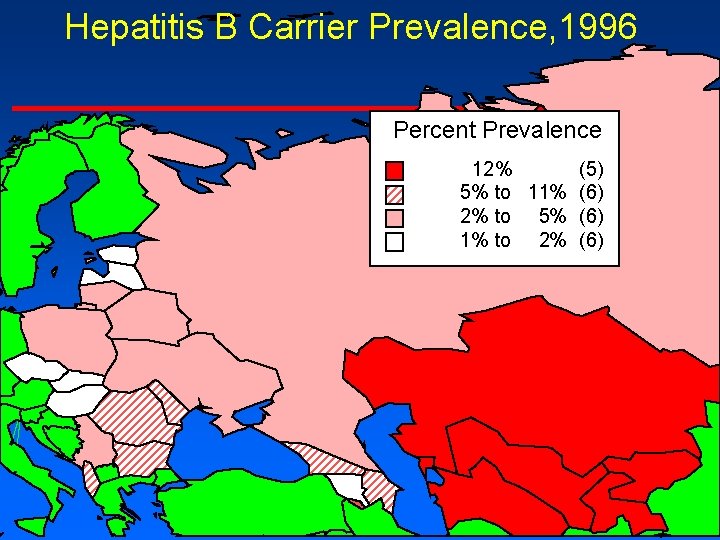
Hepatitis B Carrier Prevalence, 1996 Percent Prevalence 12% (5) 5% to 11% (6) 2% to 5% (6) 1% to 2% (6)

Hepatitis B Immunization schedules WHO/EORO, 1996 UNIVERSAL Uni INFANT Uni. INF + ADOS Uni. ADOS Consideration Selective Source WHO/EURO

Hepatitis B Implementation only 5 of the 25 countries in Central and Eastern Europe and the Newly Independent States had implemented, mainly because of economic constraints.

Recommendations to Countries 4 All countries should plan to integrate hepatitis B vaccination into their national immunization programmes as soon as possible. 4 All countries should develop a national plan for control of hepatitis B.

This plan should: 4 summarize current disease burden 4 include a strategy for routine vaccination of all infants and high-risk groups; 4 specify a time table and resources needed to implement the control programme

Recommendations to partners 4 The participants endorsed the UNICEF/ WHO strategy, calling for support of the neediest countries in obtaining hepatitis B vaccine. 4 Support should be targeted to countries with; – high disease burden, – well established EPI programmes, – a low per capita gross national product, – solid government commitment to hepatitis B prevention programmes.

Recommendations to WHO elaborate guidelines for national hepatitis B control plans, 4 provide assistance in developing and implementing these plans. 4 monitor effectiveness of hepatitis B prevention and control programmes 4 play coordinating role in working with other partners to support implementation of national plans 4

Progress since 1996. . .
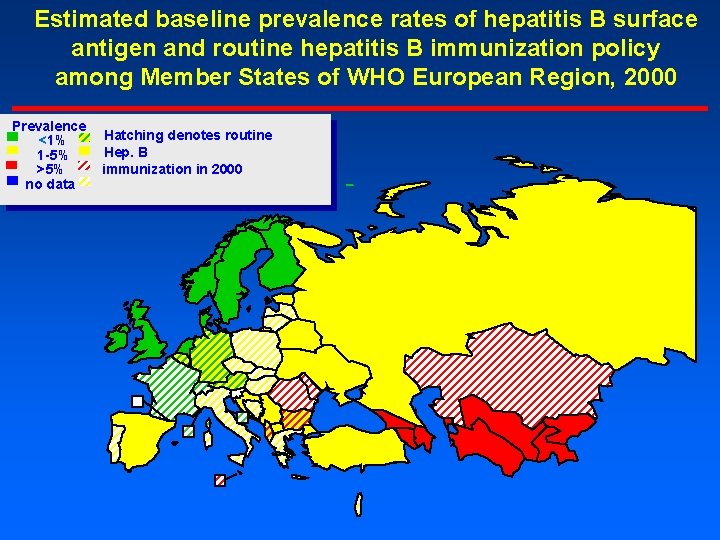
Estimated baseline prevalence rates of hepatitis B surface antigen and routine hepatitis B immunization policy among Member States of WHO European Region, 2000 Prevalence <1% 1 -5% >5% no data Hatching denotes routine Hep. B immunization in 2000

Incidence Rate of new hepatitis B cases, 1998/1999 (per 100, 000 population)
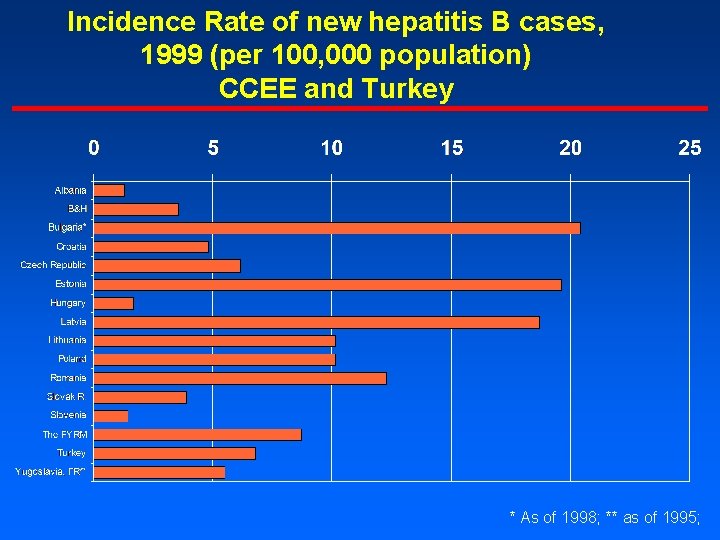
Incidence Rate of new hepatitis B cases, 1999 (per 100, 000 population) CCEE and Turkey * As of 1998; ** as of 1995;
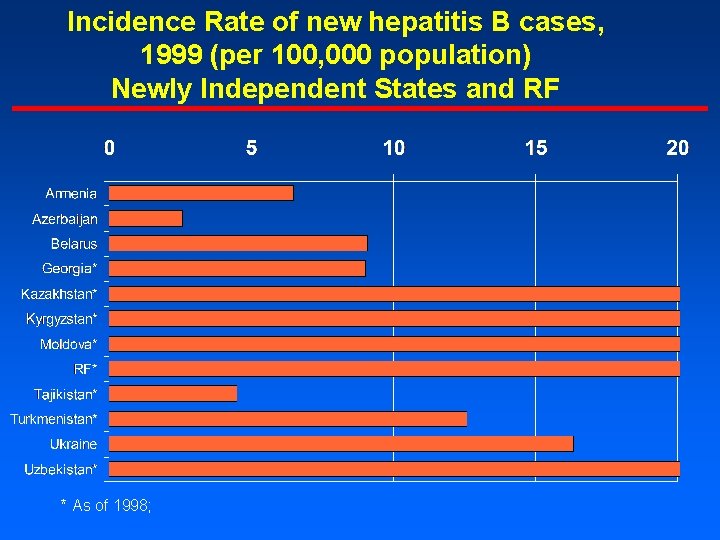
Incidence Rate of new hepatitis B cases, 1999 (per 100, 000 population) Newly Independent States and RF * As of 1998;
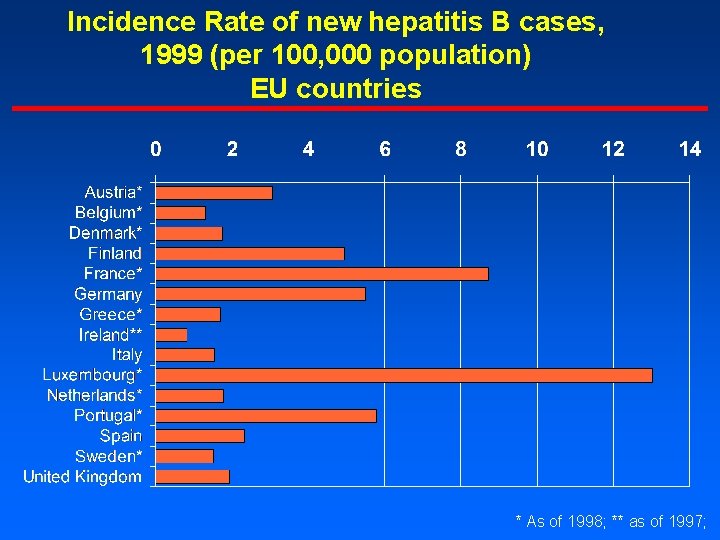
Incidence Rate of new hepatitis B cases, 1999 (per 100, 000 population) EU countries * As of 1998; ** as of 1997;

Routine hepatitis B immunization policy among Member States of WHO European Region, 2000 Universal imm Universal+ screening Risk groups Children born HBs(+) mother Adolescent
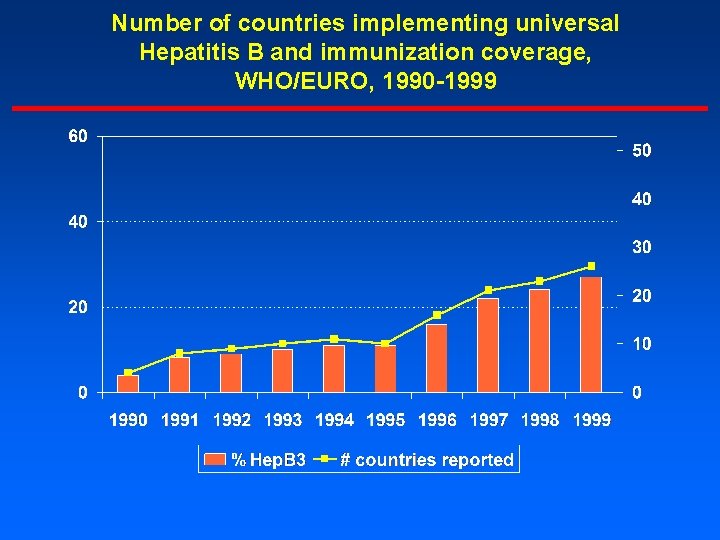
Number of countries implementing universal Hepatitis B and immunization coverage, WHO/EURO, 1990 -1999

Hepatitis B immunization Implementation in CCEE & NIS, 2000 4 High endemicity (5) – – – Albania (GF) Kazakhstan Kyrgyzstan (GF) Moldova (GF) Uzbekistan (GF) Intermediate (6) – Belarus – – – Bosnia & H. (F) Bulgaria (FYROMacedonia): Risk group Lithuania Romania 4 Low endemicity (9) – (Czech Republic) Risk group – (Croatia) Adolescents – – – Estonia: Born to HBs. Ag (+) mother Latvia Poland Slovakia Slovenia: Born to HBs. Ag (+) mother +Adolescents – Turkey – Ukraine: Born to HBs. Ag (+) mother

No Hepatitis B immunization programme, CCEE & NIS, 2000 4 High endemicity (5) – – – 4 Armenia (GF) Azerbaijan (GF) Georgia (GF) Tajikistan (GF) Turkmenistan (GF) Intermediate (1) – Russian Federation 4 Low endemicity (2) – Hungary – Yugoslavia

Hepatitis B Screening (survey + WHO/EURO database) 4 Screening of pregnant women: – universal screening recommended in 21 countries – 4 countries, selective screening – 7 countries no recommendation, because of birth dose – 18 countries no information
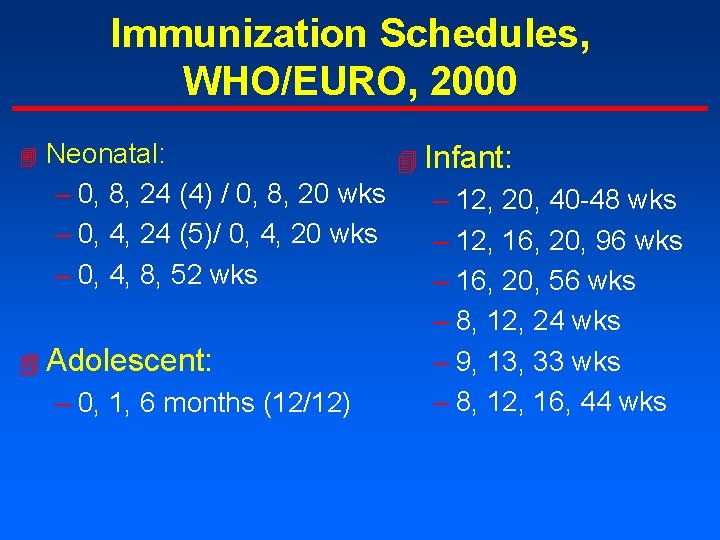
Immunization Schedules, WHO/EURO, 2000 Neonatal: 4 Infant: – 0, 8, 24 (4) / 0, 8, 20 wks – 12, 20, 40 -48 wks – 0, 4, 24 (5)/ 0, 4, 20 wks – 12, 16, 20, 96 wks – 0, 4, 8, 52 wks – 16, 20, 56 wks – 8, 12, 24 wks 4 Adolescent: – 9, 13, 33 wks – 8, 12, 16, 44 wks – 0, 1, 6 months (12/12) 4

Hepatitis B Risk group immunization (survey + WHO/EURO database) 4 Risk group programme: – information for 22/24 – 15/19 in addition to a universal programme – 6/19 risk group programme and no universal programme – no risk group programme
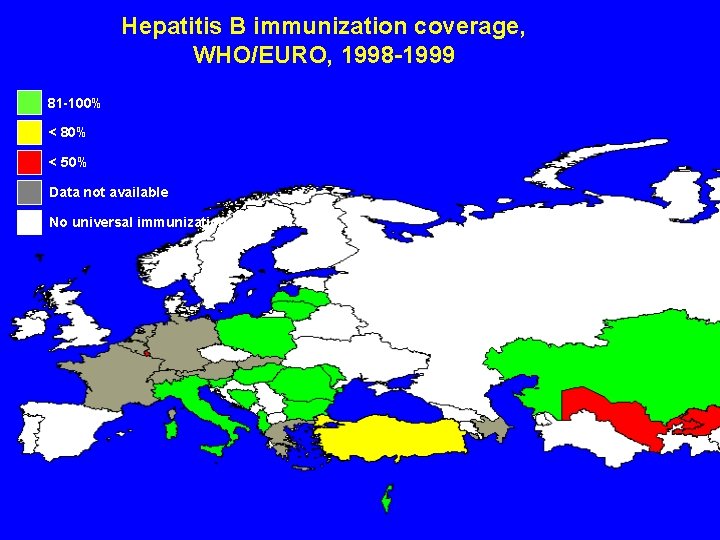
Hepatitis B immunization coverage, WHO/EURO, 1998 -1999 81 -100% < 80% < 50% Data not available No universal immunization
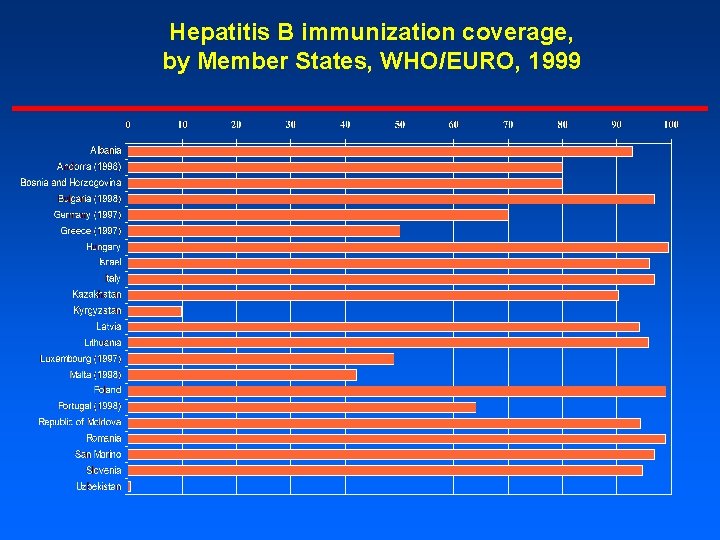
Hepatitis B immunization coverage, by Member States, WHO/EURO, 1999
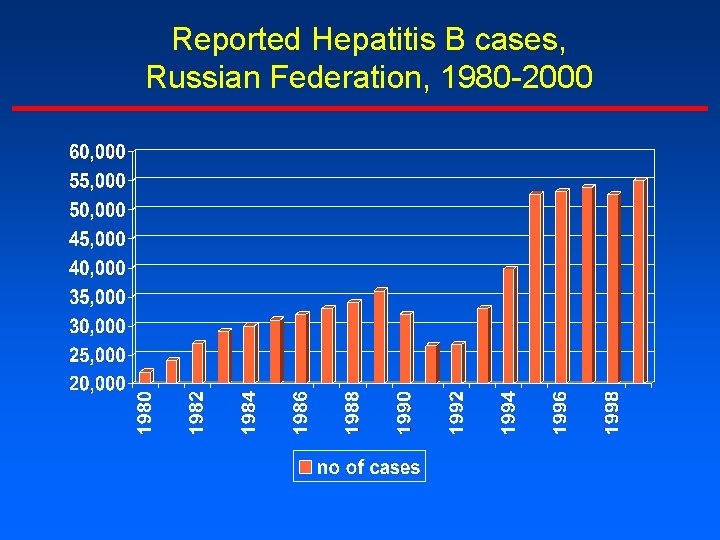
Reported Hepatitis B cases, Russian Federation, 1980 -2000
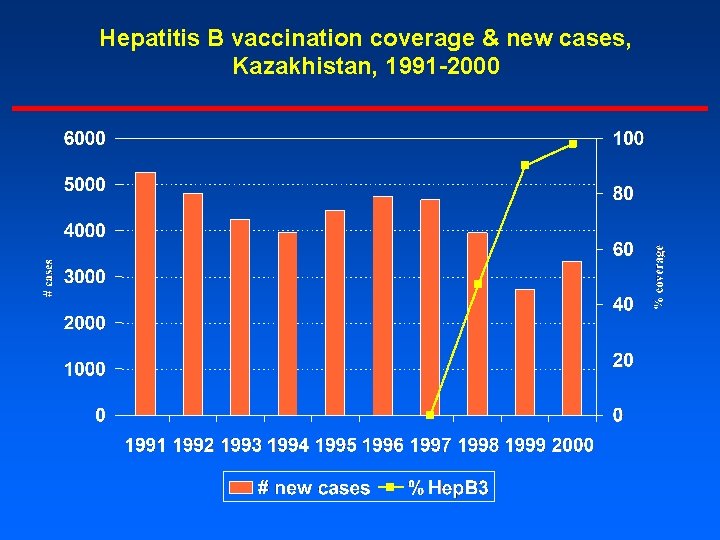
Hepatitis B vaccination coverage & new cases, Kazakhistan, 1991 -2000
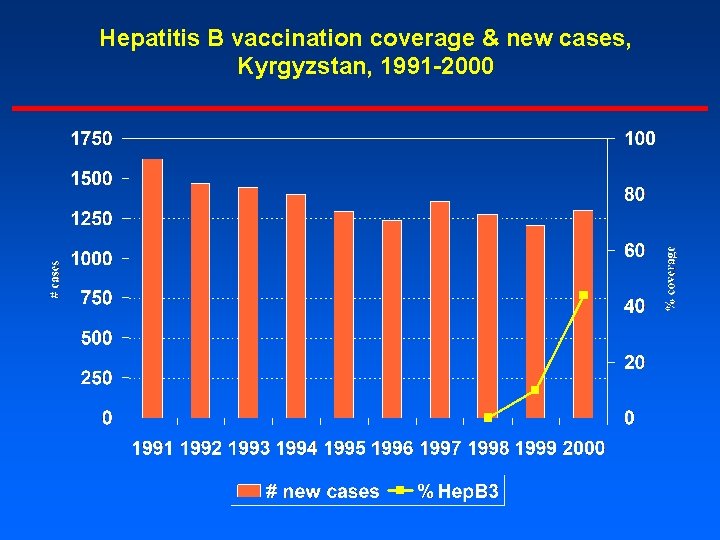
Hepatitis B vaccination coverage & new cases, Kyrgyzstan, 1991 -2000
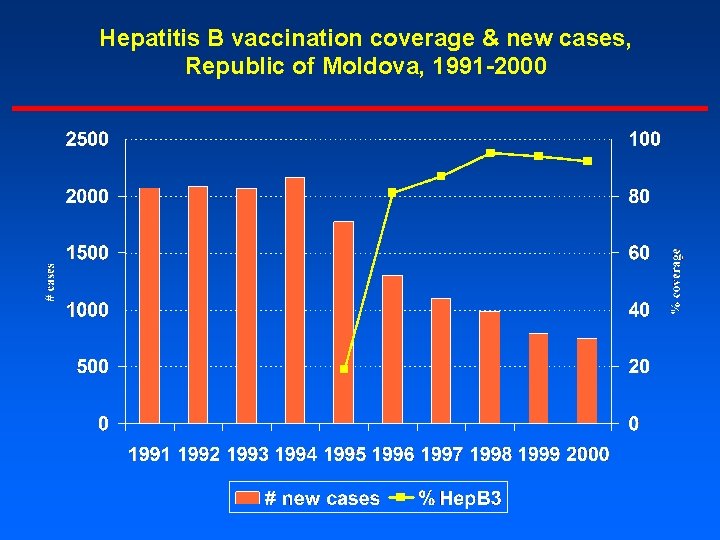
Hepatitis B vaccination coverage & new cases, Republic of Moldova, 1991 -2000
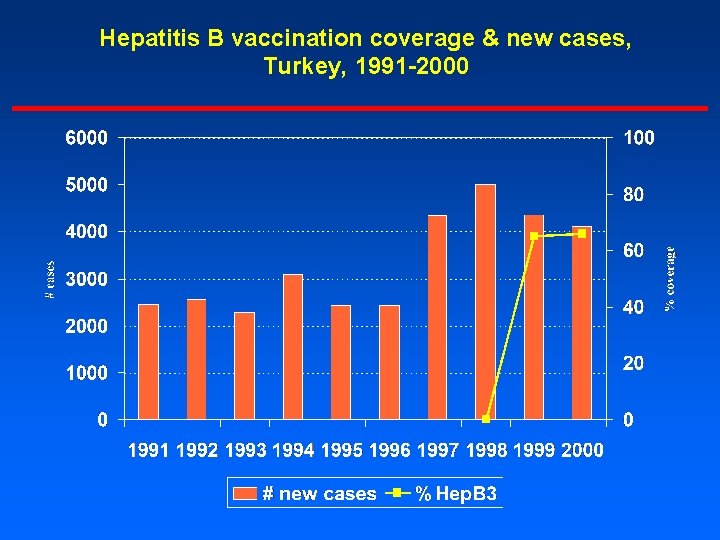
Hepatitis B vaccination coverage & new cases, Turkey, 1991 -2000

Conclusion 4 Main challenges – sustaining immunization services – increasing coverage – logistics and cold chain – safety of injections – monitoring performance – evaluation of impact
- Slides: 33