Preventable Adverse Event Reporting Vickie Gillespie PAE Clinical

Preventable Adverse Event Reporting Vickie Gillespie, PAE Clinical Specialist 04/28/15 1

Preventable Adverse Event (PAE) Reporting in Texas Objectives: – Review the background of Preventable Adverse Event reporting nationwide and in Texas. – Summarize the reporting program. – Share Texas reported events to date. Adverse Event: An event that results in unintended harm to the patient by an act of commission or omission rather than by the underlying disease or condition of the patient. 2

Why Report? • 1999 Institute of Medicine (IOM)* – Estimated 98, 000 deaths/year – Most were systemic errors – Called for a nationwide, mandatory reporting system • 2013 John T. James, Ph. D** – 2008 -2011 four studies estimated a lower limit of 210, 000 deaths/year – Newest estimate is 440, 000 deaths/year – Serious harm 10 -20 times higher than lethal harm (2 -4 Million serious harm events/year) *To err is human: building a safer health system. Kohn LT, Corrigan JM, Donaldson MS (Institute of Medicine). Washington, DC: National Academy Press, 2000. **A New, Evidence-based Estimate of Patient Harms Associated with Hospital Care, James, John T. Ph. D, Journal of Patient Safety: September 2013 - Volume 9 - Issue 3 - p 122 -128 3

Scope of the Problem • Falls— – 700, 000 -1, 000 falls annually 1 – Leading cause injury-related death 65 & older – $30 billion by 20202, 3 • Pressure Ulcers— – 257, 412 Medicare patients 20071 – 60, 000 patients die annually from HA PUs – Average charge of $43, 1804 • Medication Errors— – 1000/day in hospitalized pts 5 – 15/100 admissions— 75% preventable 6 • HAIs— – 2 Million annually in US 7 (200, 000 in Texas 8) – ~ 90, 000 deaths (8 -9000 Texas deaths) – ~ $5 billion - $ 31. 5 billion 2 healthcare costs 4

World Health Organization • Healthcare prone to accidents— – Greatest contributor is human error – Most human errors induced by system failures • Reason’s Swiss Cheese Model • One solution is reporting— – By doctor, nurse or other provider within an organization – By the organization to a broader audience • Effective reporting— – Cornerstone of safe practice – Measure of progress toward a safety culture Draft Guidelines for Adverse Event Reporting and Learning Systems, World Alliance for Patient Safety, World Health Organization, 2005 5
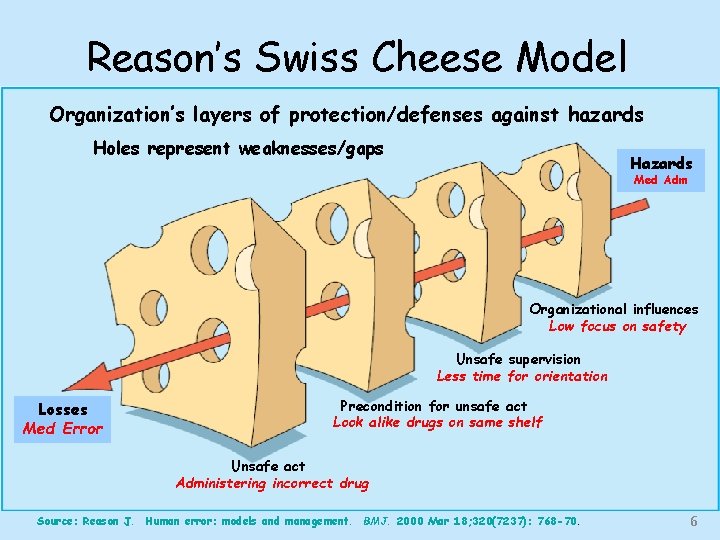
Reason’s Swiss Cheese Model Organization’s layers of protection/defenses against hazards Holes represent weaknesses/gaps Hazards Med Adm Organizational influences Low focus on safety Unsafe supervision Less time for orientation Losses Med Error Precondition for unsafe act Look alike drugs on same shelf Unsafe act Administering incorrect drug Source: Reason J. Human error: models and management. BMJ. 2000 Mar 18; 320(7237): 768 -70. 6

World Health Organization • Healthcare prone to accidents— – Greatest contributor is human error – Most human errors induced by system failures • One solution is reporting— – By doctor, nurse or other provider within an organization – By the organization to a broader audience • Effective reporting— – Cornerstone of safe practice – Measure of progress toward a safety culture Draft Guidelines for Adverse Event Reporting and Learning Systems, World Alliance for Patient Safety, World Health Organization, 2005 7

Reasons for PAE Reporting Establish Standards Transparency Systemic Learning Consumer’s right to know Evidence-based Practices Increase Patient Safety 8

AHRQ 2014 Healthcare Quality and Disparities Report • Between 2010 and 2013: ü Patient Safety improved ü 17% reduction in rates of hospital-acquired conditions. ü Half of Patient Safety measures improved ü 1. 3 million fewer harms to patients ü 50, 000 lives saved (est) ü $12 billion in cost savings (est) 9

Public Reporting of Adverse Events 2014 Guide to State Adverse Event Reporting Systems 10 National Academy for State Health Policy

PAE Reporting in Texas • 80 th Legislative Session 2007 (SB 288) – Chapter 98 Health and Safety Code – HAI Reporting, Advisory Panel • 81 st Legislative Session 2009 (SB 203) – PAE added and required: *Healthcare facilities to report certain preventable adverse events to the DSHS, AND *DSHS to make this data available to the public by facility, by type, and by number. 11

Who Must Report? • General Hospitals licensed under Chapter 241 or a hospital that provides surgeries or obstetrical services and is maintained or operated by this State. – All General Hospitals provide OB and/or Surgery. – Comprehensive Medical Rehabilitation Hospitals do not have to report. – LTAC’s must report if they are licensed as a General Hospital (provide OB and/or Surgery). – It does not include a LTAC licensed as a Special Hospital. • Ambulatory Surgery Centers licensed under Chapter 243. 12

What is Reported? • Only ACTUAL events are reported: – – – Category of Event Type of Event Date of Event MR/Patient ID#, Level of Harm • Details of the event and root cause analysis are NOT required—but may be submitted. 13
14

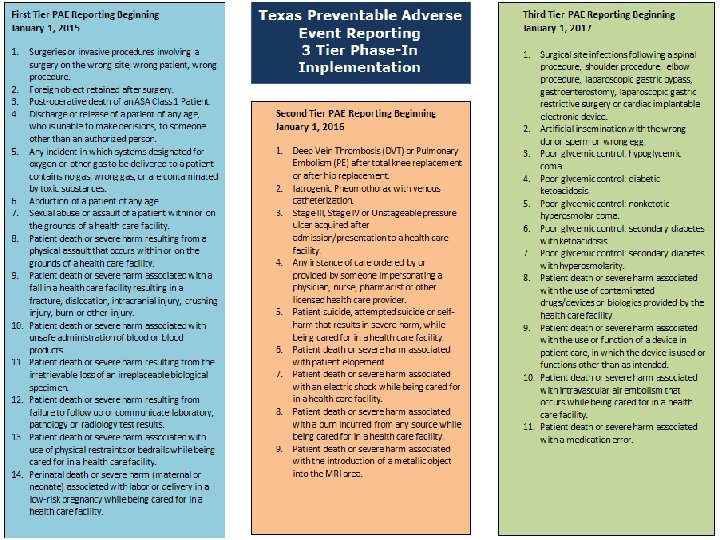
First Tier PAE Reporting January 1, 2015 SURGICAL OR INVASIVE PROCEDURE EVENTS 1. Surgeries or invasive procedures involving a surgery on the wrong site, wrong patient, wrong procedure. 2. Foreign object retained after surgery. 3. Post-operative death of an ASA Class 1 Patient. PATIENT PROTECTION EVENTS 1. Discharge or release of a patient of any age, who is unable to make decisions, to someone other than an authorized person. 15

First Tier PAE Reporting January 1, 2015 ENVIRONMENTAL EVENTS POTENTIAL CRIMINAL EVENTS 1. Any incident in which systems designated for oxygen or other gas to be delivered to a patient contains no gas, wrong gas, or are contaminated by toxic substances. 1. Abduction of a patient of any age. 2. Patient death or severe harm associated with use of physical restraints or bedrails while being cared for in a health care facility. 3. Patient death or severe harm resulting from a physical assault that occurs within or on the grounds of a health care facility. 2. Sexual abuse or assault of a patient within or on the grounds of a health care facility. 16

First Tier PAE Reporting January 1, 2015 CARE MANAGEMENT EVENTS 1. Patient death or severe harm associated with unsafe administration of blood or blood products. 2. Patient death or severe harm associated with a fall in a health care facility resulting in a fracture, dislocation, intracranial injury, crushing injury, burn or other injury. 3. Patient death or severe harm resulting from the irretrievable loss of an irreplaceable biological specimen. 4. Perinatal death or severe harm (maternal or neonatal) associated with labor or delivery in a low-risk pregnancy while being cared for in a health care facility. 5. Patient death or severe harm resulting from failure to follow up or communicate laboratory, pathology or radiology test results. 17

PAE Reporting Schematic PAE Event Report Email Reminders Access Facility Report Facilities report directly into Tx. HSN via TDSHS website portal. Facility-specific reports are then generated. 18
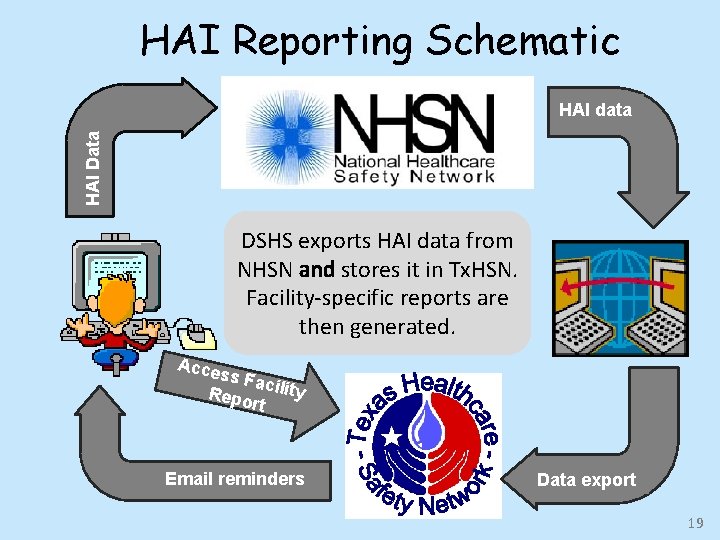
HAI Reporting Schematic HAI Data HAI data DSHS exports HAI data from NHSN and stores it in Tx. HSN. Facility-specific reports are then generated. Acce ss Fa c Repo ility rt Email reminders Data export 19
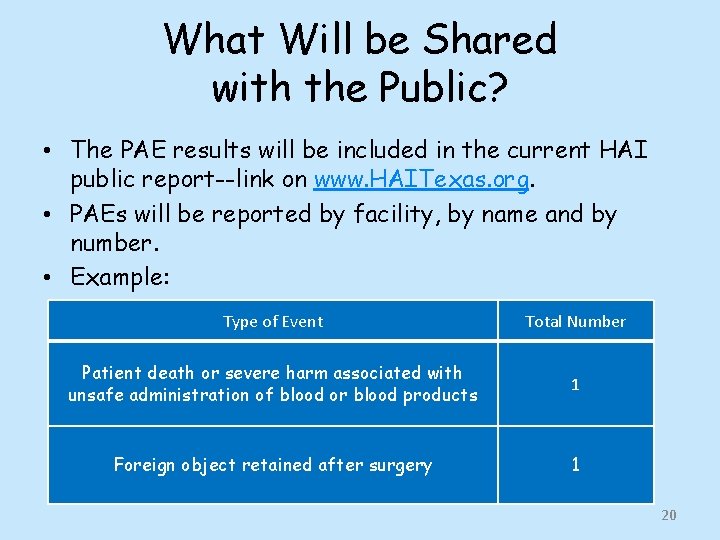
What Will be Shared with the Public? • The PAE results will be included in the current HAI public report--link on www. HAITexas. org. • PAEs will be reported by facility, by name and by number. • Example: Type of Event Total Number Patient death or severe harm associated with unsafe administration of blood or blood products 1 Foreign object retained after surgery 1 20
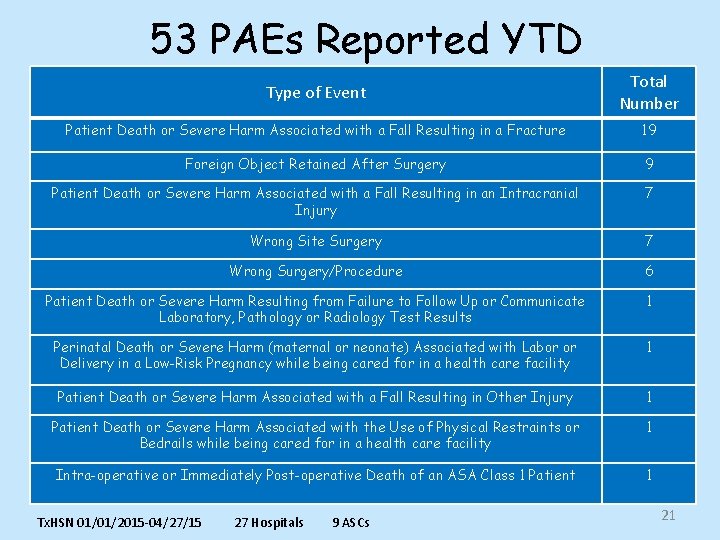
53 PAEs Reported YTD Type of Event Total Number Patient Death or Severe Harm Associated with a Fall Resulting in a Fracture 19 Foreign Object Retained After Surgery 9 Patient Death or Severe Harm Associated with a Fall Resulting in an Intracranial Injury 7 Wrong Site Surgery 7 Wrong Surgery/Procedure 6 Patient Death or Severe Harm Resulting from Failure to Follow Up or Communicate Laboratory, Pathology or Radiology Test Results 1 Perinatal Death or Severe Harm (maternal or neonate) Associated with Labor or Delivery in a Low-Risk Pregnancy while being cared for in a health care facility 1 Patient Death or Severe Harm Associated with a Fall Resulting in Other Injury 1 Patient Death or Severe Harm Associated with the Use of Physical Restraints or Bedrails while being cared for in a health care facility 1 Intra-operative or Immediately Post-operative Death of an ASA Class 1 Patient 1 Tx. HSN 01/01/2015 -04/27/15 27 Hospitals 9 ASCs 21

www. PAETexas. org PAETexas@dshs. state. tx. us 22

Questions? Thank you! 23
- Slides: 23