PRESSURE VARIABLES 1 Increase Temperature Particles move faster




PRESSURE VARIABLES 1. ) Increase Temperature Particles move faster collisions increase Pressure increases 2. ) Decrease volume Same speed collisions increase Pressure increases 3. ) Add more moles (n) Same speed Collisions increase Pressure increases
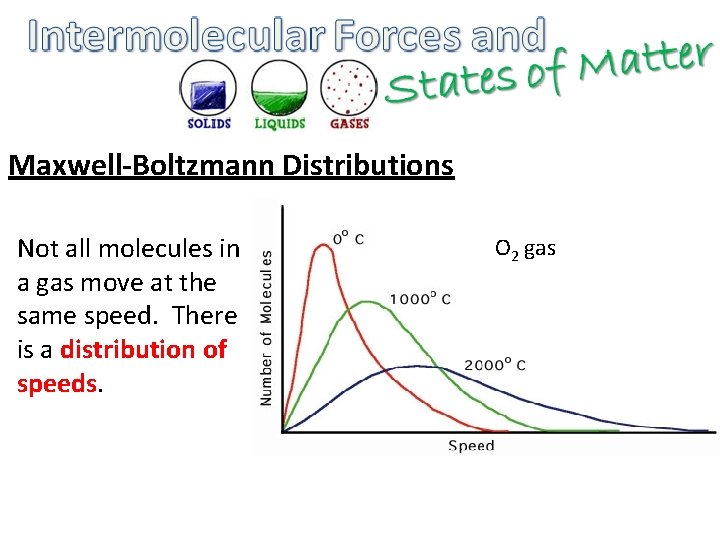
Maxwell-Boltzmann Distributions Not all molecules in a gas move at the same speed. There is a distribution of speeds. O 2 gas
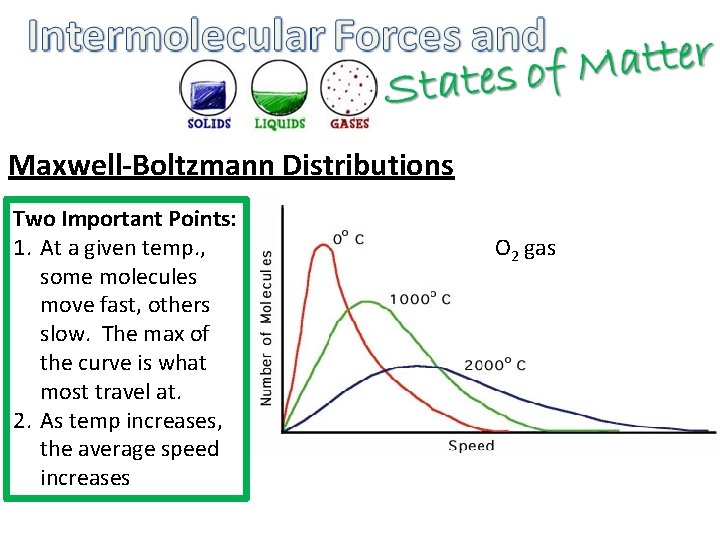
Maxwell-Boltzmann Distributions Two Important Points: 1. At a given temp. , some molecules move fast, others slow. The max of the curve is what most travel at. 2. As temp increases, the average speed increases O 2 gas

Calculations with Molecular speed: Kinetic energy of a single gas molecule depends on two things: it’s mass and it’s speed The Formula:
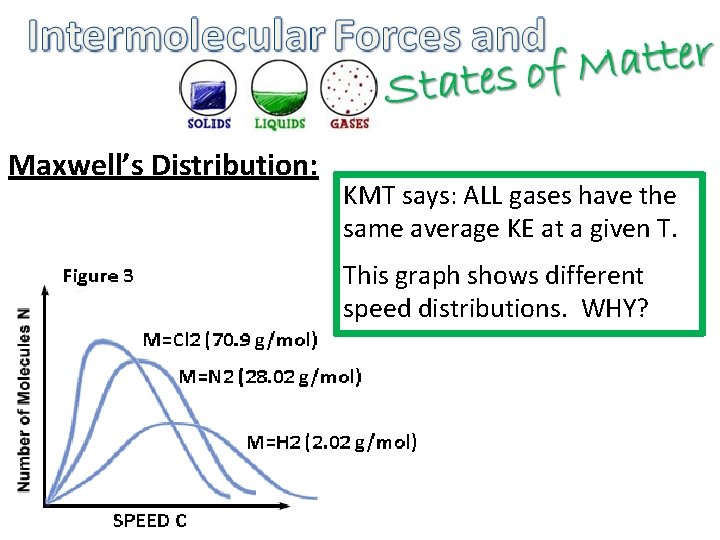
Maxwell’s Distribution: KMT says: ALL gases have the same average KE at a given T. This graph shows different speed distributions. WHY?


• MAXWELL’S DISTRIBUTION – Since all gases are at the same temperature, their kinetic energies are equal – For example, take Cl 2 N 2 and H 2 – KECl 2 = KEN 2 = KEH 2 – ½ mv 2 = ½ mv 2 – Since Cl 2’s mass is larger, it will move slower – Since H 2’s mass is smaller, it will move faster

• MAXWELL’S EQUATION R = 8. 314 J / mol K T = temperature in Kelvin M = MW in kg/mol PRACTICE: CALCULATE FOR OXYGEN AT 25°C

• LET’S WORK ON THE PRACTICE PROBLEMS…
- Slides: 14