PRESSURE Pressure is the distribution of force over

PRESSURE

Pressure is the distribution of force over a certain area P=F/A Force is measured in Newtons (N) Area is measured in m 2 Pressure is measured in N/m 2 1 N/m 2 is also called 1 pascal (Pa) 1000 Pa is called a kilopascal (k. Pa) On Earth's surface, a mass of 1 kg exerts a force of approximately 9. 81 N [down]. Therefore, if you weigh 57 kg (125 pounds), you will exert a force of about 560 N (57 x 9. 81).

There are many, many different physical situations that we encounter every day in which pressure is an important element. EXAMPLES: Walking Peeling an apple Hitting a nail

Using the formula for Pressure, explain how a thumbtack works. (hint: there are two surface areas – the top of the tack and the pin)

Using the formula for pressure, explain why it would hurt your foot more to wear this shoe: As opposed to this shoe:

Why would it be better to get an injection with a sharp syringe rather than a dull syringe?

“Bed of nail. S” versus a “bed of a nail” http: //www. youtube. com/watch? v=fq. Cd. Ib. Vmaq. M

Snowshoes Complete sheet on Pressure questions

Calculations Force(N) = mass(kg) x gravity(9. 8 m/s 2) What pressure is exerted by 150 m. L(g) of water with a surface area 0. 001 m 2? What pressure is exerted if we double the mass of the water?

Compressible and Incompressible Fluids

A fluid is any substance that has the ability to flow, or take the shape of its container. Therefore, both liquids and gases are considered fluids Confined fluids are fluids that are in a closed system. Systems can be pneumatic (gases) or hydraulic (liquids). Examples: Air or water mattress Circulatory System Braking Mechanisms Respiratory System
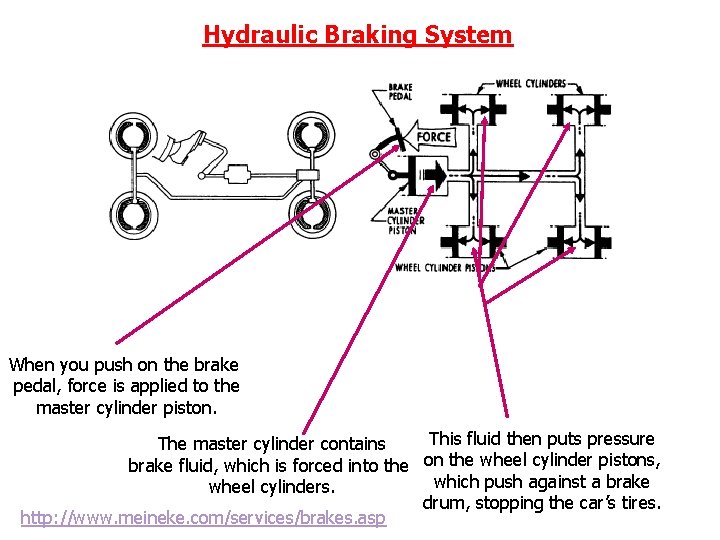
Hydraulic Braking System When you push on the brake pedal, force is applied to the master cylinder piston. This fluid then puts pressure The master cylinder contains brake fluid, which is forced into the on the wheel cylinder pistons, which push against a brake wheel cylinders. drum, stopping the car’s tires. http: //www. meineke. com/services/brakes. asp
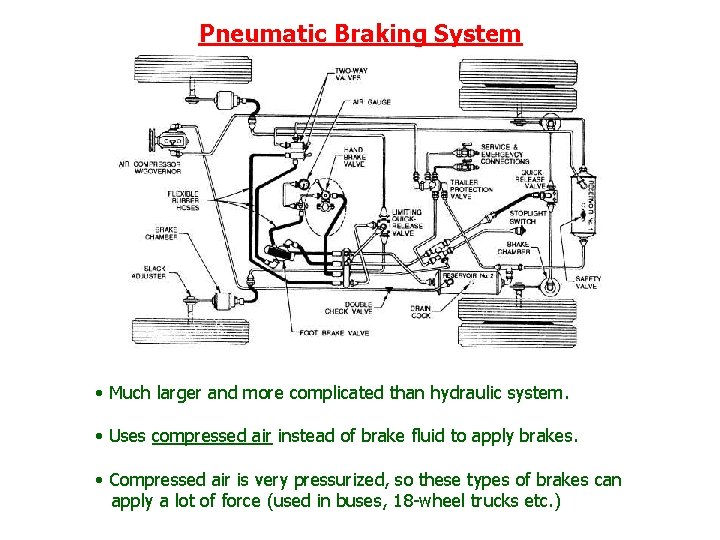
Pneumatic Braking System • Much larger and more complicated than hydraulic system. • Uses compressed air instead of brake fluid to apply brakes. • Compressed air is very pressurized, so these types of brakes can apply a lot of force (used in buses, 18 -wheel trucks etc. )

Using your knowledge of the PARTICLE THEORY (and what you learned in science last year), what is the major difference between using a pneumatic or hydraulic system? Liquids are virtually INCOMPRESSIBLE – they cannot be compressed (their volume cannot be reduced by much) Gases are COMPRESSIBLE – their volume can be reduced In a liquid, the spaces between particles are very small. When force is applied to a liquid, only a very small decrease in volume takes place. Thus they transmit force better than gases. In a gas, the particles are far away from each other. When a force is applied to a gas, the particles get pushed together and the gas is compressed. This is why there is a delay in using air filled systems – it takes time to compress the air.

Because gases are compressible and their volume can be changed, they behave in very predictable ways……….

Gas Laws: Boyle and Charles/Gay-Lussac’s Laws

Boyle’s Law In the 1700 s, scientist Robert Boyle (see above) investigated the relationship between the volume of a gas and its pressure. There are four different variables that can be changed when dealing with a sample of gas: Pressure Amount (number of particles) Volume Temperature
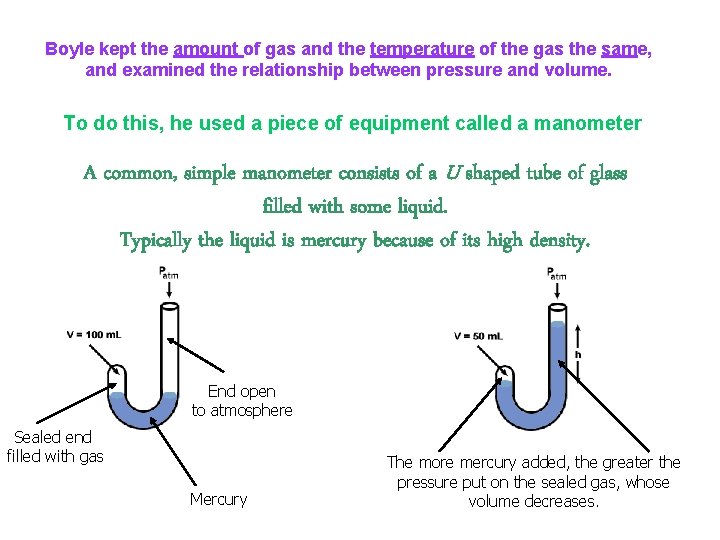
Boyle kept the amount of gas and the temperature of the gas the same, and examined the relationship between pressure and volume. To do this, he used a piece of equipment called a manometer A common, simple manometer consists of a U shaped tube of glass filled with some liquid. Typically the liquid is mercury because of its high density. End open to atmosphere Sealed end filled with gas Mercury The more mercury added, the greater the pressure put on the sealed gas, whose volume decreases.

He found that as the volume of a gas decreased, the pressure on the gas increased as long as the temperature of the gas and the amount of gas is kept constant. OR P 1 V 1=P 2 V 2 This is known as Boyle’s Law
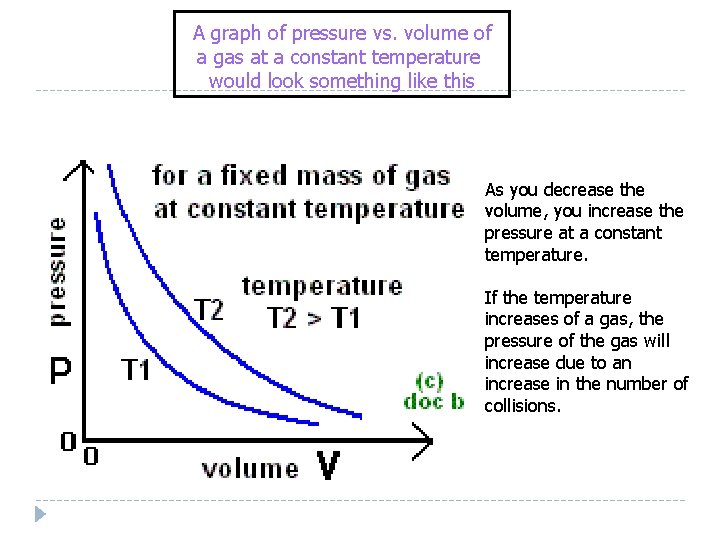
A graph of pressure vs. volume of a gas at a constant temperature would look something like this As you decrease the volume, you increase the pressure at a constant temperature. If the temperature increases of a gas, the pressure of the gas will increase due to an increase in the number of collisions.
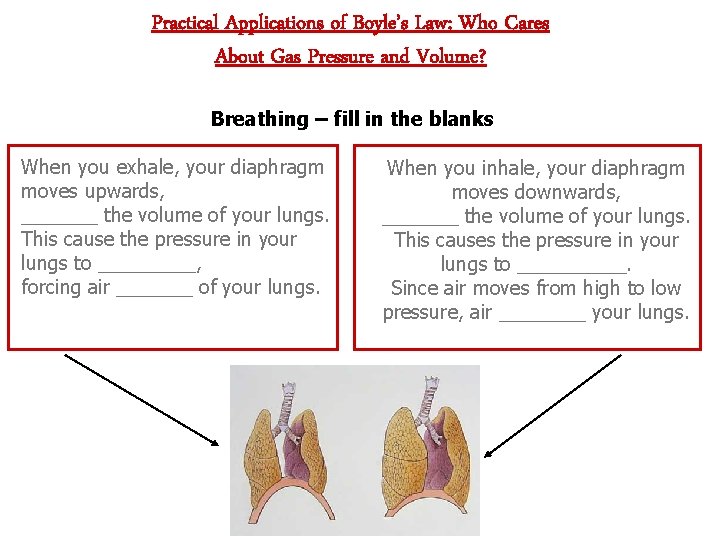
Practical Applications of Boyle’s Law: Who Cares About Gas Pressure and Volume? Breathing – fill in the blanks When you exhale, your diaphragm moves upwards, _______ the volume of your lungs. This cause the pressure in your lungs to _____, forcing air _______ of your lungs. When you inhale, your diaphragm moves downwards, _______ the volume of your lungs. This causes the pressure in your lungs to _____. Since air moves from high to low pressure, air ____ your lungs.
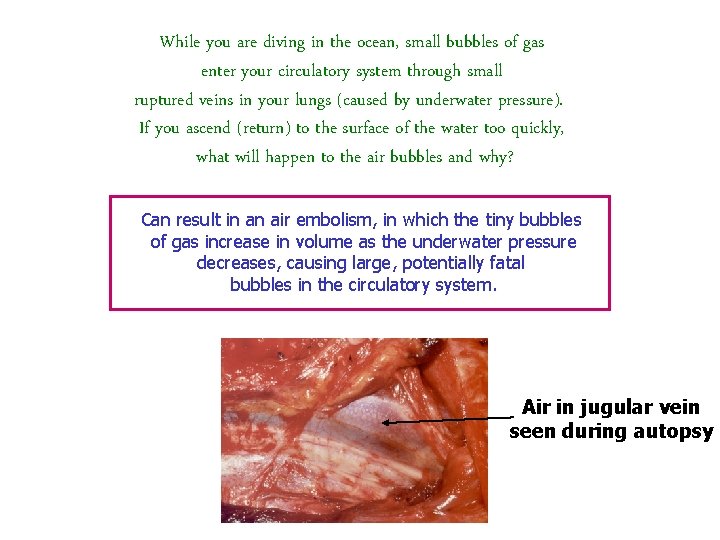
While you are diving in the ocean, small bubbles of gas enter your circulatory system through small ruptured veins in your lungs (caused by underwater pressure). If you ascend (return) to the surface of the water too quickly, what will happen to the air bubbles and why? Can result in an air embolism, in which the tiny bubbles of gas increase in volume as the underwater pressure decreases, causing large, potentially fatal bubbles in the circulatory system. Air in jugular vein seen during autopsy

We need oxygen to breathe. The concentration of oxygen in air is about 21%. You go on a hiking trip up Mt. Kilimanjaro, which is 4600 m tall. About half way to the top, you become short of breath, dizzy, and develop a severe headache. You have altitude sickness because your body is not getting enough oxygen, even though the oxygen level on the mountain is still 21%! What is the cause? (hint: the atmospheric pressure at 3600 m is half that at sea level)

You have a balloon filled with air to near-bursting at sea level. If you were to take this balloon to the top of a very high mountain, ( 3000 m) what might happen and why? (assume the temperature is kept the same)

While the SI unit for pressure is the pascal (Pa), when discussing gases, pressure is measured in atmospheres (atm). Atmospheres are related to atmospheric pressure. 1 atm = 1. 0123 x 105 Pa (101 325 Pa) If 50 m. L of oxygen gas is compressed from 20 atm of pressure to 40 atm of pressure at constant temperature, what is the new volume? (remember P 1 V 1=P 2 V 2) P 1 V 1=P 2 V 2 P 1 = 20 atm V 1 = 50 m. L P 2 = 40 atm V 2 = x Complete Boyle’s Law Worksheet

Charles/Gay-Lussac’s Law Relationship between temperature and volume of gases. At constant pressure, as the temperature of a gas in increases, the volume also increases proportionally. Joseph Gay-Lussac Jacques Charles V 1/T 1=V 2/T 2

To use the equation for Charles/Gay-Lussac’s Law, ALL TEMPERATURES MUST BE CONVERTED INTO KELVIN The Kelvin is the SI unit of temperature Zero Kelvin is called “absolute zero” and is the coldest temperature possible 0 K = -273. 15ºC To convert from Kelvin to Celsius: °C = K − 273. 15 To convert from Celsius to Kelvin: K = °C + 273. 15 Convert from Celsius to Kelvin: Convert from Kelvin to Celsius 50°C - 62 °C -46°C 480°C - 273. 15 K 51 K 0 K 560 K -

Hot Air Balloons: Air is heated inside the balloon, causing it to expand. As the air expands, the balloon gets bigger, and eventually the balloon is able to overcome gravity and rise. Make your own! Standard hot air balloon What will happen if you put a balloon in a car on a hot day? On a cold day? Why?

A gas occupies a volume of 100 m. L at 300 K. At what temperature will the gas have a volume of 200 m. L? (remember V 1/T 1 = V 2/T 2) V 1 – 100 m. L T 1 – 300 K V 2 – 200 m. L T 2 – x 100/300 = 200/x x= 600 K Complete Charles/Gay-Lussac’s Law Worksheet

Cheat Sheet Force - N Area - m 2 Pressure - N/m 2 1 N/m 2 = 1 pascal (Pa) 1000 Pa = 1 kilopascal (k. Pa) 1 kg = 9. 81 N 1 atm = 1. 10132 x 105 Pa P=F/A P 1 V 1=P 2 V 2 Kelvin to Celsius: °C = K − 273. 15 Celsius to Kelvin: K = °C + 273. 15 V 1/T 1 = V 2/T 2
- Slides: 30