Pressure By Prof Ahmed Mostafa Professor of anesthesia

Pressure By Prof. Ahmed Mostafa Professor of anesthesia and ICU

Definitions - Pressure is defined as force per unit area. - The unit of force is the newton (N), 1 N being that force which will accelerate a (frictionless) mass of 1 kg at 1 m/s 2 (in a vacuum). - The SI unit of pressure is the pascal (Pa), - 1 pascal being a force of 1 N acting over an area of 1 m 2.

Definitions - Gravity gives any mass an acceleration of 9. 81 m/s 2, so the force acting on 1 kg is 9. 81 N. - 1 N is therefore equivalent to 102 g. - This is a small pressure, hence the use of the kilopascal (k. Pa) as the main unit of physiological pressure. - Pressures are still quoted in bar [1 bar = 100 k. Pa = 1 atmosphere absolute (ata) = 760 mm. Hg = 760 torr = 1030 cm H 2 O = 14. 7 psi].

The relationship between force and pressure explains why a small syringe can generate far higher pressures than a larger one. The pressure developed is force divided by area. The smaller the area represented by the plunger in the syringe, then the greater is the pressure generated for a given applied force; hence a 2 ml syringe is much more effective than a 10 ml syringe in flushing a blocked intravenous catheter.
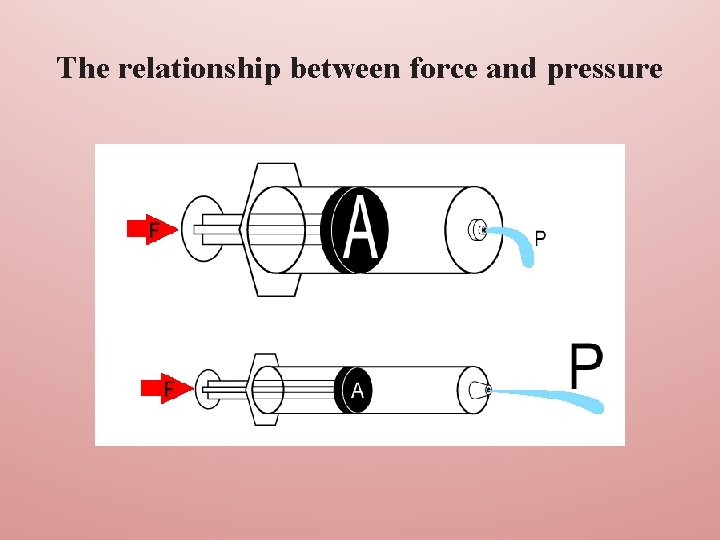
The relationship between force and pressure

Absolute pressure and gauge pressure • An empty gas cylinder has a gauge pressure of zero, but the ambient pressure inside the cylinder is 1 atmosphere. Absolute pressure, therefore, is given by the gauge pressure plus atmospheric pressure.

Measurement of pressure Liquid manometry: the pressure in the column is equal to the product of the height of the column, the density of the liquid and the force of gravity. The width and shape of the column have no effect on the pressure reading. Surface tension provides a potential source of error in columns less than 10 mm in diameter, but in the clinical context of central venous pressure measurement, in which trends are commonly more important than absolute numbers, this is not significant.

Measurement of pressure Aneroid gauges: Examples include the Bourdon gauge for high pressures, which comprises a flattened coiled tube which unwinds as pressures increase. Diaphragm gauges: These are used for many physiological pressures. Pressure changes cause movement in a flexible diaphragm, and these are either read directly or transduced. Electromechanical devices are probably the commonest, employing wire strain gauges whose resistance changes in response to pressure. The sensing diaphragm can also be incorporated as one plate of a capacitor, the other being fixed. The charge that is carried varies with the separation of the plates.

Intravascular pressures (Laplace’s law) In a tube, such as the aorta, the transmural pressure gradient is given by the wall tension divided by the radius (P = T/r). For a sphere the relationship is P = 2 T/r. This pressure relationship explains why an expanding aortic aneurysm is increasingly likely to rupture as the aorta dilates, and why a reservoir bag on a breathing circuit does not cause barotrauma to normal lungs if it is allowed to distend by tightening the valve.

Thank you
- Slides: 10