Presented by Matthew Brons Soomi Hwang Manish Khullar

Presented by: Matthew Brons Soomi Hwang Manish Khullar Sukhjinder Sidhu

Systematic Review Outcomes • Primary outcomes – Incidence of recurrent VTE • Secondary outcomes – Incidence of major bleeding – Mortality

Hierarchy of Outcomes • • 1. Mortality 2. Serious adverse events (ie. major bleeding) 3. Recurrence of VTE 4. Withdrawal due to adverse events

Methods • Inclusion criteria – randomly allocated to different durations of oral anticoagulant therapy – symptomatic VTE

Methods • Exclusion criteria – duplicate or preliminary reports – not using objective tests to confirm diagnosis – if different target INR ranges used – continuous use of other anticoagulants/antiplatelet – no data for thromboembolic events + bleeding – unblinded assessors – no breakdown of minor/major bleeding

Methods • Search Strategy – Medline, Embase, CINAHL, Central, handsearching, conference proceedings – Provided search terms • Data collection – 2 reviewers – assessed adequacy of concealment of allocation

Methods Statistical analysis • odds ratio calculated separately for each trial • ORs combined using the Mantel-Haenszel procedure – weight given to the # of events – fixed treatment effect assumed
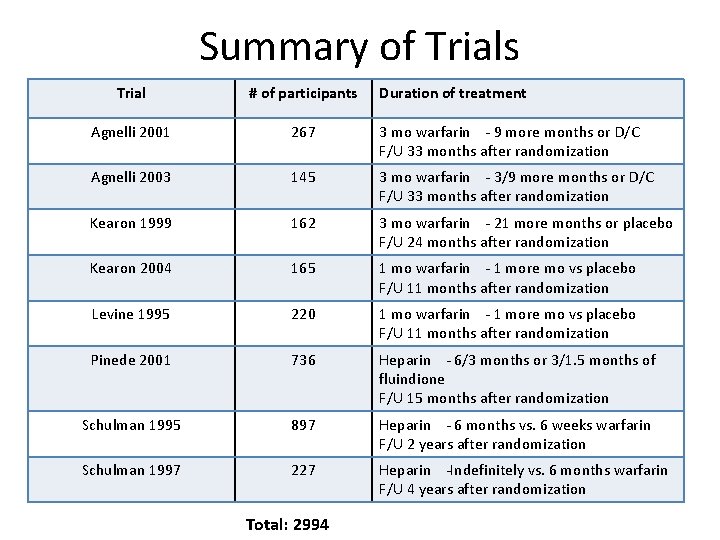
Summary of Trials Trial # of participants Agnelli 2001 267 3 mo warfarin - 9 more months or D/C F/U 33 months after randomization Agnelli 2003 145 3 mo warfarin - 3/9 more months or D/C F/U 33 months after randomization Kearon 1999 162 3 mo warfarin - 21 more months or placebo F/U 24 months after randomization Kearon 2004 165 1 mo warfarin - 1 more mo vs placebo F/U 11 months after randomization Levine 1995 220 1 mo warfarin - 1 more mo vs placebo F/U 11 months after randomization Pinede 2001 736 Heparin - 6/3 months or 3/1. 5 months of fluindione F/U 15 months after randomization Schulman 1995 897 Heparin - 6 months vs. 6 weeks warfarin F/U 2 years after randomization Schulman 1997 227 Heparin -Indefinitely vs. 6 months warfarin F/U 4 years after randomization Total: 2994 Duration of treatment

The Analyses of Studies
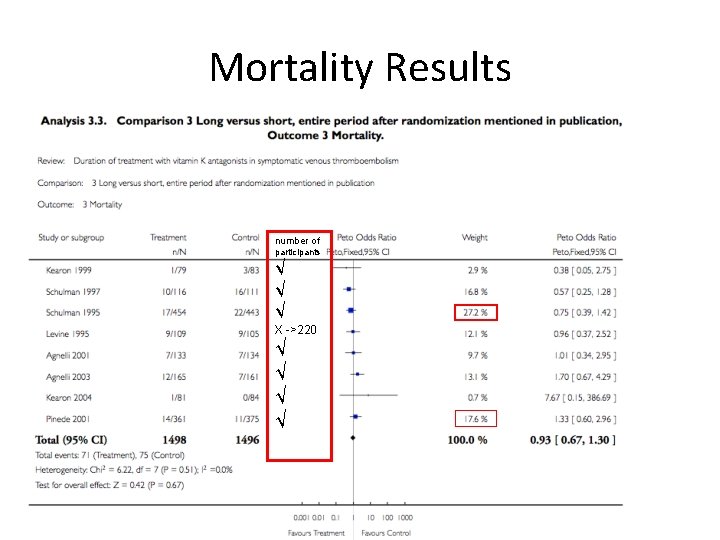
Mortality Results number of participants √ √ √ X ->220 √ √

Serious Adverse Events number of participants √ √ √ X ->220 √ √

Recurrence of VTE number of participants √ √ √ X ->220 √ √

Systematic Review Conclusion • Prolonged treatment reduces the risk of recurrent VTE for as long as it is used • Efficacy during continuing treatment decreases while the risk for major bleeding remains • Our thoughts?

Questions?
- Slides: 14