Presented by Kris V Kowdley MD Director Liver

Presented by Kris V. Kowdley, MD Director Liver Care Network and Organ Care Research Swedish Medical Center Seattle, Washington Seth N. Sclair, MD Clinical Assistant Professor Division of Gastroenterology and Liver Disease Case Western Reserve University School of Medicine University Hospitals Cleveland Medical Center Cleveland, Ohio

Content Areas • Diagnostic considerations • Initial treatment with ursodeoxycholic acid • Assessing response to ursodeoxycholic acid • Obeticholic acid • Investigational treatments • Managing symptoms and complications

Diagnostic Considerations Differential for Cholestatic Liver Biochemistry 3 Spectrum of Autoimmune Liver Injuries 1 • Autoimmune hepatitis 1 • Primary biliary cirrhosis 1 • Primary sclerosing cholangitis 1 • Ig. G 4 -related disease 2 • • • Drug-induced liver injury Inherited cholestasis Idiopathic ductopenia Malignant infiltration Nonalcoholic fatty liver disease Obstructive biliary lesion Primary biliary cholangitis Primary sclerosing cholangitis Sarcoidosis 1. Trivedi PJ, et al. Aliment Pharmacol Ther. 2012; 36: 517 -533. 2. Joshi D, et al. Aliment Pharmacol Ther. 2014; 40: 1251 -1261. 3. Hirschfield GM, et al. Best Pract Res Clin Gastroenterol. 2011; 25: 701 -712.

What Are the Diagnostic Markers in Primary Biliary Cholangitis? • Primary biliary cholangitis (PBC) is the most common adult autoimmune liver disease • The overwhelming majority of patients are women in middle age who have circulating antimitochondrial antibodies • Cholestasis is usually reflected as a predominant rise in alkaline phosphatase level • At presentation, most patients are largely asymptomatic Over time, however, symptoms such as pruritus and fatigue significantly impact patient quality of life

PBC Phenotype Age Usually > 45 years Gender Female > Male (9: 1) Serology AMA in ~95%; disease-specific ANA in ~30 -50%; ASMA may be present Immunoglobulin MRCP Ig. M typically elevated Normal Liver Histology Lymphocytic infiltrate; inflammatory duct lesion; granuloma may be present Coexisting IBD Not typical AMA, antimitochondrial antibody; ANA, antinuclear antibody; ASMA, anti-smooth-muscle antibody; IBD, inflammatory bowel disease; MRCP, magnetic resonance cholangiography; PBC, primary biliary cholangitis. Trivedi PJ, et al. Aliment Pharmacol Ther. 2012; 36: 517 -533.

Take a Good History • Symptom burden 1, 2 ◦ ◦ ◦ Pruritus Fatigue Sicca syndrome: dry eyes/mouth Abdominal pain Arthralgias Remember: some patients remain asymptomatic • Relevant medical history 1 ◦ ◦ Autoimmunity (personal or family) Smoking Recurrent urinary tract infection Pruritus during pregnancy 1. Hirschfield GM, et al. Best Pract Res Clin Gastroenterol. 2011; 25: 701 -712. 2. Trivedi PJ, et al. Aliment Pharmacol Ther. 2012; 36: 517 -533.

Interpretation of AMA, ANA, and Immunoglobulin Testing in PBC • AMA ◦ Positive in >90% of patients with PBC, depending on assay 1 ◦ In the correct context, AMA reactivity, with an elevated ALP and no significant elevation in AST, is associated with a >95% PPV of histologic PBC 2 • ANA ◦ 2 ANA immunofluorescent patterns are specific to PBC: multiple nuclear dots and perinuclear/rim-like membranous 3 ◦ Automated ANA assays will likely not detect these reactivities ◦ Laboratories should perform immunofluorescence if ELISA-based assays for gp 210 and sp 100 are not available • Immunoglobulins ◦ Elevated Ig. M is a sensitive but not specific characteristic of PBC 1 ◦ Elevated Ig. G is primarily observed in AIH 1 AIH, autoimmune hepatitis; ALP, alkaline phosphatase; AMA, antimitochondrial antibody; ANA, antinuclear antibody; AST, aspartate aminotransferase; ELISA, enzymelinked immunosorbent assay; Ig. G, immunoglobulin G; Ig. M, immunoglobulin M; PPV, positive predictive value. 1. Trivedi PJ, et al. Aliment Pharmacol Ther. 2012; 36: 517 -533. 2. Zein CO, et al. Clin Gastroenterol Hepatol. 2003; 1: 89 -95. 3. Zeman MV, et al. Can J Gastroenterol. 2010; 24: 225– 231.
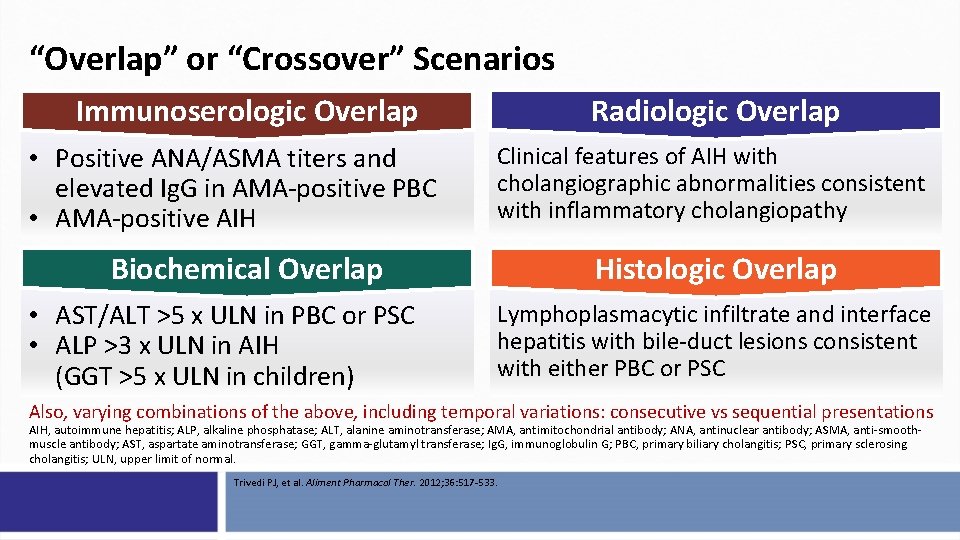
“Overlap” or “Crossover” Scenarios Radiologic Overlap Immunoserologic Overlap • Positive ANA/ASMA titers and elevated Ig. G in AMA-positive PBC • AMA-positive AIH Clinical features of AIH with cholangiographic abnormalities consistent with inflammatory cholangiopathy Biochemical Overlap • AST/ALT >5 x ULN in PBC or PSC • ALP >3 x ULN in AIH (GGT >5 x ULN in children) Histologic Overlap Lymphoplasmacytic infiltrate and interface hepatitis with bile-duct lesions consistent with either PBC or PSC Also, varying combinations of the above, including temporal variations: consecutive vs sequential presentations AIH, autoimmune hepatitis; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AMA, antimitochondrial antibody; ANA, antinuclear antibody; ASMA, anti-smoothmuscle antibody; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase; Ig. G, immunoglobulin G; PBC, primary biliary cholangitis; PSC, primary sclerosing cholangitis; ULN, upper limit of normal. Trivedi PJ, et al. Aliment Pharmacol Ther. 2012; 36: 517 -533.

Predictive Significance of Alkaline Phosphatase and Bilirubin Values Survival at 15 Years (%) 100 80 [VALUE]% 60 40 [VALUE]% 20 0 1 2 ALP ≤ 2 x ULN ALP, alkaline phosphatase; ULN, upper limit of normal. Lammers WJ, et al. Gastroenterology. 2014; 147: 1338 -1349. 3 4 ALP >2 x ULN

Influence of PBC-Specific Antinuclear Antibodies on Disease Prognosis • Retrospective/prospective Japanese cohort study to assess significance of ANAs for progression of PBC (N = 276) ◦ Prevalence of anti-gp 210 in PBC = 26. 1% • Anti-gp 210 antibodies strongest ANA predictor for progression to end-stage hepatic failure ◦ Odds ratio 33. 777 (95% CI, 5. 930− 636. 745) • Frequency of death significantly higher in patients positive for anti-gp 210 than negative (P = 1. 3 x 10 -7) ANA, antinuclear antibody; CI, confidence interval. Nakamura M, et al. Hepatology. 2007; 45: 118 -127.
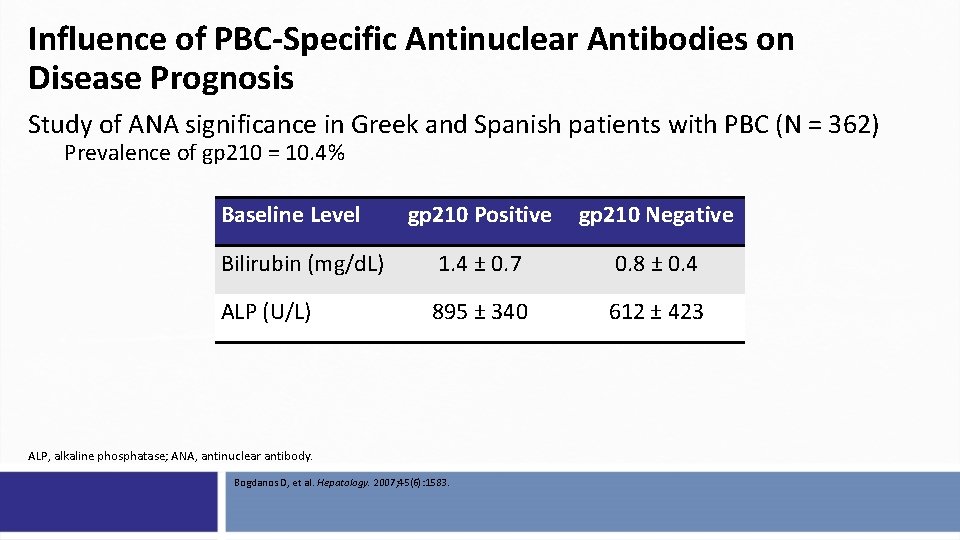
Influence of PBC-Specific Antinuclear Antibodies on Disease Prognosis Study of ANA significance in Greek and Spanish patients with PBC (N = 362) Prevalence of gp 210 = 10. 4% Baseline Level gp 210 Positive gp 210 Negative Bilirubin (mg/d. L) 1. 4 ± 0. 7 0. 8 ± 0. 4 ALP (U/L) 895 ± 340 612 ± 423 ALP, alkaline phosphatase; ANA, antinuclear antibody. Bogdanos D, et al. Hepatology. 2007; 45(6): 1583.
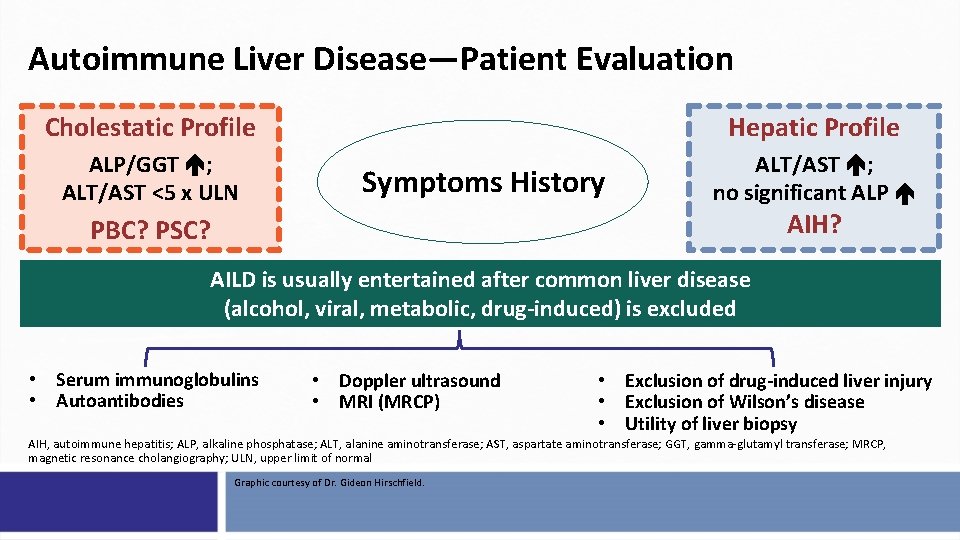
Autoimmune Liver Disease—Patient Evaluation Cholestatic Profile Hepatic Profile ALP/GGT ; ALT/AST <5 x ULN ALT/AST ; no significant ALP Symptoms History AIH? PBC? PSC? AILD is usually entertained after common liver disease (alcohol, viral, metabolic, drug-induced) is excluded • Serum immunoglobulins • Autoantibodies • Doppler ultrasound • MRI (MRCP) • Exclusion of drug-induced liver injury • Exclusion of Wilson’s disease • Utility of liver biopsy AIH, autoimmune hepatitis; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase; MRCP, magnetic resonance cholangiography; ULN, upper limit of normal Graphic courtesy of Dr. Gideon Hirschfield.
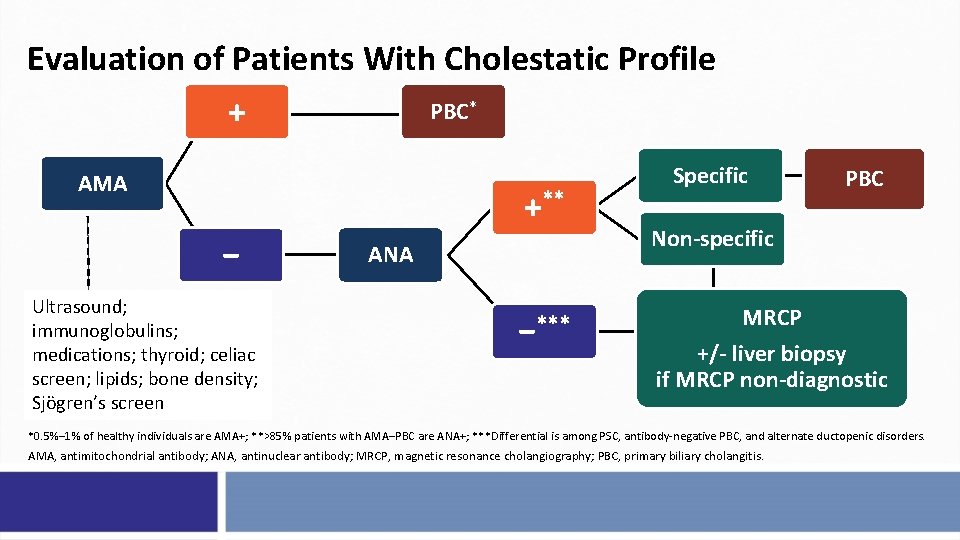
Evaluation of Patients With Cholestatic Profile PBC* + AMA +** − Ultrasound; immunoglobulins; medications; thyroid; celiac screen; lipids; bone density; Sjögren’s screen ANA −*** Specific PBC Non-specific MRCP +/- liver biopsy if MRCP non-diagnostic *0. 5%– 1% of healthy individuals are AMA+; **>85% patients with AMA–PBC are ANA+; ***Differential is among PSC, antibody-negative PBC, and alternate ductopenic disorders. AMA, antimitochondrial antibody; ANA, antinuclear antibody; MRCP, magnetic resonance cholangiography; PBC, primary biliary cholangitis.

Overall Management of PBC • Start ursodeoxycholic acid (UDCA) and assess response • Determine stage of disease ◦ Institute HCC and variceal screening for cirrhotics • Assess and address ◦ ◦ ◦ Osteoporosis Fat-soluble vitamin deficiency Fatigue Pruritus Sicca syndrome • Be aware of extrahepatic manifestations ◦ Common: thyroid disease, renal disease, gallstones, arthritis ◦ Uncommon: lichen planus, ulcerative colitis, anemias HCC, hepatocellular carcinoma; PBC, primary biliary cirrhosis. Lindor KD, et al. Hepatology. 2009; 50: 291 -308.

UDCA—Current Standard-of-Care for Primary Biliary Cholangitis • UDCA is 1 of 2 therapies approved by FDA for PBC Obeticholic acid approved in combination with UDCA in adults with an inadequate response to UDCA, or as monotherapy in adults unable to tolerate UDCA • Recommended adult dosage is 13− 15 mg/kg/day • Typically administered in 2 divided doses FDA, US Food and Drug Administration; PBC, primary biliary cholangitis; UDCA, ursodeoxycholic acid or ursodiol. Lindor KD, et al. Hepatology. 2009; 50: 291 -308.
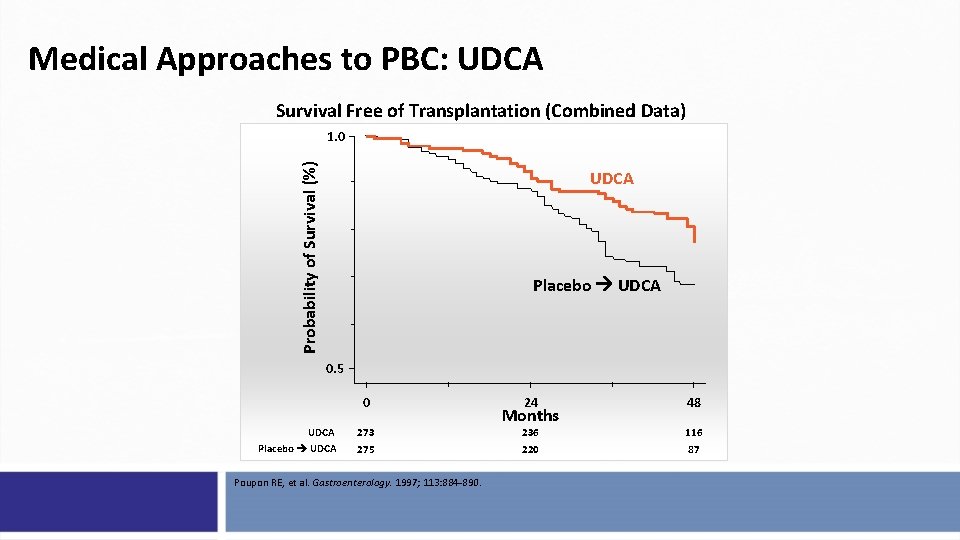
Medical Approaches to PBC: UDCA Survival Free of Transplantation (Combined Data) Probability of Survival (%) 1. 0 UDCA Placebo UDCA 0. 5 0 UDCA Placebo UDCA 273 275 Poupon RE, et al. Gastroenterology. 1997; 113: 884 -890. 24 48 236 220 116 87 Months
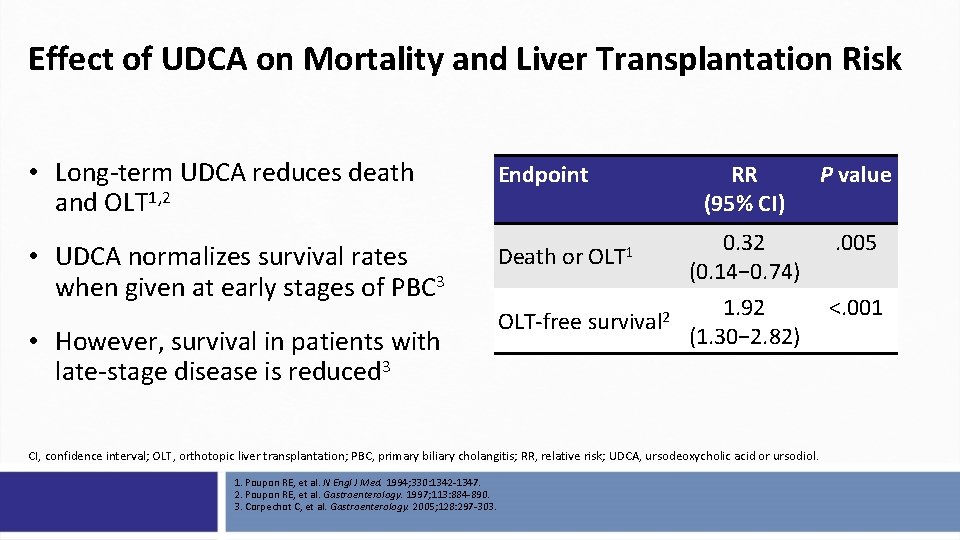
Effect of UDCA on Mortality and Liver Transplantation Risk • Long-term UDCA reduces death and OLT 1, 2 • UDCA normalizes survival rates when given at early stages of PBC 3 • However, survival in patients with late-stage disease is reduced 3 Endpoint RR (95% CI) P value Death or OLT 1 0. 32 (0. 14− 0. 74) . 005 OLT-free survival 2 1. 92 (1. 30− 2. 82) <. 001 CI, confidence interval; OLT, orthotopic liver transplantation; PBC, primary biliary cholangitis; RR, relative risk; UDCA, ursodeoxycholic acid or ursodiol. 1. Poupon RE, et al. N Engl J Med. 1994; 330: 1342 -1347. 2. Poupon RE, et al. Gastroenterology. 1997; 113: 884 -890. 3. Corpechot C, et al. Gastroenterology. 2005; 128: 297 -303.

What Are Appropriate Baseline Factors and Treatment Endpoints to Assess Risk of Progression in PBC? • High confidence and applicability ◦ Baseline and on-treatment ALP, bilirubin ◦ Baseline and on-treatment AST/platelet ratio ◦ On-treatment biochemical response criteria • Intermediate confidence and applicability ◦ Presenting age ◦ Baseline disease-specific ANA ◦ Baseline and on-treatment transient elastography • Indeterminate confidence and applicability ◦ Gender and baseline symptom profile ◦ Baseline and on-treatment novel histologic scores ◦ On-treatment direct portal pressure measurement ALP, alkaline phosphatase; ANA, antinuclear antibody; AST, aspartate aminotransferase; ELF, enhanced liver fibrosis; PBC, primary biliary cirrhosis. Trivedi PJ, et al. Hepatology. 2016; 63(2): 644 -659.

Established Response Criteria Models for UDCA Barcelona 1 ALP decreased by >40% from baseline or normalized after 1 year UDCA Paris-I 2 All 3 of the following: ALP ≤ 3 x ULN; AST ≤ 2 x ULN; and bilirubin ≤ 1 mg/d. L after 1 year UDCA Rotterdam 3 Albumin and bilirubin normalization when 1 or both were abnormal at baseline; albumin OR bilirubin normalization when both were abnormal at baseline after 1 year UDCA (2006) (2008) (2009) Toronto 4 (2010) ALP <1. 67 x ULN after 2 years UDCA ALP, alkaline phosphatase; AST, aspartate aminotransferase; UDCA, ursodeoxycholic acid or ursodiol; ULN, upper limit of normal. 1. Parés A, et al. Gastroenterology. 2006; 130: 715 -720. 2. Corpechot C, et al. Hepatology. 2008; 48: 871 -877. 3. Kuiper EM, et al. Gastroenterology. 2009; 136: 1281 -1287. 4. Kumagi T, et al. Am J Gastroenterol. 2010; 105: 2186 -2194.

Modifications of Biochemical Response Criteria Models Paris-II 1 (2011) All 3 of the following: ALP ≤ 1. 5 x ULN; AST ≤ 1. 5 x ULN; and bilirubin ≤ 1 mg/d. L after 1 year UDCA Early Biochemical Response 2 Barcelona, Paris-I, or Toronto criteria met at 6 months UDCA (2013) ALP, alkaline phosphatase; AST, aspartate aminotransferase; UDCA, ursodeoxycholic acid or ursodiol; ULN, upper limit of normal. 1. Corpechot C. J Hepatol. 2011; 55: 1361 -1367. 2. Zhang LN, et al. Hepatology. 2013; 58: 264 -272.

Optimized Response Criteria Models Biochemical + APRI 1 Biochemical response (Barcelona, Paris-I/II, or Toronto) and APRI ≤ 0. 54 (2014) after 1 year UDCA UK-PBC Risk Score 2 Prognostic index comprising baseline albumin and platelet count, plus bilirubin, ALT or AST, and ALP after 1 year UDCA GLOBE Score 3 Prognostic index comprising baseline age, and bilirubin, ALP, albumin, and platelet count after 1 year UDCA (2015) ALP, alkaline phosphatase; ALT, alanine aminotransferase; APRI, AST/platelet ratio index; AST, aspartate aminotransferase; UDCA, ursodeoxycholic acid or ursodiol. 1. Trivedi PJ, et al. J Hepatol. 2014; 60: 1249 -1258. 2. Carbone M, et al. Hepatology. 2016; 63(3): 930 -950. 3. Lammers WJ, et al. Gastroenterology. 2015; 149(7): 1804 -1812.

GLOBE Score Calculation Score comprising ◦ age at start of UDCA ◦ beta coefficients of identified variables ◦ natural logarithm transformation of BILI, ALP, ALB, and PLT after 1 year of UDCA ◦ baseline survival estimate (0. 044378 * age at start of UDCA therapy + 0. 93982 * NL(BILIx. ULN) at 1 year)) + (0. 335648 * NL(ALPx. ULN at 1 year))— 2. 266708 * ALBx. LLN at 1 year— 0. 002581 * PLT at 1 year) + 1. 216865 ALB, albumin; ALP, alkaline phosphatase; BILI, bilirubin, LLN, lower limit of normal; NL, normal logarithm; PLT, platelet count; ULN, upper limit of normal. Online calculator available at: http: //www. globalpbc. com/globe. Lammers WJ, et al. Gastroenterology. 2015; 149(7): 1804 -1812.
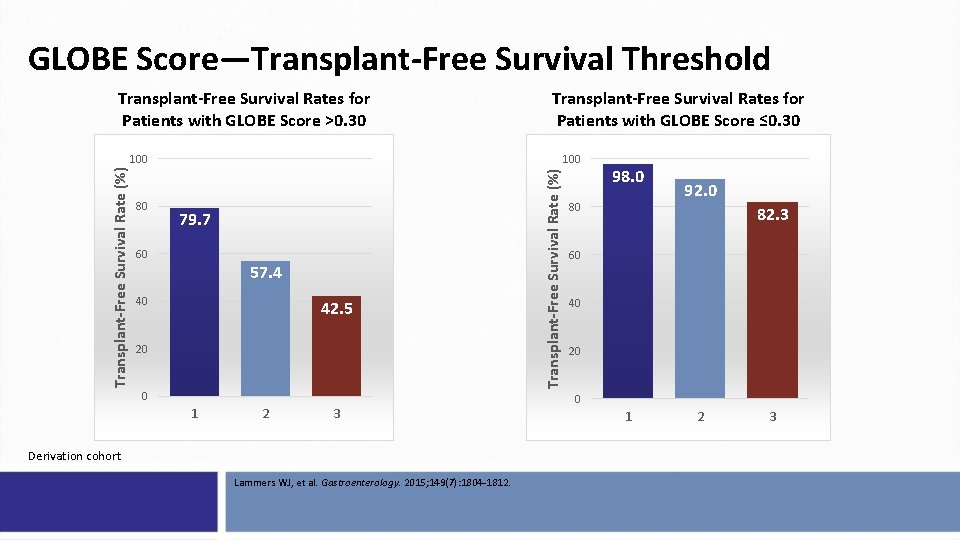
GLOBE Score—Transplant-Free Survival Threshold Transplant-Free Survival Rates for Patients with GLOBE Score >0. 30 Transplant-Free Survival Rates for Patients with GLOBE Score ≤ 0. 30 80 100 79. 7 60 57. 4 40 42. 5 20 0 1 2 3 Derivation cohort Lammers WJ, et al. Gastroenterology. 2015; 149(7): 1804 -1812. Transplant-Free Survival Rate (%) 100 98. 0 80 92. 0 82. 3 60 40 20 0 1 2 3
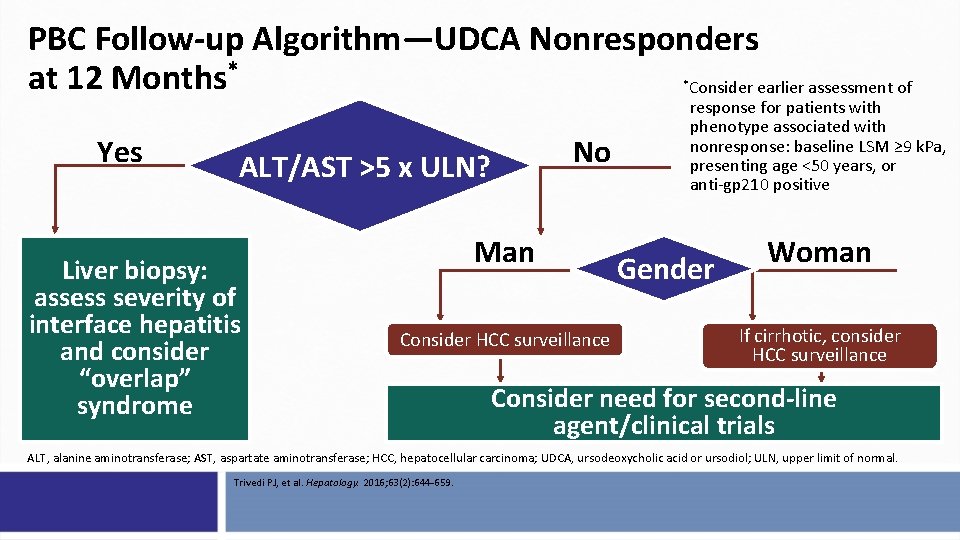
PBC Follow-up Algorithm—UDCA Nonresponders at 12 Months* Consider earlier assessment of * Yes No ALT/AST >5 x ULN? Liver biopsy: assess severity of interface hepatitis and consider “overlap” syndrome Man Consider HCC surveillance response for patients with phenotype associated with nonresponse: baseline LSM ≥ 9 k. Pa, presenting age <50 years, or anti-gp 210 positive Gender Woman If cirrhotic, consider HCC surveillance Consider need for second-line agent/clinical trials ALT, alanine aminotransferase; AST, aspartate aminotransferase; HCC, hepatocellular carcinoma; UDCA, ursodeoxycholic acid or ursodiol; ULN, upper limit of normal. Trivedi PJ, et al. Hepatology. 2016; 63(2): 644 -659.
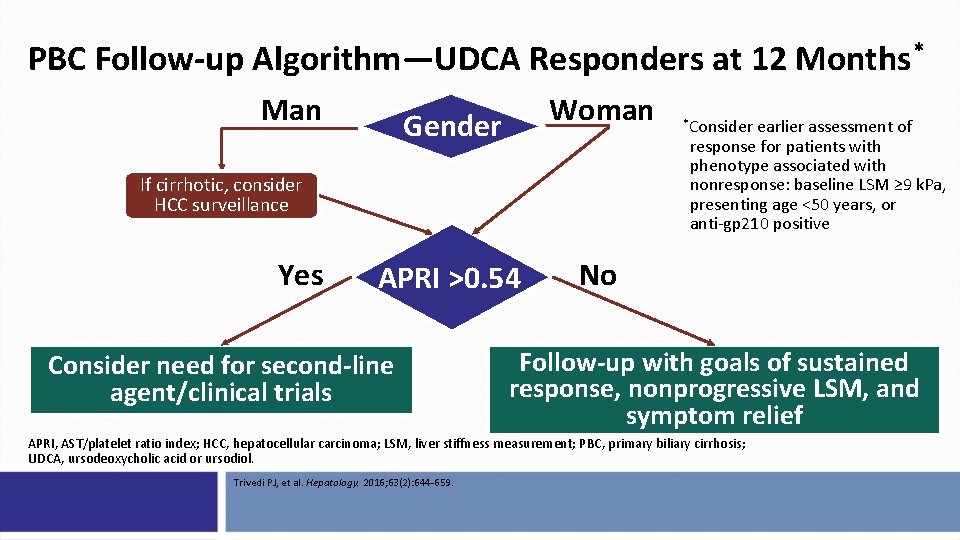
PBC Follow-up Algorithm—UDCA Responders at 12 Months* Man Woman Gender If cirrhotic, consider HCC surveillance Yes APRI >0. 54 Consider need for second-line agent/clinical trials *Consider earlier assessment of response for patients with phenotype associated with nonresponse: baseline LSM ≥ 9 k. Pa, presenting age <50 years, or anti-gp 210 positive No Follow-up with goals of sustained response, nonprogressive LSM, and symptom relief APRI, AST/platelet ratio index; HCC, hepatocellular carcinoma; LSM, liver stiffness measurement; PBC, primary biliary cirrhosis; UDCA, ursodeoxycholic acid or ursodiol. Trivedi PJ, et al. Hepatology. 2016; 63(2): 644 -659.
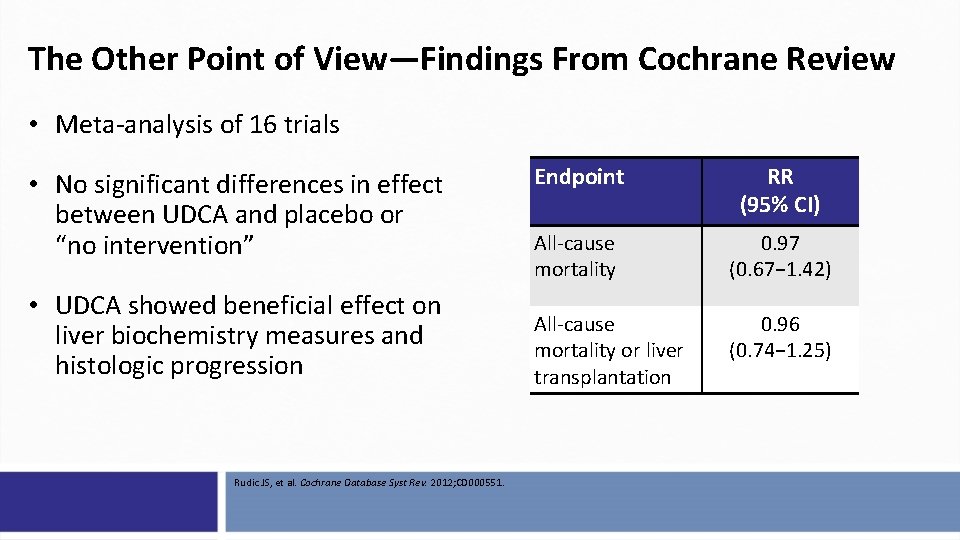
The Other Point of View—Findings From Cochrane Review • Meta-analysis of 16 trials • No significant differences in effect between UDCA and placebo or “no intervention” • UDCA showed beneficial effect on liver biochemistry measures and histologic progression Rudic JS, et al. Cochrane Database Syst Rev. 2012; CD 000551. Endpoint RR (95% CI) All-cause mortality 0. 97 (0. 67− 1. 42) All-cause mortality or liver transplantation 0. 96 (0. 74− 1. 25)

Reported Incidence of UDCA Treatment Failure Study Pells et al, 20131 (UK-PBC group) Treatment Failure (%) • 60% of patients presenting at age <40 years • 10% of patients presenting at age >70 years Corpechot et al, 20112 13%– 37%* Kuiper et al, 20093 34%– 38%* Corpechot et al, 20084 35%– 39%* *Depending on criteria used. 1. Pells G, et al. J Hepatol. 2013; 59: 67 -73. 2. Corpechot C, et al. J Hepatol. 2011; 55: 1361 -1367. 3. Kuiper EM, et al. Gastroenterology. 2009; 136: 1281 -1287. 4. Corpechot C, et al. Hepatology. 2008; 48: 871 -877.

What Can Clinicians Do Next for Patients With Suboptimal Response to UDCA? • Query patient for adherence 1 • Barriers to adherence: weight gain, loose stools, hair loss • Confirm UDCA dosage 13– 15 mg/kg 1 Doubling UDCA dose has not shown benefit 2 • Check for comorbid liver disease 1 • Avoid coadministration of bile acid sequestrant 1 • Second-line therapy with obeticholic acid 1. Lindor KD, et al. Hepatology. 2009; 50: 291 -308. 2. Angulo P, et al. Am J Gastroenterol. 2001; 96: 3152 -3157.
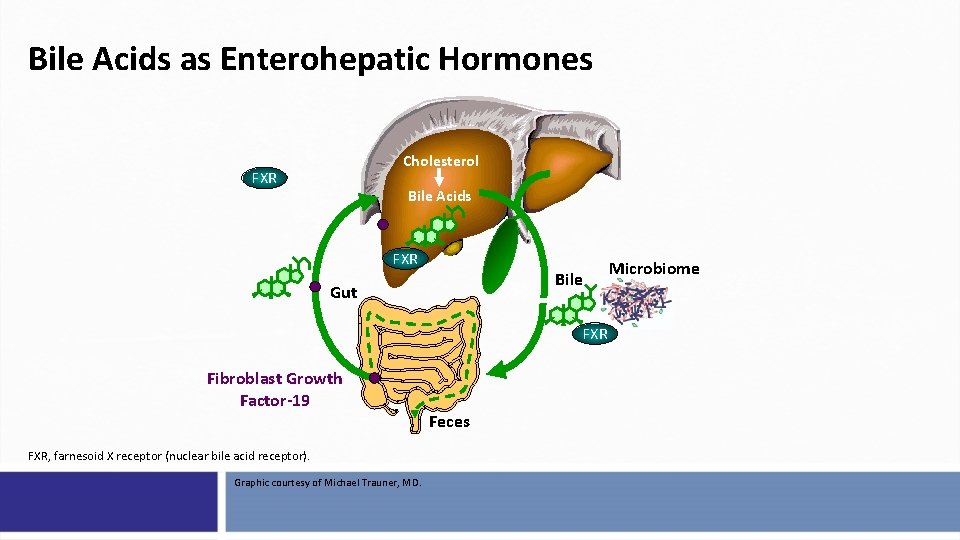
Bile Acids as Enterohepatic Hormones Cholesterol FXR Bile Acids FXR Bile Gut FXR Fibroblast Growth Factor-19 FXR, farnesoid X receptor (nuclear bile acid receptor). Graphic courtesy of Michael Trauner, MD. Feces Microbiome
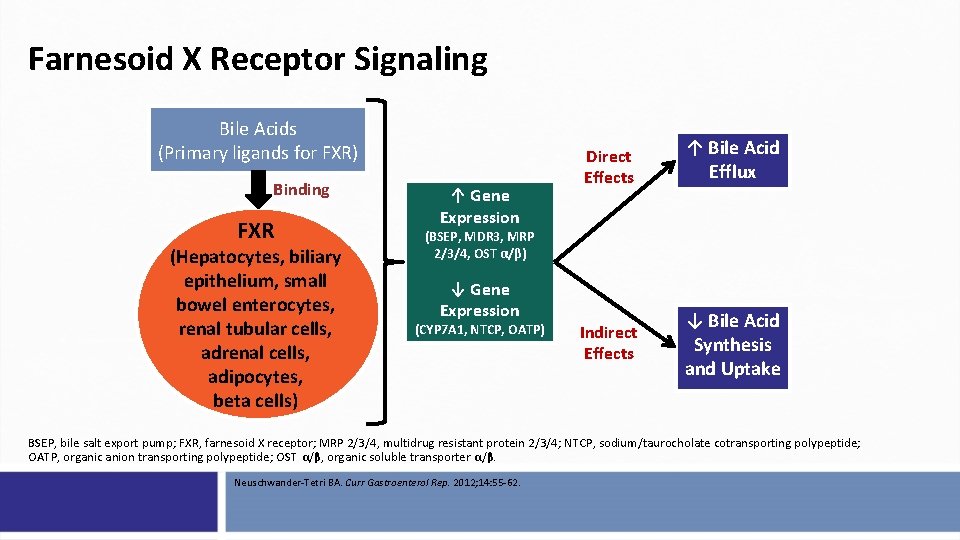
Farnesoid X Receptor Signaling Bile Acids (Primary ligands for FXR) Binding FXR (Hepatocytes, biliary epithelium, small bowel enterocytes, renal tubular cells, adrenal cells, adipocytes, beta cells) ↑ Gene Expression Direct Effects ↑ Bile Acid Efflux Indirect Effects ↓ Bile Acid Synthesis and Uptake (BSEP, MDR 3, MRP 2/3/4, OST α/β) ↓ Gene Expression (CYP 7 A 1, NTCP, OATP) BSEP, bile salt export pump; FXR, farnesoid X receptor; MRP 2/3/4, multidrug resistant protein 2/3/4; NTCP, sodium/taurocholate cotransporting polypeptide; OATP, organic anion transporting polypeptide; OST α/β, organic soluble transporter α/β. Neuschwander-Tetri BA. Curr Gastroenterol Rep. 2012; 14: 55 -62.
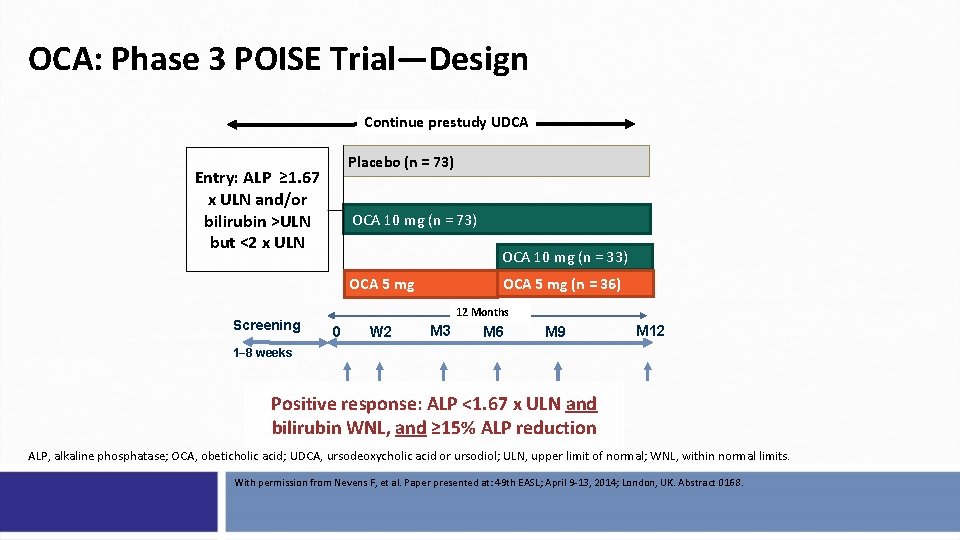
OCA: Phase 3 POISE Trial—Design Continue prestudy UDCA Placebo (n = 73) Entry: ALP ≥ 1. 67 x ULN and/or bilirubin >ULN but <2 x ULN OCA 10 mg (n = 73) OCA 10 mg (n = 33) OCA 5 mg (n = 36) OCA 5 mg Screening 12 Months 0 W 2 M 3 M 6 M 9 M 12 1− 8 weeks Positive response: ALP <1. 67 x ULN and bilirubin WNL, and ≥ 15% ALP reduction ALP, alkaline phosphatase; OCA, obeticholic acid; UDCA, ursodeoxycholic acid or ursodiol; ULN, upper limit of normal; WNL, within normal limits. With permission from Nevens F, et al. Paper presented at: 49 th EASL; April 9 -13, 2014; London, UK. Abstract 0168.
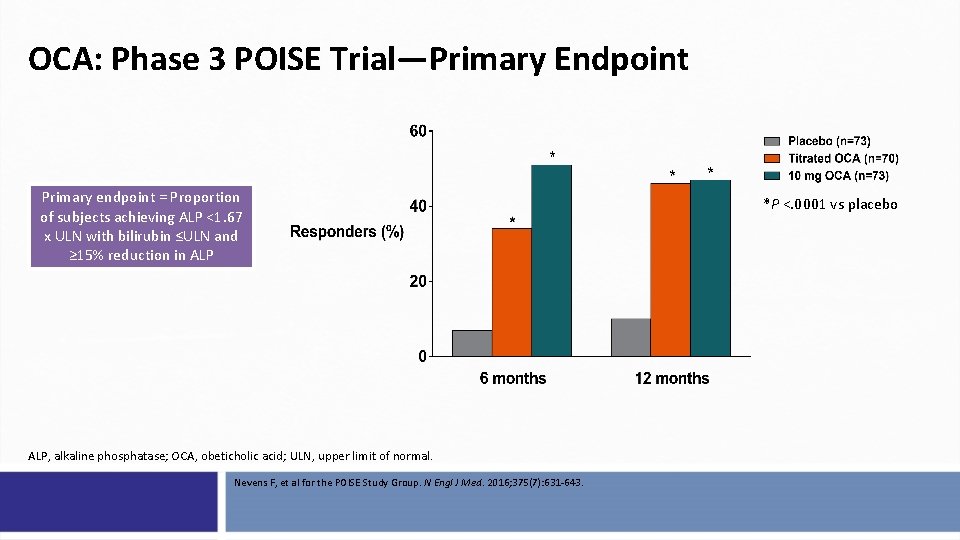
OCA: Phase 3 POISE Trial—Primary Endpoint Primary endpoint = Proportion of subjects achieving ALP <1. 67 x ULN with bilirubin ≤ULN and ≥ 15% reduction in ALP, alkaline phosphatase; OCA, obeticholic acid; ULN, upper limit of normal. Nevens F, et al for the POISE Study Group. N Engl J Med. 2016; 375(7): 631 -643. *P <. 0001 vs placebo
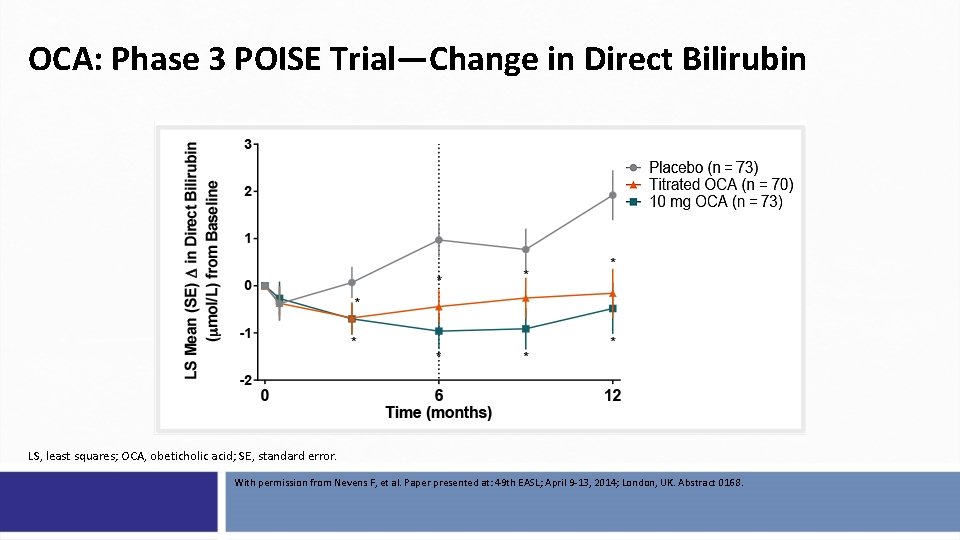
OCA: Phase 3 POISE Trial—Change in Direct Bilirubin LS, least squares; OCA, obeticholic acid; SE, standard error. With permission from Nevens F, et al. Paper presented at: 49 th EASL; April 9 -13, 2014; London, UK. Abstract 0168.
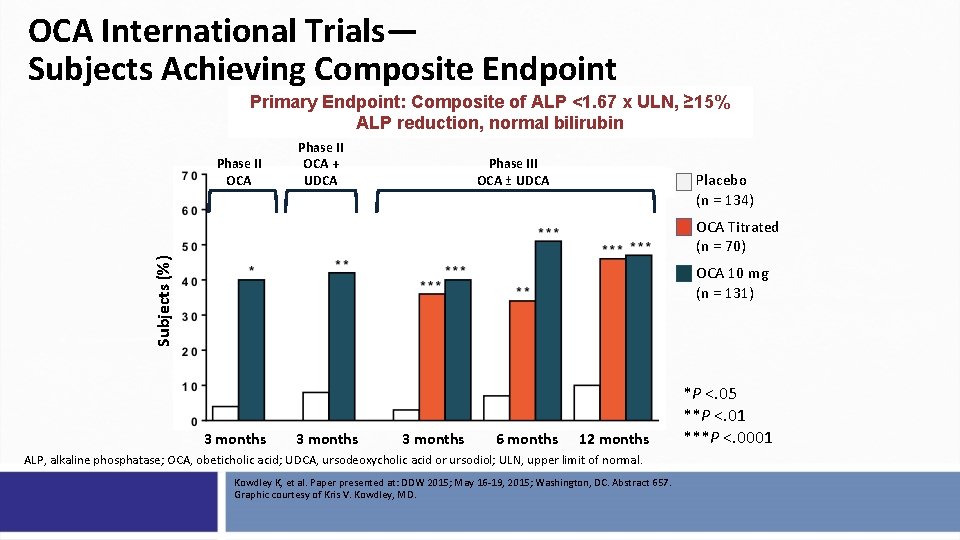
OCA International Trials— Subjects Achieving Composite Endpoint Primary Endpoint: Composite of ALP <1. 67 x ULN, ≥ 15% ALP reduction, normal bilirubin Phase II OCA + UDCA Phase III OCA ± UDCA Placebo (n = 134) Subjects (%) OCA Titrated (n = 70) OCA 10 mg (n = 131) 3 months 6 months 12 months ALP, alkaline phosphatase; OCA, obeticholic acid; UDCA, ursodeoxycholic acid or ursodiol; ULN, upper limit of normal. Kowdley K, et al. Paper presented at: DDW 2015; May 16 -19, 2015; Washington, DC. Abstract 657. Graphic courtesy of Kris V. Kowdley, MD. *P <. 05 **P <. 01 ***P <. 0001
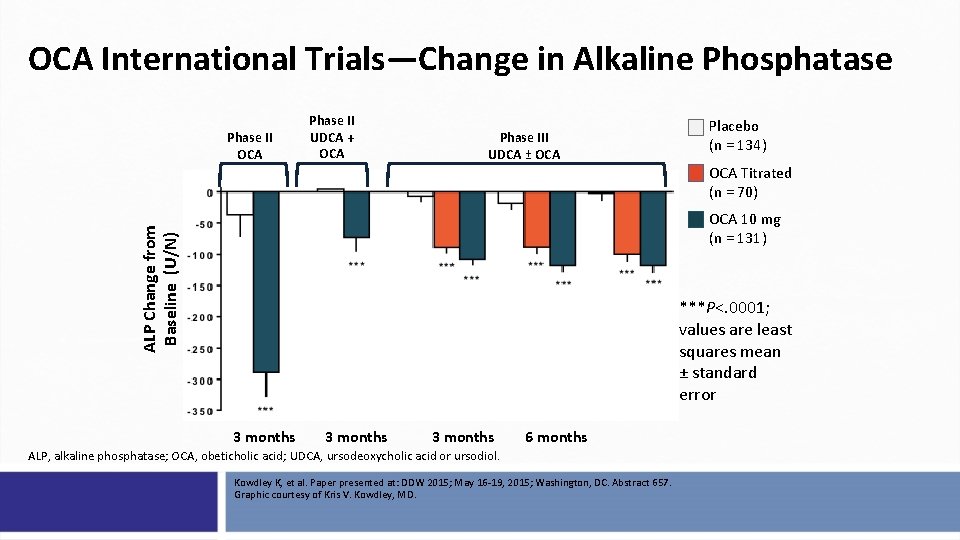
OCA International Trials—Change in Alkaline Phosphatase Phase II OCA Phase II UDCA + OCA Phase III UDCA ± OCA Placebo (n = 134) OCA Titrated (n = 70) ALP Change from Baseline (U/N) OCA 10 mg (n = 131) ***P<. 0001; values are least squares mean ± standard error 3 months 6 months ALP, alkaline phosphatase; OCA, obeticholic acid; UDCA, ursodeoxycholic acid or ursodiol. Kowdley K, et al. Paper presented at: DDW 2015; May 16 -19, 2015; Washington, DC. Abstract 657. Graphic courtesy of Kris V. Kowdley, MD.
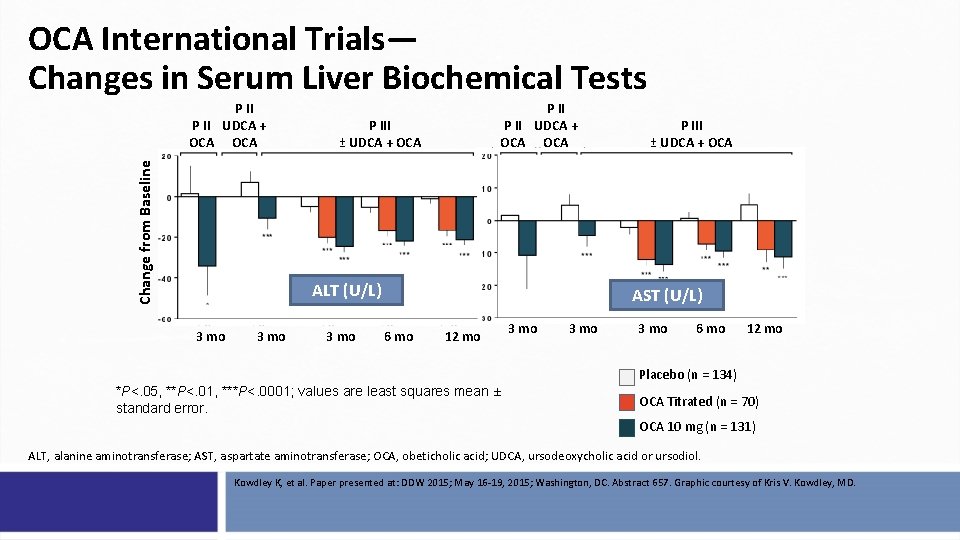
OCA International Trials— Changes in Serum Liver Biochemical Tests Change from Baseline P II UDCA + OCA OCA P III ± UDCA + OCA ALT (U/L) 3 mo P III ± UDCA + OCA AST (U/L) 6 mo 12 mo 3 mo 6 mo 12 mo Placebo (n = 134) *P<. 05, **P<. 01, ***P<. 0001; values are least squares mean ± standard error. OCA Titrated (n = 70) OCA 10 mg (n = 131) ALT, alanine aminotransferase; AST, aspartate aminotransferase; OCA, obeticholic acid; UDCA, ursodeoxycholic acid or ursodiol. Kowdley K, et al. Paper presented at: DDW 2015; May 16 -19, 2015; Washington, DC. Abstract 657. Graphic courtesy of Kris V. Kowdley, MD.
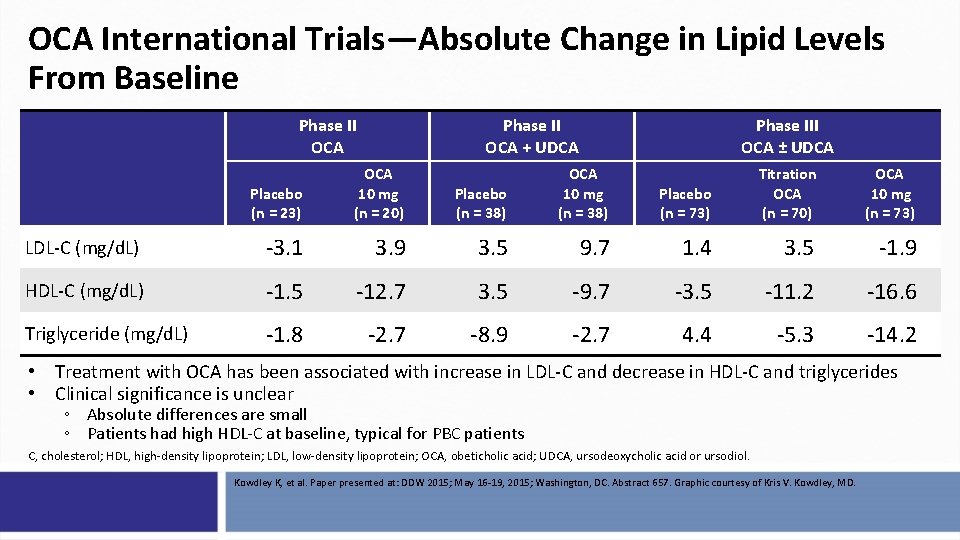
OCA International Trials—Absolute Change in Lipid Levels From Baseline Phase II OCA + UDCA Placebo (n = 23) OCA 10 mg (n = 20) LDL-C (mg/d. L) -3. 1 HDL-C (mg/d. L) Triglyceride (mg/d. L) Phase III OCA ± UDCA Placebo (n = 38) OCA 10 mg (n = 38) Placebo (n = 73) Titration OCA (n = 70) OCA 10 mg (n = 73) 3. 9 3. 5 9. 7 1. 4 3. 5 -1. 9 -1. 5 -12. 7 3. 5 -9. 7 -3. 5 -11. 2 -16. 6 -1. 8 -2. 7 -8. 9 -2. 7 4. 4 -5. 3 -14. 2 • Treatment with OCA has been associated with increase in LDL-C and decrease in HDL-C and triglycerides • Clinical significance is unclear ◦ Absolute differences are small ◦ Patients had high HDL-C at baseline, typical for PBC patients C, cholesterol; HDL, high-density lipoprotein; LDL, low-density lipoprotein; OCA, obeticholic acid; UDCA, ursodeoxycholic acid or ursodiol. Kowdley K, et al. Paper presented at: DDW 2015; May 16 -19, 2015; Washington, DC. Abstract 657. Graphic courtesy of Kris V. Kowdley, MD.

OCA International Trials—Summary of Adverse Events • ≥ 1 Treatment-emergent serious adverse event ◦ OCA 10 mg (6%) • None drug-related ◦ Placebo (4%) • Pruritus was the most common adverse event reported across all treatment groups ◦ Most pruritus treatment-emergent adverse events were mild or moderate in severity ◦ Uptitrating OCA dose from 5 mg to 10 mg at 6 months mitigated the incidence of pruritus and improved tolerability as assessed by patient discontinuation rate due to pruritus • 1% in titration group • 9% in 10 -mg group OCA, obeticholic acid. Kowdley K, et al. Paper presented at: DDW 2015; May 16 -19, 2015; Washington, DC. Abstract 657.

OCA: Drug Interactions • Bile acid binding resins ◦ Take OCA >4 hours before or after resin • Warfarin ◦ ↓ International normalized ratio • CYP 1 A 2 substrates with narrow therapeutic index ◦ ↓ Clearance of CYP 1 A 2 substrates CYP 1 A 2, cytochrome P 450 1 A 2 enzyme; OCA, obeticholic acid. Ocaliva [package insert]. New York, NY: Intercept Pharmaceuticals, Inc. ; 2016.

Peroxisome Proliferator-Activated Receptor Alpha Activity • Regulates bile acid synthesis and detoxification • Modulates phospholipid secretion, which helps protect bile duct epithelium by formation of micelles Zollner G, et al. Br J Pharmacol. 2009; 156: 7 -27.

Fibroblast Growth Factor 19 Signaling • Fibroblast growth factor 19 (FGF 19) is an endocrine hormone that helps regulate bile acids, and carbohydrate, lipid, and energy metabolism • FGF 19 also has a role in regulating hepatic cell proliferation FGF 19 -FGFR 4 signaling is associated with hepatocellular tumorigenesis • An engineered FGF 19 variant has been shown to be capable of targeting the bile acid homeostasis function, but not the proliferative function Luo J, et al. Sci Transl Med. 2014; 6(247): 247 ra 100.
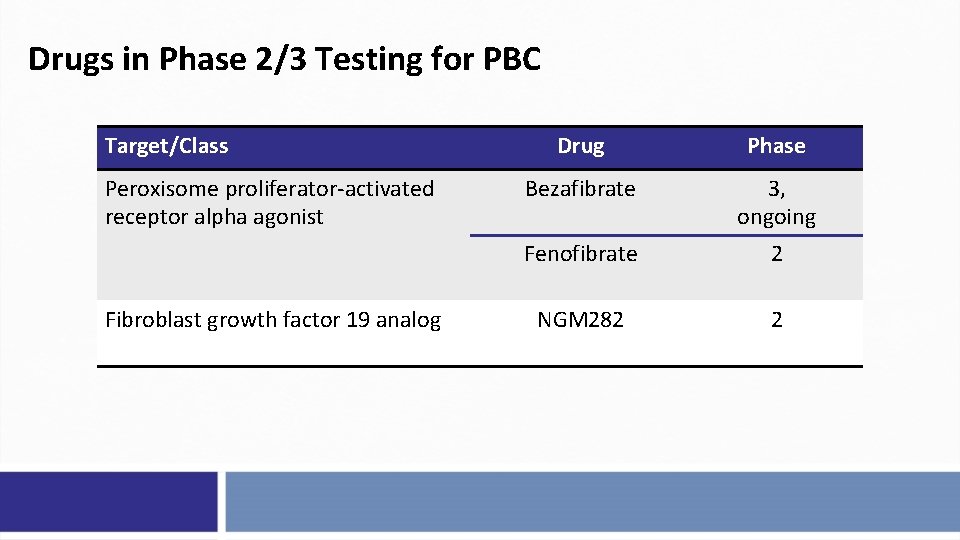
Drugs in Phase 2/3 Testing for PBC Target/Class Peroxisome proliferator-activated receptor alpha agonist Fibroblast growth factor 19 analog Drug Phase Bezafibrate 3, ongoing Fenofibrate 2 NGM 282 2

Bezafibrate + UDCA—Long-term Outcome in UDCA Nonresponders With Dyslipidemia • Prospective, randomized, controlled, multicenter study (N = 27) • Continued administration of UDCA vs bezafibrate add-on to UDCA after ≥ 24 weeks; therapy continued through 8 years • Primary endpoints ◦ ALP level ◦ Mayo risk score ◦ Total bilirubin, AST, albumin • Other endpoints ◦ Overall survival ◦ HCC incidence ◦ Creatinine—safety endpoint ALP, alkaline phosphatase; AST, aspartate aminotransferase; HCC, hepatocellular carcinoma; UDCA, ursodeoxycholic acid or ursodiol. Hosonuma K, et al. Am J Gastroenterol. 2015; 110: 423 -431.

Bezafibrate + UDCA—Key Long-term Outcomes at 8 Years • Mayo risk score (bezafibrate + UDCA vs UDCA) ◦ 0. 91 vs 1. 42 (P <. 05) • Mortality rate and incidence of HCC were not significantly different between the 2 groups • Creatinine levels at 8 years (bezafibrate + UDCA vs UDCA) ◦ 0. 94 vs 0. 56 mg/d. L (P <. 05) • “We should pay close attention to adverse events during this long-term combination therapy” HCC, hepatocellular carcinoma; UDCA, ursodeoxycholic acid or ursodiol. Hosonuma K, et al. Am J Gastroenterol. 2015; 110: 423 -431.
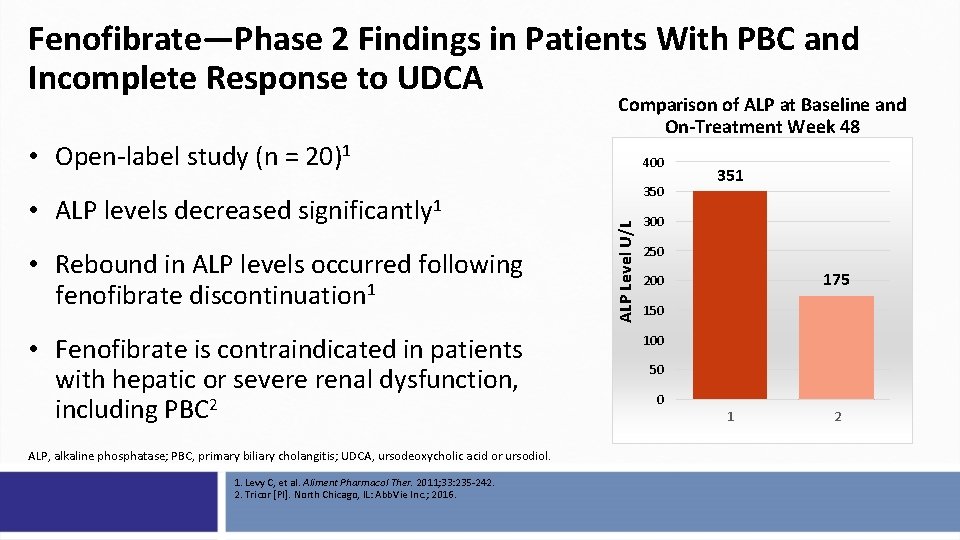
Fenofibrate—Phase 2 Findings in Patients With PBC and Incomplete Response to UDCA Comparison of ALP at Baseline and On-Treatment Week 48 • Open-label study (n = 20)1 • Rebound in ALP levels occurred following fenofibrate discontinuation 1 • Fenofibrate is contraindicated in patients with hepatic or severe renal dysfunction, including PBC 2 ALP, alkaline phosphatase; PBC, primary biliary cholangitis; UDCA, ursodeoxycholic acid or ursodiol. 1. Levy C, et al. Aliment Pharmacol Ther. 2011; 33: 235 -242. 2. Tricor [PI]. North Chicago, IL: Abb. Vie Inc. ; 2016. 350 ALP Level U/L • ALP levels decreased significantly 1 400 351 300 250 175 200 150 100 50 0 1 2
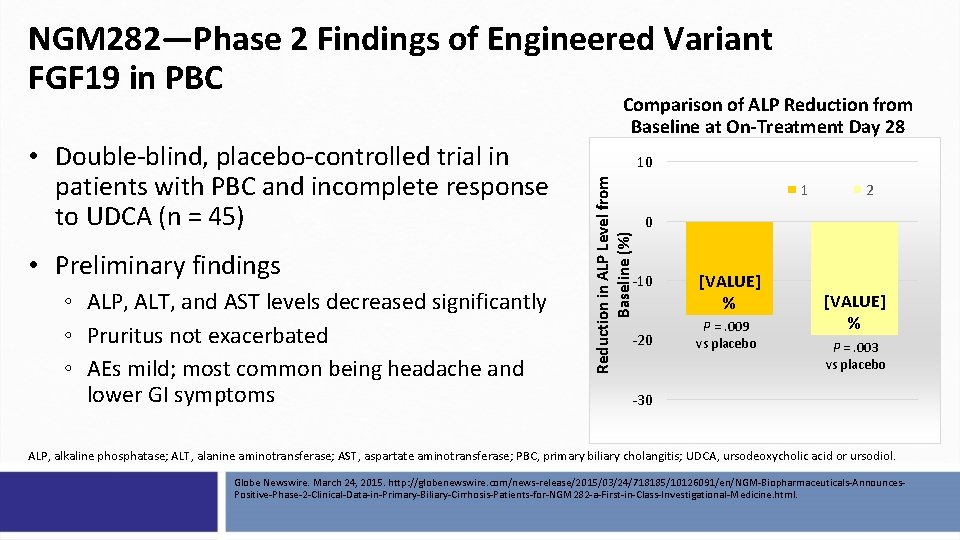
NGM 282—Phase 2 Findings of Engineered Variant FGF 19 in PBC • Preliminary findings ◦ ALP, ALT, and AST levels decreased significantly ◦ Pruritus not exacerbated ◦ AEs mild; most common being headache and lower GI symptoms 10 Reduction in ALP Level from Baseline (%) • Double-blind, placebo-controlled trial in patients with PBC and incomplete response to UDCA (n = 45) Comparison of ALP Reduction from Baseline at On-Treatment Day 28 1 2 0 -10 -20 [VALUE] % P =. 009 vs placebo [VALUE] % P =. 003 vs placebo -30 ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; PBC, primary biliary cholangitis; UDCA, ursodeoxycholic acid or ursodiol. Globe Newswire. March 24, 2015. http: //globenewswire. com/news-release/2015/03/24/718185/10126091/en/NGM-Biopharmaceuticals-Announces. Positive-Phase-2 -Clinical-Data-in-Primary-Biliary-Cirrhosis-Patients-for-NGM 282 -a-First-in-Class-Investigational-Medicine. html.

Fatigue in Patients With PBC • Common at all stages of PBC • Autonomic dysfunction • Accelerated reduction in muscle function Mitochondrial dysfunction? PBC, primary biliary cholangitis. Griffiths L, et al. Dig Dis. 2014; 32: 615 -625.
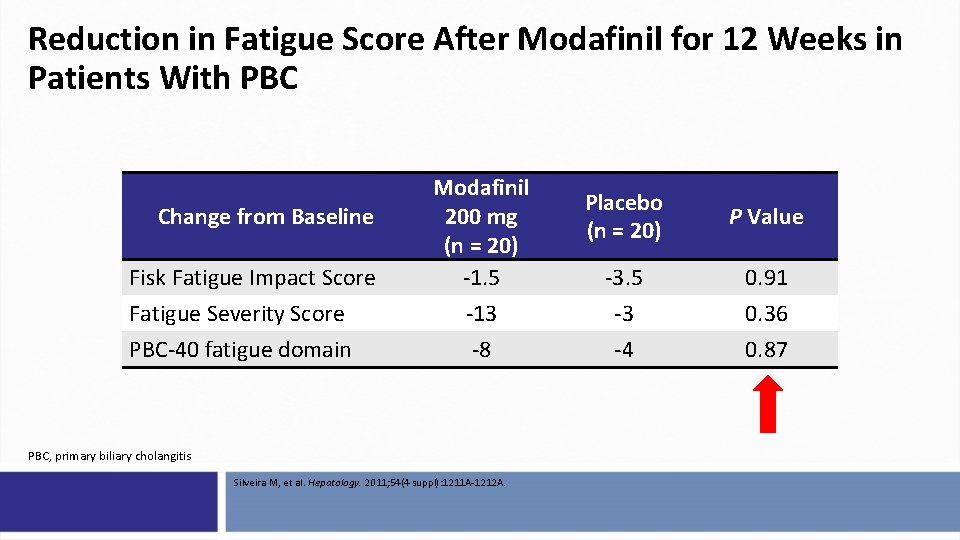
Reduction in Fatigue Score After Modafinil for 12 Weeks in Patients With PBC Change from Baseline Fisk Fatigue Impact Score Fatigue Severity Score PBC-40 fatigue domain Modafinil 200 mg (n = 20) -1. 5 -13 -8 PBC, primary biliary cholangitis Silveira M, et al. Hepatology. 2011; 54(4 suppl): 1211 A-1212 A. Placebo (n = 20) P Value -3. 5 -3 -4 0. 91 0. 36 0. 87

Pruritus in Patients With PBC • Occurs early in cholestatic diseases, 1 later in hepatocellular diseases • Localized vs generalized ◦ Palms and soles • No primary rash 1 • Exacerbated by ◦ Pressure ◦ Heat 1 • Circadian rhythm (worse in evenings)1, 2 • Periodic exacerbations and improvements 2 PBC, primary biliary cholangitis 1. Rishe E, et al. Acta Derm Venereol. 2008; 88: 34 -37. 2. Mayo MJ, et al. Primary biliary cirrhosis. In: Yamada T, ed. Textbook of Gastroenterology, 4 th ed. Oxford, UK: Lippincott Williams & Wilkins; 2003.

Behavioral Modifications for Pruritus in Patients With PBC • Wear loose, absorbent clothes • Seek cool (not dry) environment • Use cool emollients frequently • Avoid pruritogenic medications (opioids) • Trim nails • Limit sun/ultraviolet exposure PBC, primary biliary cholangitis.
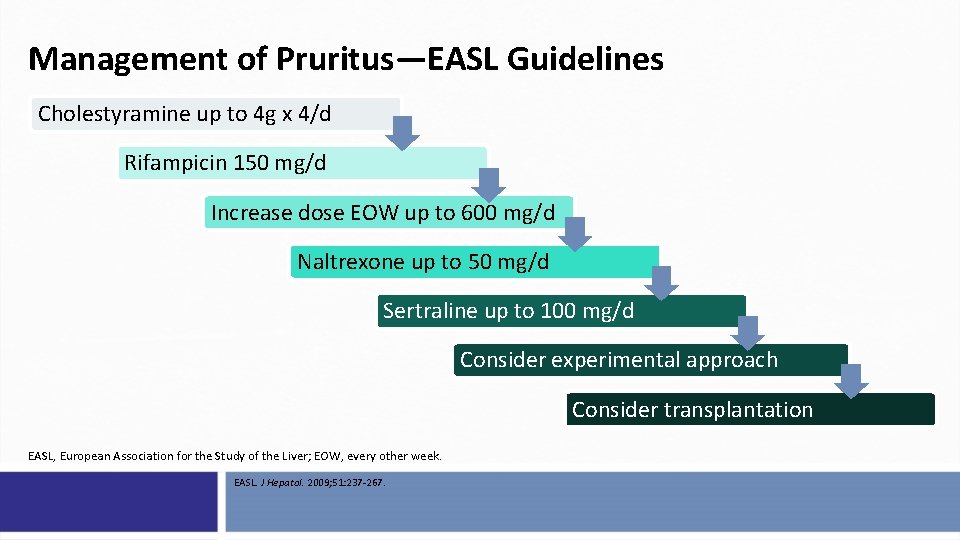
Management of Pruritus—EASL Guidelines Cholestyramine up to 4 g x 4/d Rifampicin 150 mg/d Increase dose EOW up to 600 mg/d Naltrexone up to 50 mg/d Sertraline up to 100 mg/d Consider experimental approach Consider transplantation EASL, European Association for the Study of the Liver; EOW, every other week. EASL. J Hepatol. 2009; 51: 237 -267.

Sicca Syndrome Management—Dry Mouth • Professional dental cleaning every 6 months • Sugar-free candy/gum • Rinsing with water • Saliva substitutes • Pilocarpine or cevimeline if refractory to all of the above Lindor KD, et al. Hepatology. 2009; 50: 291 -308. Graphic courtesy of National Institutes of Health.
Sicca Syndrome Management—Dry Eyes • Artificial tears • Pilocarpine or cevimeline if refractory to artificial tears • Cyclosporine eye drops if refractory to all of the above • Tear duct plugs Lindor KD, et al. Hepatology. 2009; 50: 291 -308. Graphic courtesy of National Institutes of Health.
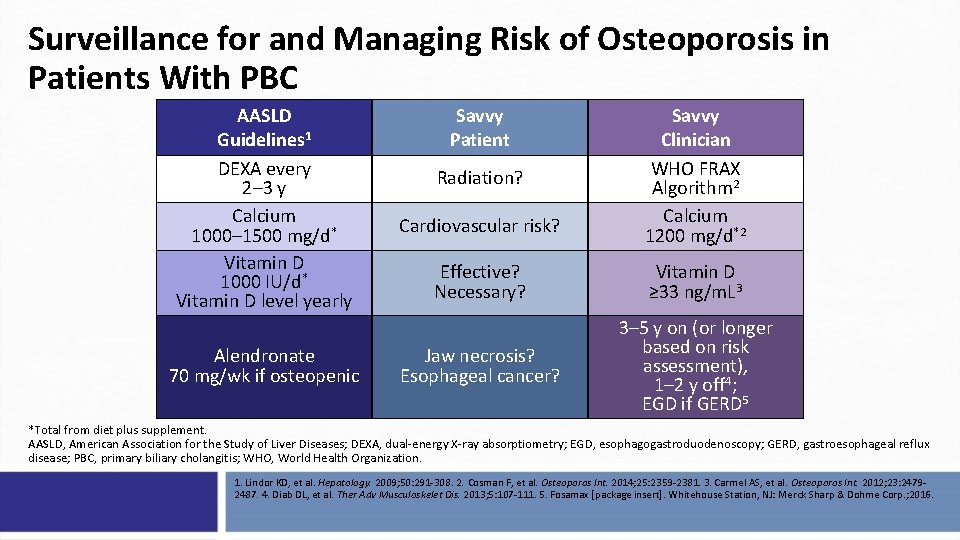
Surveillance for and Managing Risk of Osteoporosis in Patients With PBC AASLD Guidelines 1 DEXA every 2– 3 y Calcium 1000– 1500 mg/d* Vitamin D 1000 IU/d* Vitamin D level yearly Alendronate 70 mg/wk if osteopenic Savvy Patient Cardiovascular risk? Savvy Clinician WHO FRAX Algorithm 2 Calcium 1200 mg/d*2 Effective? Necessary? Vitamin D ≥ 33 ng/m. L 3 Jaw necrosis? Esophageal cancer? 3– 5 y on (or longer based on risk assessment), 1– 2 y off 4; EGD if GERD 5 Radiation? *Total from diet plus supplement. AASLD, American Association for the Study of Liver Diseases; DEXA, dual-energy X-ray absorptiometry; EGD, esophagogastroduodenoscopy; GERD, gastroesophageal reflux disease; PBC, primary biliary cholangitis; WHO, World Health Organization. 1. Lindor KD, et al. Hepatology. 2009; 50: 291 -308. 2. Cosman F, et al. Osteoporos Int. 2014; 25: 2359 -2381. 3. Carmel AS, et al. Osteoporos Int. 2012; 23: 24792487. 4. Diab DL, et al. Ther Adv Musculoskelet Dis. 2013; 5: 107 -111. 5. Fosamax [package insert]. Whitehouse Station, NJ: Merck Sharp & Dohme Corp. ; 2016.
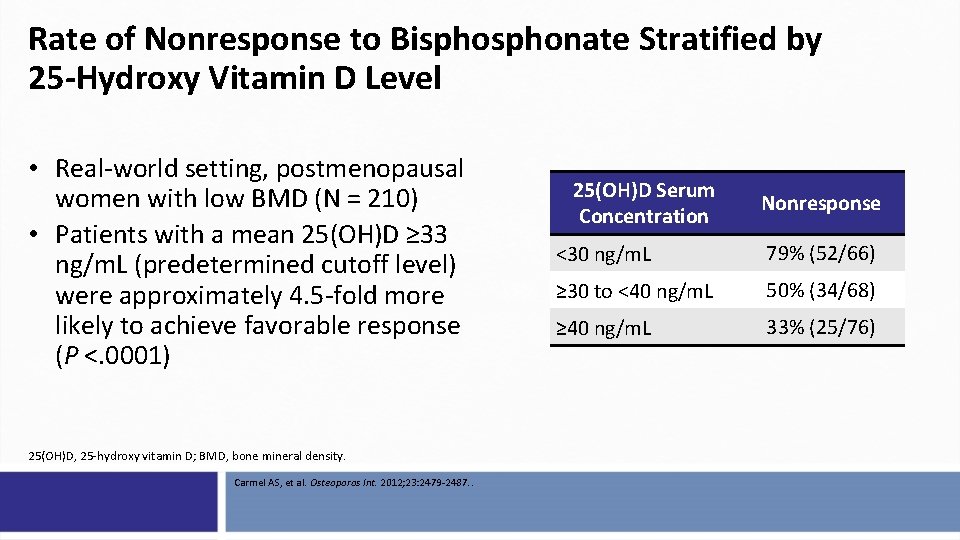
Rate of Nonresponse to Bisphonate Stratified by 25 -Hydroxy Vitamin D Level • Real-world setting, postmenopausal women with low BMD (N = 210) • Patients with a mean 25(OH)D ≥ 33 ng/m. L (predetermined cutoff level) were approximately 4. 5 -fold more likely to achieve favorable response (P <. 0001) 25(OH)D, 25 -hydroxy vitamin D; BMD, bone mineral density. Carmel AS, et al. Osteoporos Int. 2012; 23: 2479 -2487. . 25(OH)D Serum Concentration Nonresponse <30 ng/m. L 79% (52/66) ≥ 30 to <40 ng/m. L 50% (34/68) ≥ 40 ng/m. L 33% (25/76)

Surveillance for Fat-Soluble Vitamin Deficiencies in Patients With PBC • Decreased bile acid secretion may lead to impaired absorption of fat-soluble vitamins ADEK 1 • Monitoring guidelines ◦ AASLD: no specific recommendation 2 ◦ Medicare: no more than once annually 3 • Reasonable practice: annual vitamin A, D, PT (surrogate marker for K) • May need to increase frequency of surveillance with new bile acid pool-lowering therapies Vitamin Proportion of PBC Patients with Deficiency 1 (N = 180) A D E K 33. 5% 13. 2% 1. 9% 7. 8% AASLD, American Association for the Study of Liver Diseases; ADEK, vitamins A, D, E, and K; PBC, primary biliary cholangitis; PT, prothrombin time. 1. Phillips JR, et al. Am J Gastroenterol. 2001; 96: 2745 -2750. 2. Lindor KD, et al. Hepatology. 2009; 50: 291 -308. 3. https: //www. cms. gov/medicare-coverage-database/details/lcddetails. aspx? LCDId=29510&Contr. Id=269&ver=38&Contr. Ver=1&Date=08%2 f 04%2 f 2014&Doc. ID=L 29510&bc=AAAAAAg. AAAAAAA%3 d%3 d&

Surveillance for Hepatocellular Carcinoma in Patients With PBC • American Association for the Study of Liver Disease Patients with cirrhosis should be screened every 6− 12 months using ultrasound with (or without) alpha fetoprotein 1, 2 • Beware the older male nonresponder, even if not cirrhotic!3 1. Lindor KD, et al. Hepatology. 2009; 50: 291 -308. 2. Bruix J, et al. Hepatology. 2011; 53(3): 1020 -1022. 3. Trivedi PJ, et al. Gut. 2016; 65(2): 321 -329.
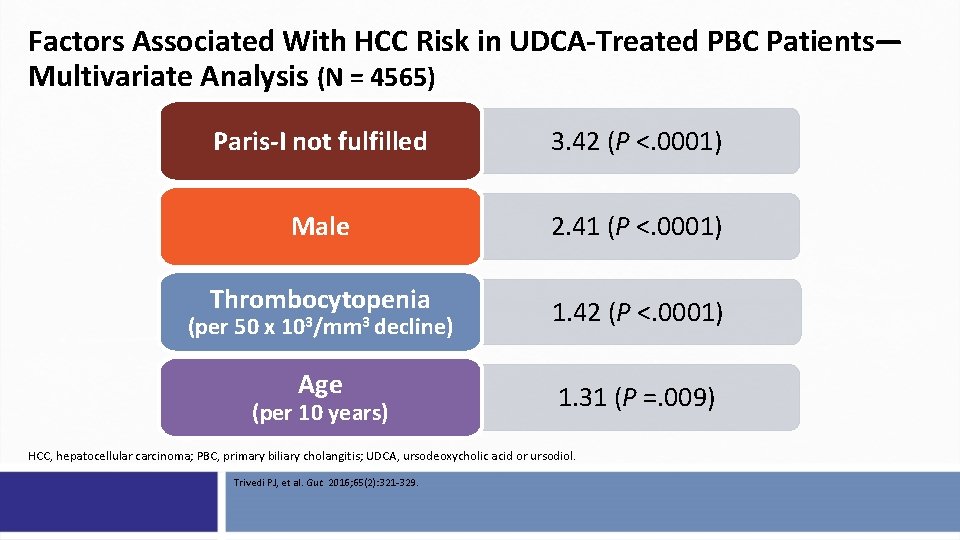
Factors Associated With HCC Risk in UDCA-Treated PBC Patients— Multivariate Analysis (N = 4565) Paris-I not fulfilled 3. 42 (P <. 0001) Male 2. 41 (P <. 0001) Thrombocytopenia 1. 42 (P <. 0001) Age 1. 31 (P =. 009) (per 50 x 103/mm 3 decline) (per 10 years) HCC, hepatocellular carcinoma; PBC, primary biliary cholangitis; UDCA, ursodeoxycholic acid or ursodiol. Trivedi PJ, et al. Gut. 2016; 65(2): 321 -329.

Surveillance for Varices • Patients with cirrhosis should be screened every 1− 3 years using esophagogastroduodenoscopy 1 Interval based on decompensation 2 • Varices with bleeding may occur in noncirrhotic PBC patients 3 1. Lindor KD, et al. Hepatology. 2009; 50: 291 -308. 2. Garcia-Tsao G, et al. Hepatology. 2007; 46: 922 -938. 3. Vlachogiannakos J, et al. Eur J Gastroenterol Hepatol. 2009; 21: 701 -707.
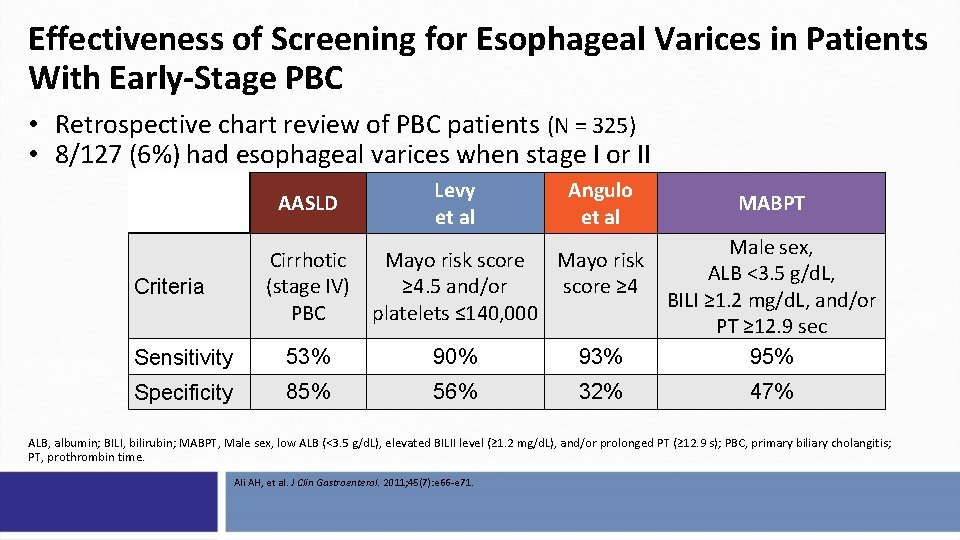
Effectiveness of Screening for Esophageal Varices in Patients With Early-Stage PBC • Retrospective chart review of PBC patients (N = 325) • 8/127 (6%) had esophageal varices when stage I or II AASLD Levy et al Angulo et al MABPT Sensitivity 53% 90% 93% Male sex, ALB <3. 5 g/d. L, BILI ≥ 1. 2 mg/d. L, and/or PT ≥ 12. 9 sec 95% Specificity 85% 56% 32% 47% Criteria Cirrhotic (stage IV) PBC Mayo risk score Mayo risk ≥ 4. 5 and/or score ≥ 4 platelets ≤ 140, 000 ALB, albumin; BILI, bilirubin; MABPT, Male sex, low ALB (<3. 5 g/d. L), elevated BILII level (≥ 1. 2 mg/d. L), and/or prolonged PT (≥ 12. 9 s); PBC, primary biliary cholangitis; PT, prothrombin time. Ali AH, et al. J Clin Gastroenterol. 2011; 45(7): e 66 -e 71.
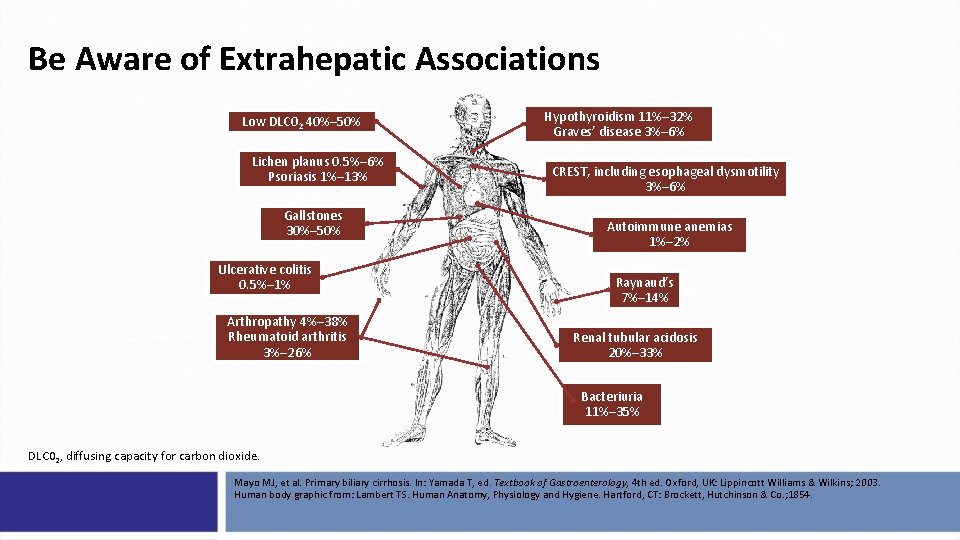
Be Aware of Extrahepatic Associations Low DLC 02 40%– 50% Lichen planus 0. 5%– 6% Psoriasis 1%– 13% Gallstones 30%– 50% Ulcerative colitis 0. 5%– 1% Arthropathy 4%– 38% Rheumatoid arthritis 3%– 26% Hypothyroidism 11%– 32% Graves’ disease 3%– 6% CREST, including esophageal dysmotility 3%– 6% Autoimmune anemias 1%– 2% Raynaud’s 7%– 14% Renal tubular acidosis 20%– 33% Bacteriuria 11%– 35% DLC 02, diffusing capacity for carbon dioxide. Mayo MJ, et al. Primary biliary cirrhosis. In: Yamada T, ed. Textbook of Gastroenterology, 4 th ed. Oxford, UK: Lippincott Williams & Wilkins; 2003. Human body graphic from: Lambert TS. Human Anatomy, Physiology and Hygiene. Hartford, CT: Brockett, Hutchinson & Co. ; 1854.
- Slides: 62