Presentation by Liquid Chromatography Mass Spectrometry Pari Karami

Presentation by: Liquid Chromatography – Mass Spectrometry Pari Karami www. darsonline. org

Outline �Introduction � Instrumentation �Application

Introduction ØWhy Liquid Chromatography? ØAnalysis of nonvolatile and thermally fragile analytes ØAnalysis of more polar compounds without derivatization. ØAnalysis of significantly higher masses v. Why Mass Spectrometry ? v. It measures the masses of individual molecules v. The mass spectrum of each compound is unique and can be used as chemical “fingerprint”

q. LC with detectors like Refractive index, electrochemical, fluorescence, and ultravioletvisible (UV-Vis) detectors generate two dimensional data; that is, data representing signal strength as a function of time q. When coupled with MS In addition to signal strength, they generate mass spectral data that can provide valuable information about the

LC Problem in combining LC & MS MS ØLiquid phase operation v. Vacuum operation Ø 25 - 50 deg. C v 200 - 300 deg. C ØNo mass range limitations v. Up to 4000 Da for quadrup ØInorganic buffers v. Requires volatile buffers Ø 1 ml/min eluent flow is equivalent to 500 ml/min of v. Accepts 10 ml/min gas flow

Fundamental incompatibility between the Gas-phase world of MS, the Bird, and the Liquid-phase world of LC, the Fish First LC-MS interface developed in 1969 Ray Scott
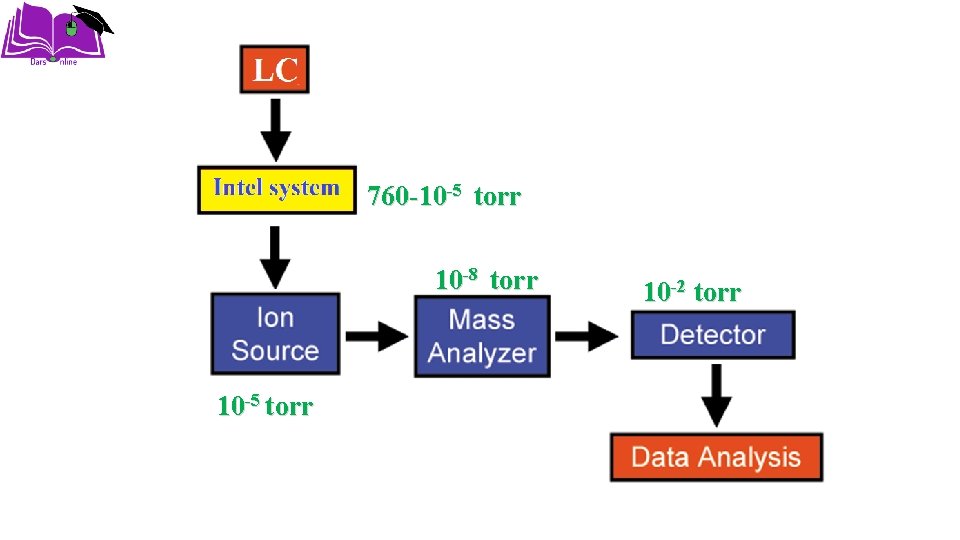
760 -10 -5 torr 10 -8 torr 10 -5 torr 10 -2 torr
Interfacing systems

Interfacing systems Ø Moving Belt Ø Thermospray ionization (TSI) Ø Particle Beam Interface (PB) Ø Atmospheric Pressure ionization (API) • Electrospray ionization (ESI) • Atmospheric pressure chemical ionization (APCI) • Atmospheric pressure photoionization(APPI)
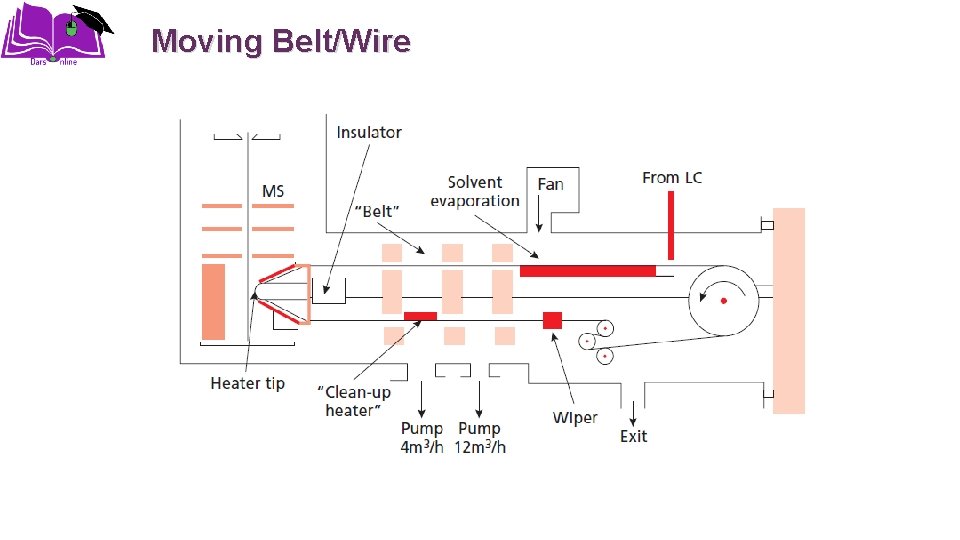
Moving Belt/Wire
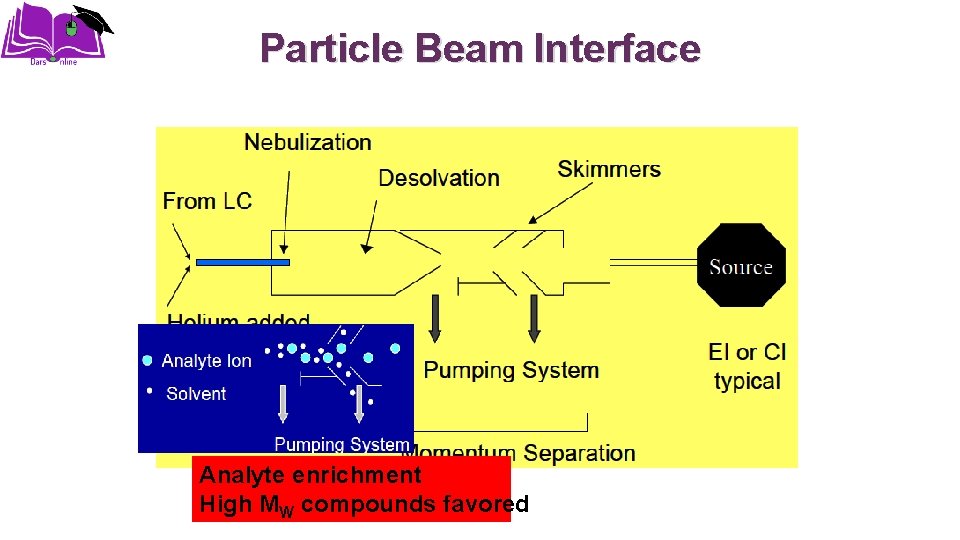
Particle Beam Interface Analyte enrichment High MW compounds favored
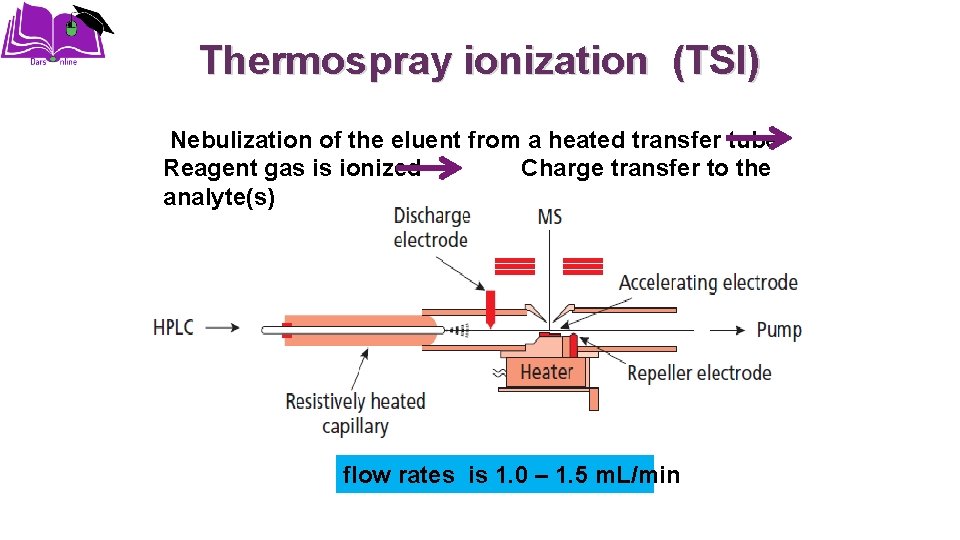
Thermospray ionization (TSI) Nebulization of the eluent from a heated transfer tube Reagent gas is ionized Charge transfer to the analyte(s) flow rates is 1. 0 – 1. 5 m. L/min
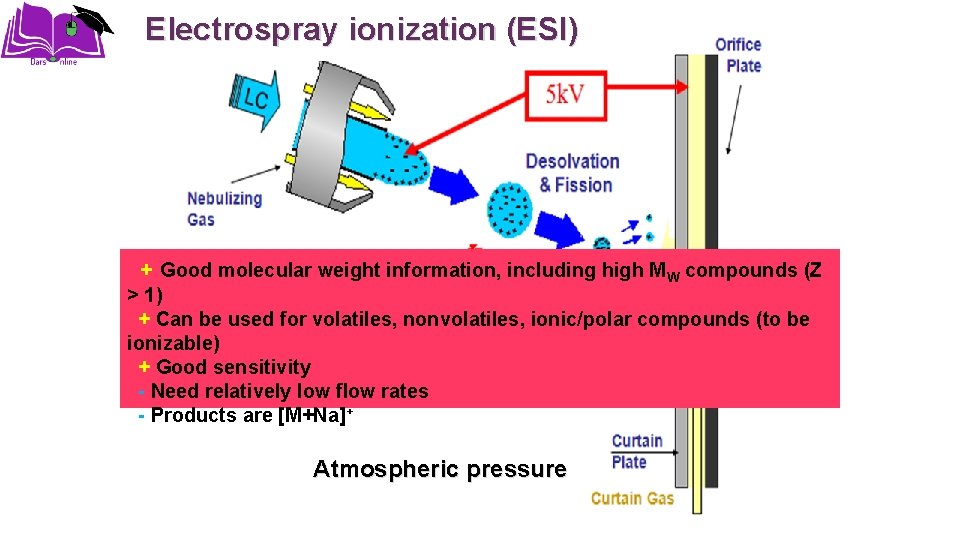
Electrospray ionization (ESI) + Good molecular weight information, including high MW compounds (Z > 1) + Can be used for volatiles, nonvolatiles, ionic/polar compounds (to be ionizable) + Good sensitivity - Need relatively low flow rates - Products are [M+Na]+ Atmospheric pressure
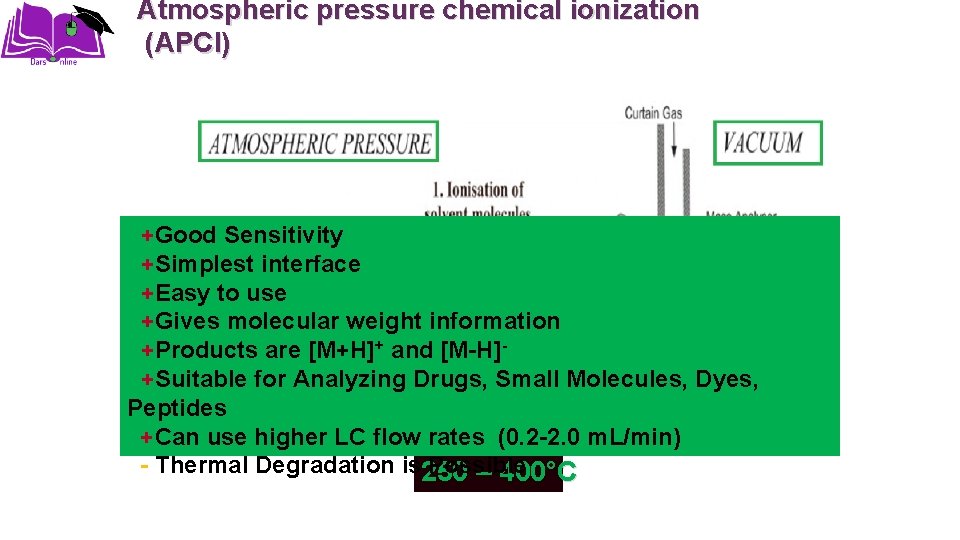
Atmospheric pressure chemical ionization (APCI) +Good Sensitivity +Simplest interface +Easy to use +Gives molecular weight information +Products are [M+H]+ and [M-H]+Suitable for Analyzing Drugs, Small Molecules, Dyes, Peptides +Can use higher LC flow rates (0. 2 -2. 0 m. L/min) - Thermal Degradation is 250 Possible – 400°C

Atmospheric pressure photoionization (APPI) +For highly non polar compounds +Low flow rates (<100 μl/min)
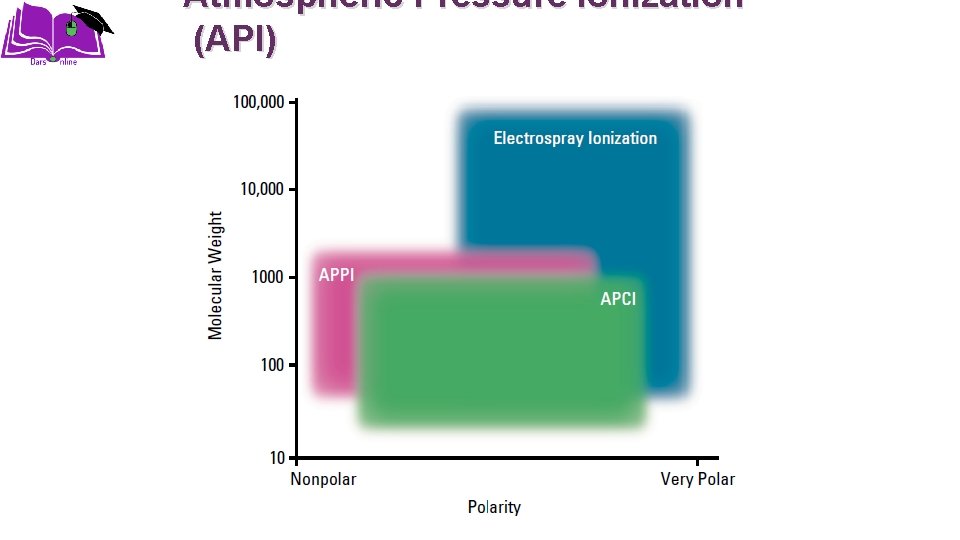
Atmospheric Pressure ionization (API)
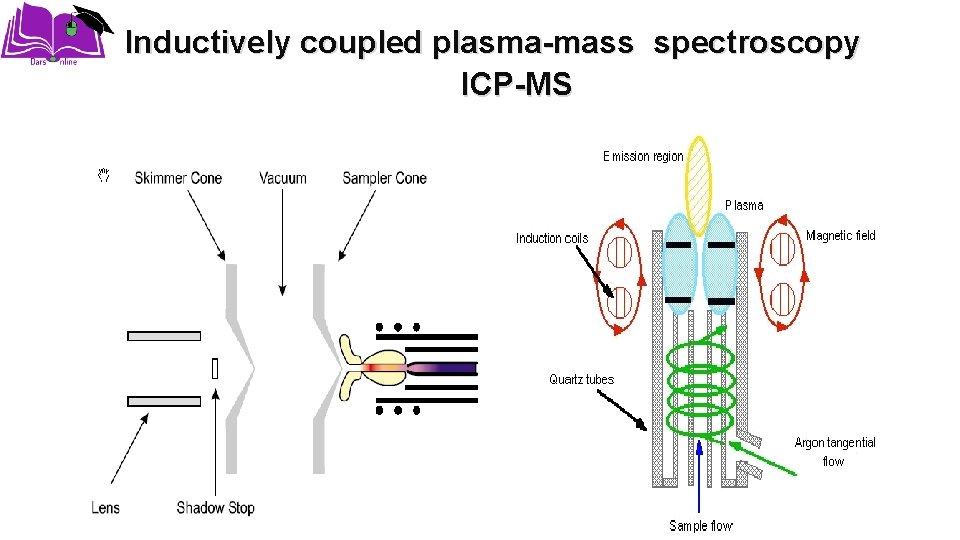
Inductively coupled plasma-mass spectroscopy ICP-MS
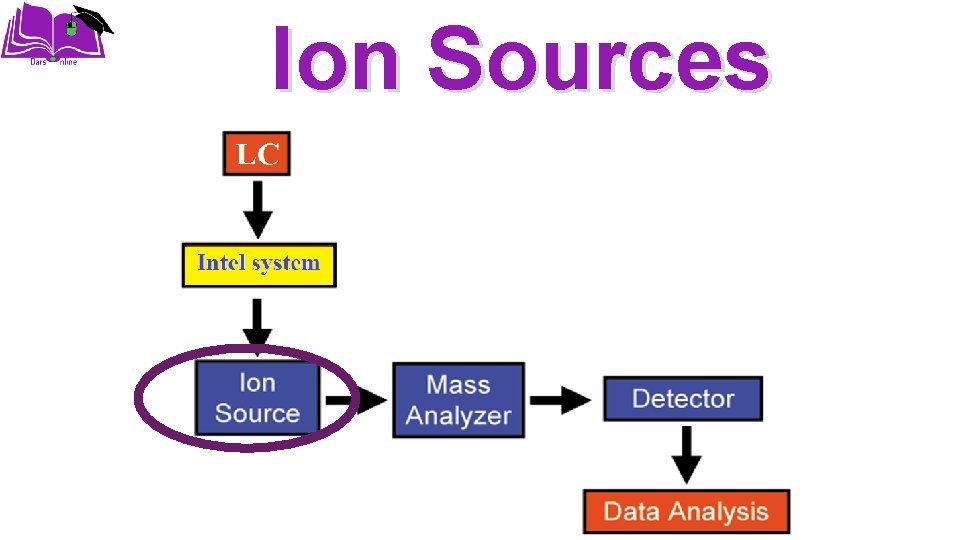
Ion Sources
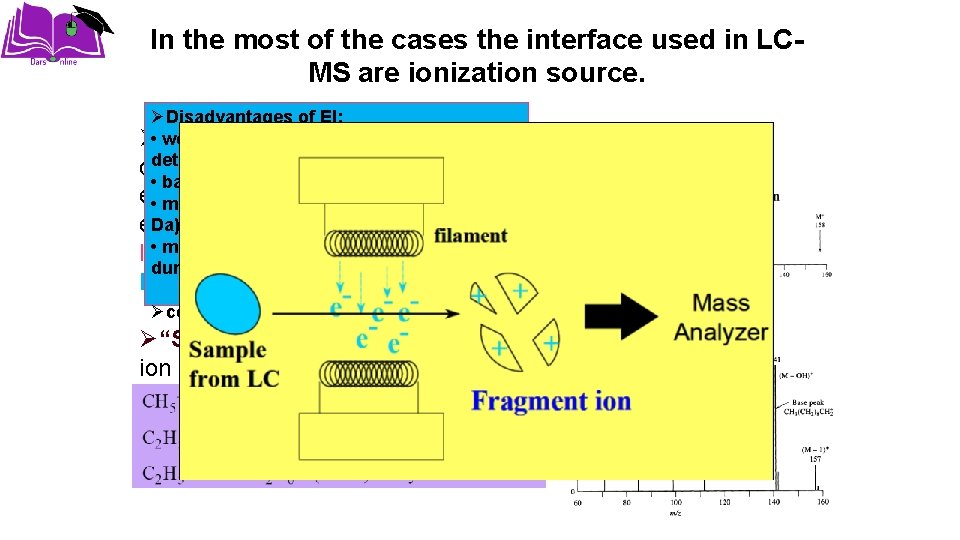
In the most of the cases the interface used in LCMS are ionization source. ØDisadvantages of EI: +peak inhibits or absent Ø • weak “Hard” -(EI)-M(PB, MB) determination of MW considerable fragmentation, leave • base peak m/z « M+ excess energy –W < 103 • molecules must in be molecule vaporized (M Da) extensive fragmentation. Standard • molecules be thermally stable libraries aremust available during vaporization Poor Detection Limits Øcomplex spectra helps identification Ø“Soft” -(CI)- (API, TSI), molecular ion is main product little excess energy in of molecule – reduced EI ionization reactant gas (methane) • most common ions (M+1)+ and (M-1)+ fragmentation • sometimes (M+17)+ (addition of CH 5+ or OH) or (M+29)+ (addition of C 2 H 5+)
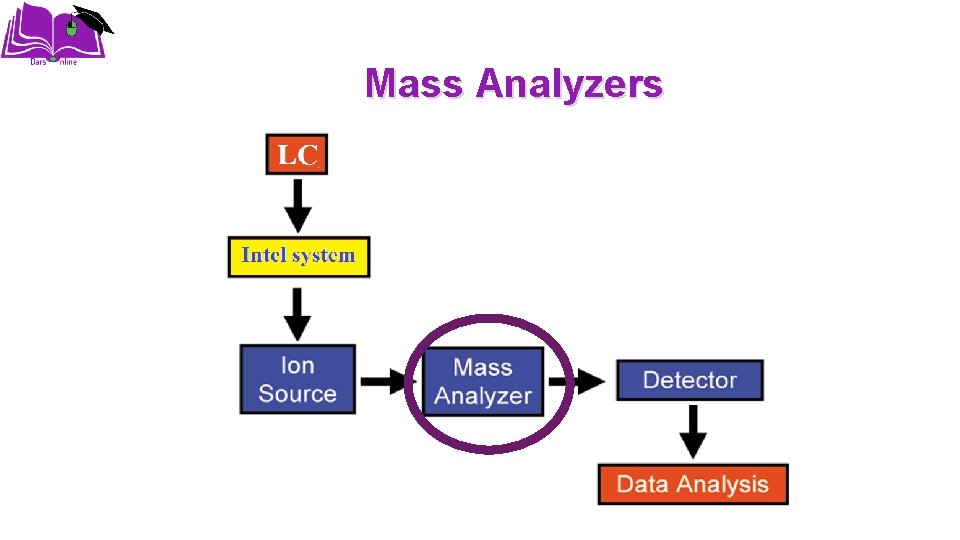
Mass Analyzers
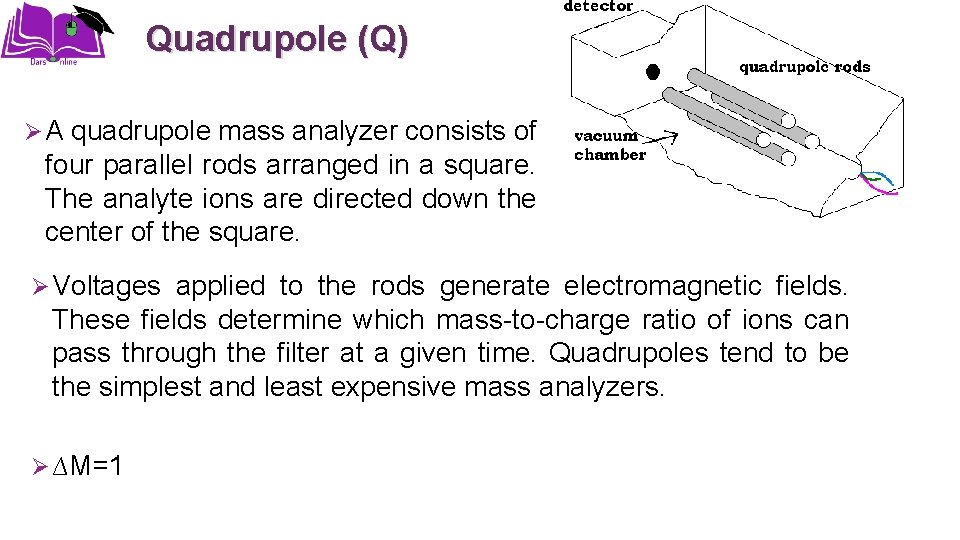
Quadrupole (Q) Ø A quadrupole mass analyzer consists of four parallel rods arranged in a square. The analyte ions are directed down the center of the square. Ø Voltages applied to the rods generate electromagnetic fields. These fields determine which mass-to-charge ratio of ions can pass through the filter at a given time. Quadrupoles tend to be the simplest and least expensive mass analyzers. Ø ∆M=1
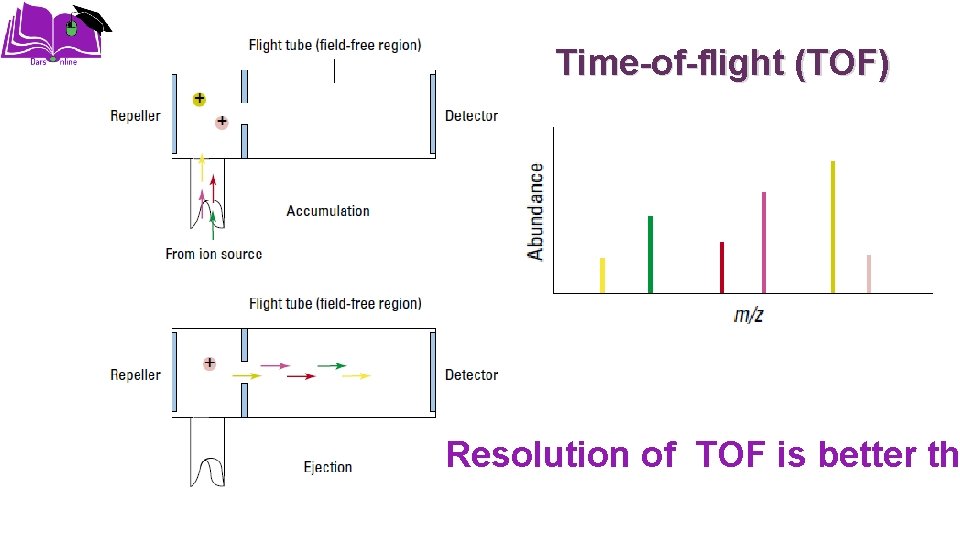
Time-of-flight (TOF) Resolution of TOF is better th
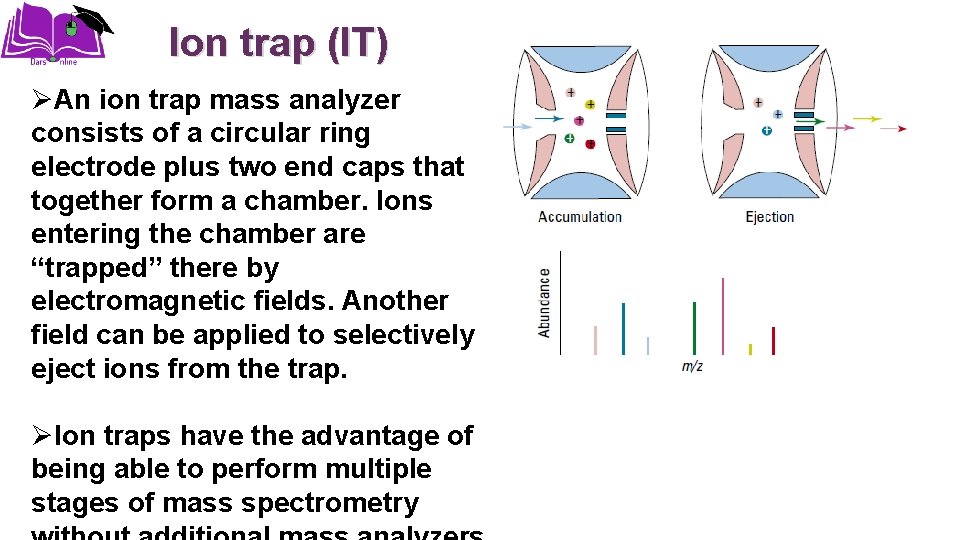
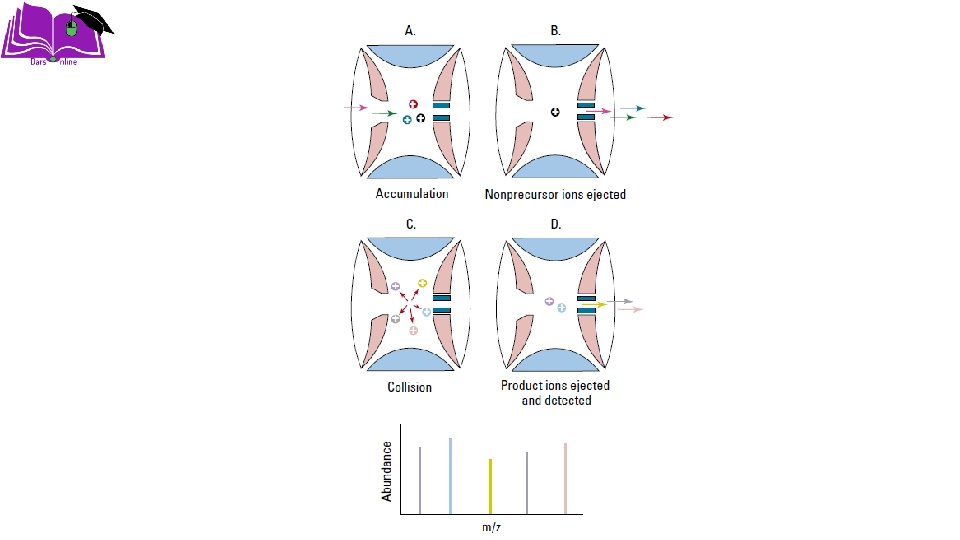
Ion trap (IT) ØAn ion trap mass analyzer consists of a circular ring electrode plus two end caps that together form a chamber. Ions entering the chamber are “trapped” there by electromagnetic fields. Another field can be applied to selectively eject ions from the trap. ØIon traps have the advantage of being able to perform multiple stages of mass spectrometry
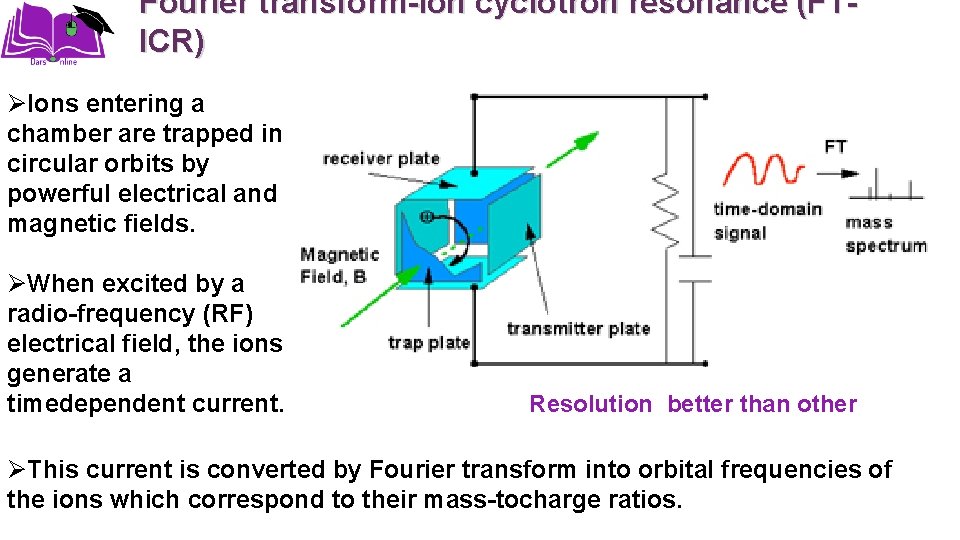
Fourier transform-ion cyclotron resonance (FTICR) ØIons entering a chamber are trapped in circular orbits by powerful electrical and magnetic fields. ØWhen excited by a radio-frequency (RF) electrical field, the ions generate a timedependent current. Resolution better than other ØThis current is converted by Fourier transform into orbital frequencies of the ions which correspond to their mass-tocharge ratios.
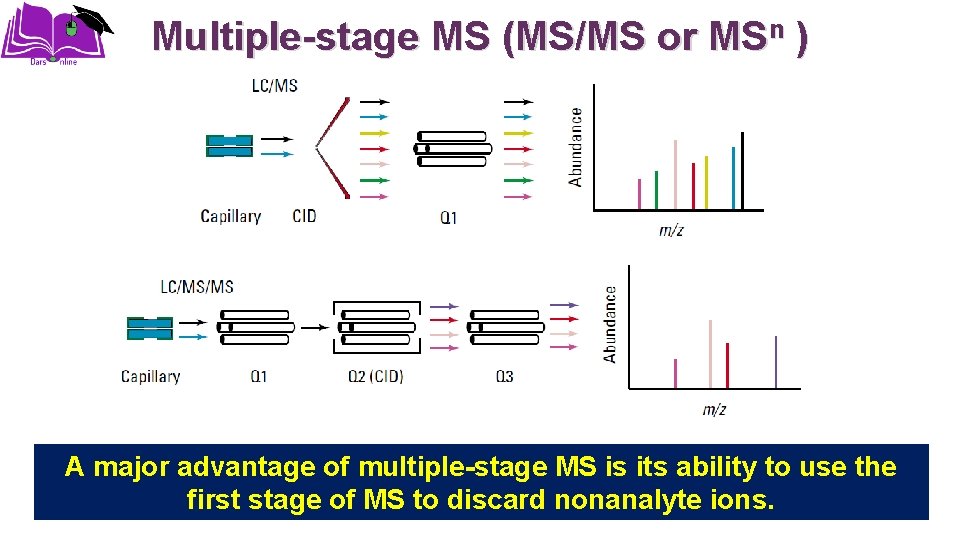
Multiple-stage MS (MS/MS or MSn ) A major advantage of multiple-stage MS is its ability to use the first stage of MS to discard nonanalyte ions.
Detector

Detection Electron multiplier Dynolyte Photomultiplier Microchannel plates Photographic plate Faraday cage Charge collectors
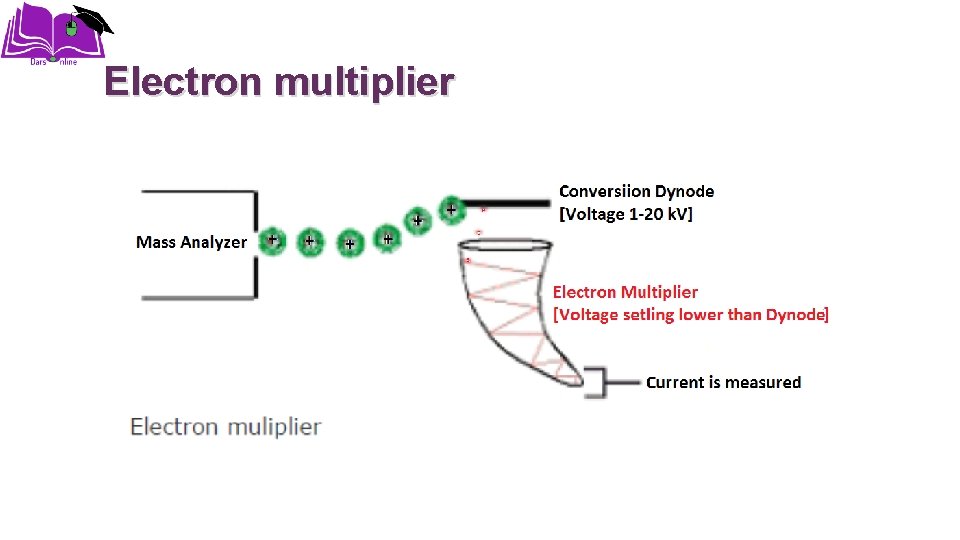
Electron multiplier

Dynolyte photomultiplier

Micro channel plate

Sample preparation
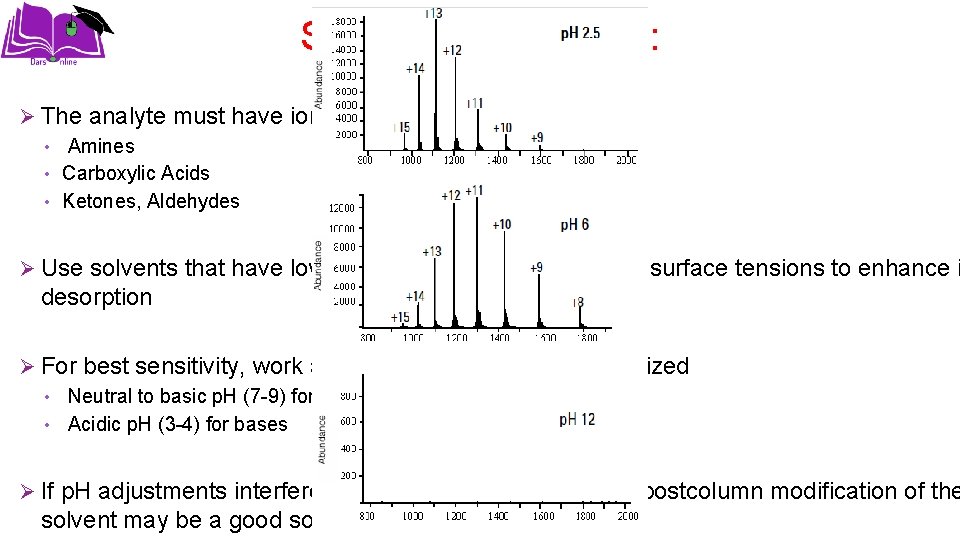
Sample preparation: Ø The analyte must have ionizable groups • Amines • Carboxylic Acids • Ketones, Aldehydes Ø Use solvents that have low heats of vaporization and low surface tensions to enhance i desorption Ø For best sensitivity, work at a p. H where the analyte is ionized • Neutral to basic p. H (7 -9) for acids • Acidic p. H (3 -4) for bases Ø If p. H adjustments interfere with proper chromatography, postcolumn modification of the solvent may be a good solution.

• Ensure adequate analyte concentration • Removing compounds that can cause background ion or suppress ionization. • Make sure that gas-phase reactions do not neutralize ions through proton transfer or ion pair reactions Ø Example of sample preparation include (1) On –column concentration to increase analyte concentration. (2) Filtration to separate a low molecular-weight (3) Desalting to reduce the sodium and potassium adduct
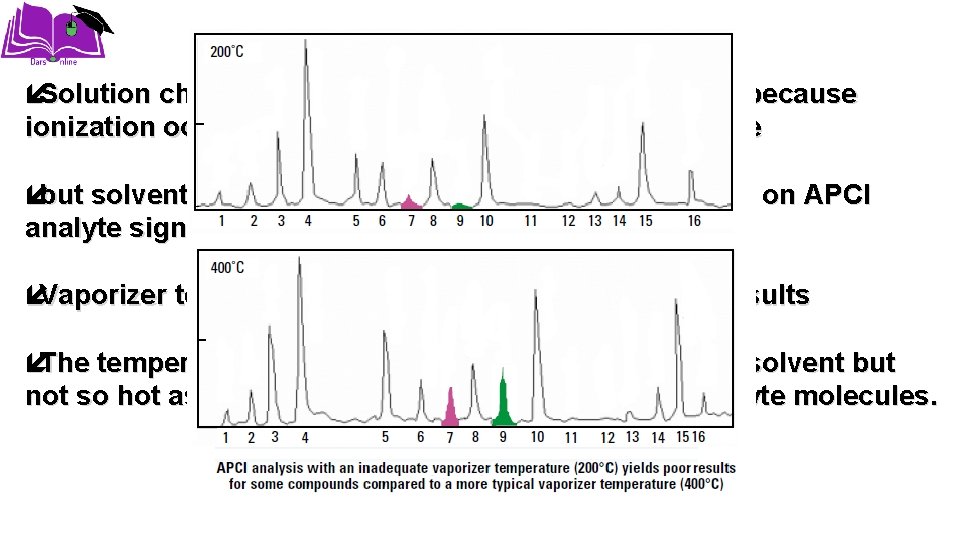
Solution chemistry is less critical for APCI operation because ionization occurs in the gas phase, not the liquid phase but solvent selection can still have a significant effect on APCI analyte signal response. Vaporizer temperature also affects APCI ionization results The temperature must be hot enough to vaporize the solvent but not so hot as to cause thermal degradation of the analyte molecules.
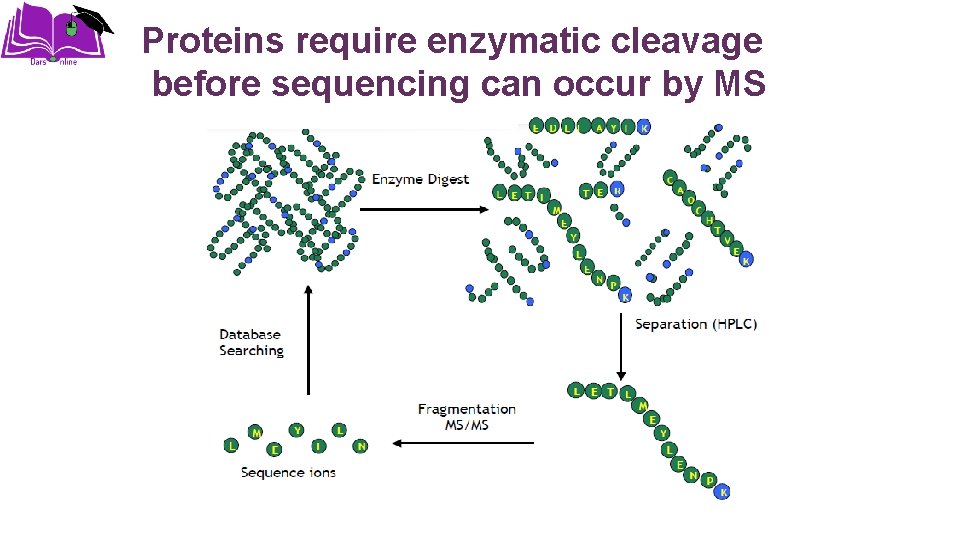
Proteins require enzymatic cleavage before sequencing can occur by MS

Application
Applications �Structural Determination �Molecular Weight Determination �Pharmaceutical Applications �Biochemical Applications �Clinical Applications �Food Applications �Environmental Applications
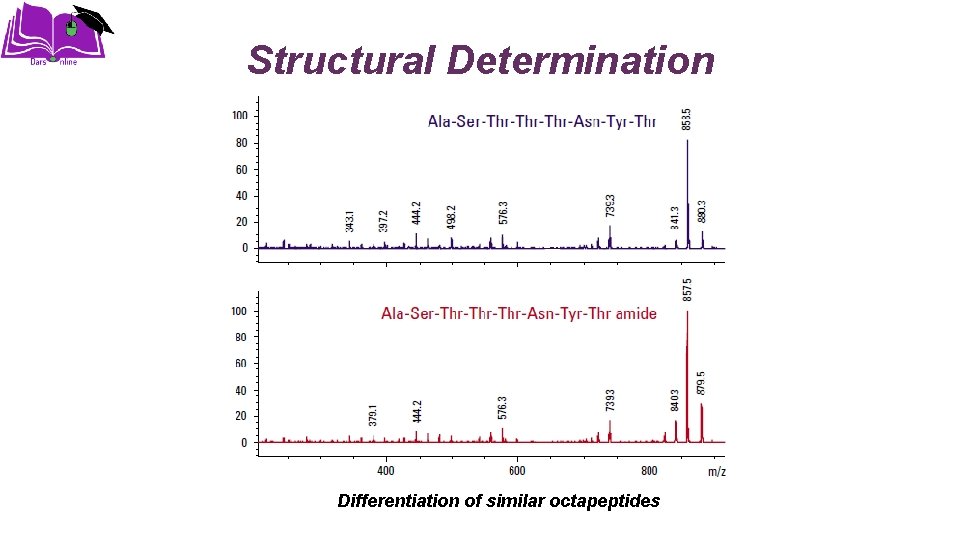
Structural Determination Differentiation of similar octapeptides
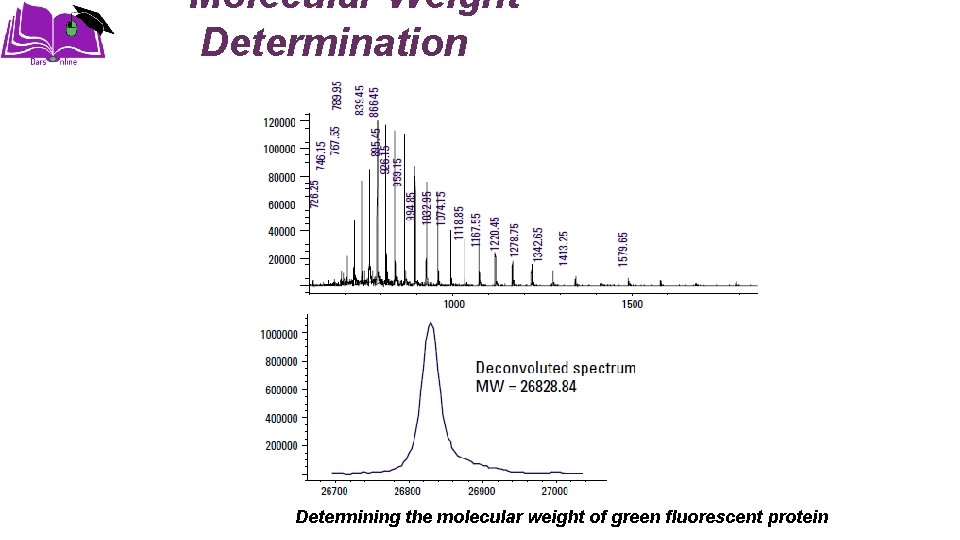
Molecular Weight Determination Determining the molecular weight of green fluorescent protein
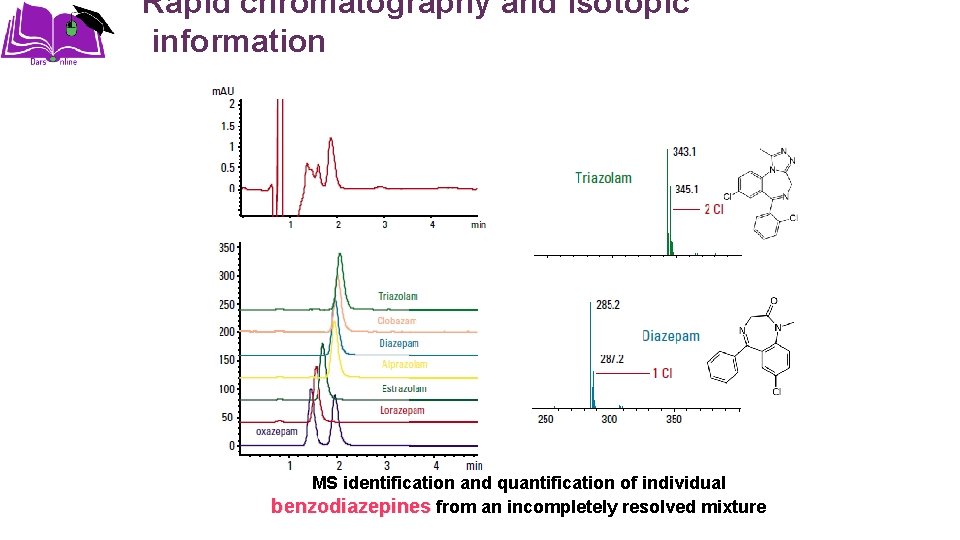
Rapid chromatography and isotopic information MS identification and quantification of individual benzodiazepines from an incompletely resolved mixture
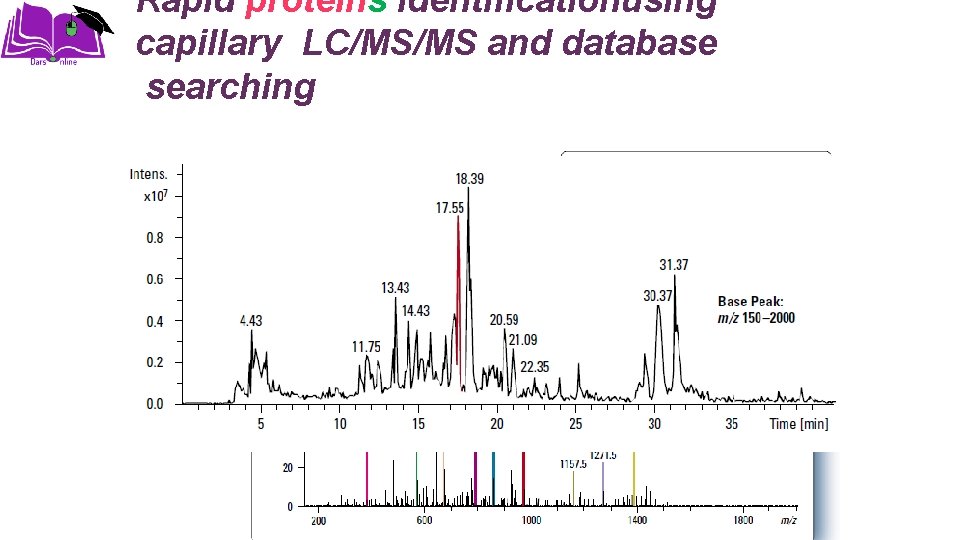
Rapid proteins identificationusing capillary LC/MS/MS and database searching Base peak chromatogram
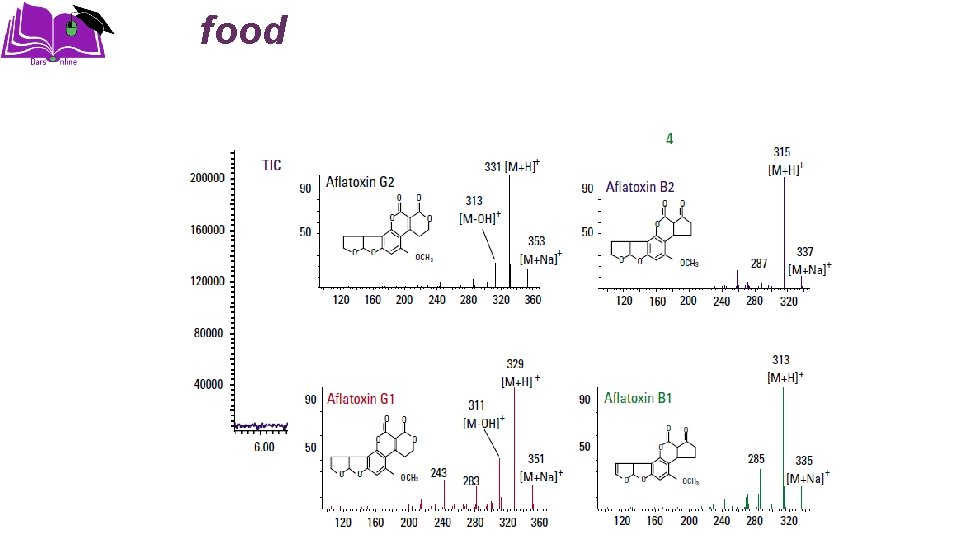
food
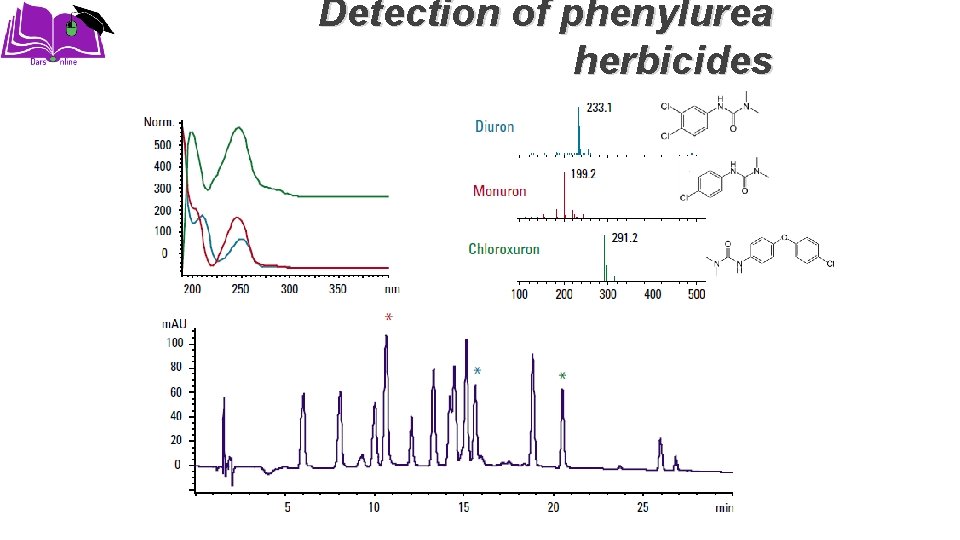
Detection of phenylurea herbicides

Conclusion �LC–MS makes qualitative and quantitative determination of compounds or their metabolites possible even at very low concentration.


The slide presentation available for download at (WWW. darsonline. org)
- Slides: 47