PRESENTATION BY DR NAVEED DEPARTMENT OF FORENSIC MEDICINE


PRESENTATION BY DR. NAVEED DEPARTMENT OF FORENSIC MEDICINE & TOXICOLOGY

USE OF ANTI DOTES IN FORENSIC MEDICINE & TOXICOLOGY Ø Ø a) b) c) Antidotes are remedies which counteract or neutralize the effect of poisons with out causing appreciable harm to the body. They need to be used because The poison may not have been completely remove by emesis or gastric lavage or these procedures are contraindicated The poison is already absorbed The poison has been given by route other then ingestion

� They 1. 2. 3. 4. can be classified into four groups in accordance with their mode of action Mechanical or physical Chemical Physiological or pharmacological Universal

Mechanical or Physical Antidotes � � These are substances which impede the absorption by their presence for example demulcents, bulky food and activated charcoal Demulcents are substances such as fats, oils, milk etc which prevent the absorption of the poison by forming a coating on mucous membrane of the stomach. Activated charcoal is specially useful in adsorbing alkaloidal poisons such as strychnine. It is given in a dose of 30 -60 gms in children and 60 -100 gms in adults in five times the quantity of water

Chemical Antidotes � � � These are substances which act either by direct chemical action or by oxidizing the poison to from a non-toxic or an insoluble compound. Some of the examples are as follows: Dilute acetic acid or vinegar neutralize alkalis by direct chemical action. Canned fruit is an useful alternative for the purpose. Dilute alkalis, eg, milk of magnesia, will neutralize acids, however, bicarbonates should not be used owing to the risk of rapture of stomach from the librated carbon dioxide, tannin (strong tea) produce insoluble compounds with most alkaloids, glucosides and metals

� � Potassium permanganate in an oxidizing agent effective against all oxidisable poison, such as most of alkaloids, amidopyrin antipyrine, barbiturates, phosphorus, cyanides, etc. The usual strength of the solution is 1: 100 approximately ( 1 gm in litre ) and even 100 -150 ml of this solution ca be left in the stomach with out ay harm. A weak solution of iodine , 15 drops of the tincture to a tumblerful (200 ml) of water, may be helpful in the absence of permanganate.

Physiological or Pharmacological Antidotes � These agents produce effects which are opposite to that of the poison. Example of the physiological antagonists are atropine for pilocaroine, diazepam for strtchnine, naloxone for morphine, atropine and oximes for organophosphorus compounds, Nacetyle cysteine for acetaminophen and mazicon for benzodiazepins. However, the antagonism is usual nor complete and the remedy , mat itself produce most undesirable side affects.

� Certain chelating agents are widely used as specific antidotes against some heavy metals. These are substances which produce a firm non ionized cyclic complex (chelate) with cations. Such compounds can form stable, soluble, non toxic complexes with calcium and certain heavy metals. The important amongst them are BAL, EDTA (ethylene diamine tetra acetate) and versenate (calcium diethyl tetra acetate) for Arsenic, EDTA and versenate for mercury and lead, N- penicillamine for mercury, lead and copper, and desferrioxamine-B for iron.

� BAL (brithish anti-lewisite), also called dimercaprol is used in the treatment of certain types of heavy metal poisoning. It is given deep intra muscularly as a 10% solution in arachis oil with benzyl benzoate. In severe poisoning a dose of 3 mg/kg is given at 4 hourly interval for the first 2 days, and 6 hourly interval during the 3 rd day, and at 12 hourly interval thereafter for about 10 days. BAL is contraindicated if the liver extensively demerge.

� EDTA is chelating agent and is affective in lead, mercury, and copper poisoning. The usual adult dose is 1 gram twice daily for periods up to 5 days given by slow intravenous infusion in isotonic glucose saline. After an interval of 2 days this course of treatment may be repeated, EDTA has been shown to be superior to BAL in some respects for the treatment of poisoning by arsenic and mercury. It is contraindicated in renal damage.

� Penicillamine ( cuprimine) is a degradation product of penicillin and has the advantage that it can be given orally, continually, and that it is much less toxic then EDTA. It is the treatment of choice in copper, lead and mercury poisoning. A dose of 30 mg/kg body weight up to a total of 2 gms per day in 4 divided doses given orally is satisfactory. It is especially useful in hepatolenticular degeneration( Wilsons disease) which is caused by a disorder of copper metabolism.

� Desferrioxamine chelates iron. It is cheifly valuable in the treatment of acute iron poisoning. In certain chronic diseases such as haemochromatosis characterized by excessive retention of iron in the tissues, desferrioxamine is useful in accelerating the removal of iron from the body.

Universal Antidote. � It is an antidote that is used in those cases where the nature of ingested poison is unknown or where it is suspected that a combination of 2 or more poisons has been taken. It cosists of a mixture of readily available substances, as follows.
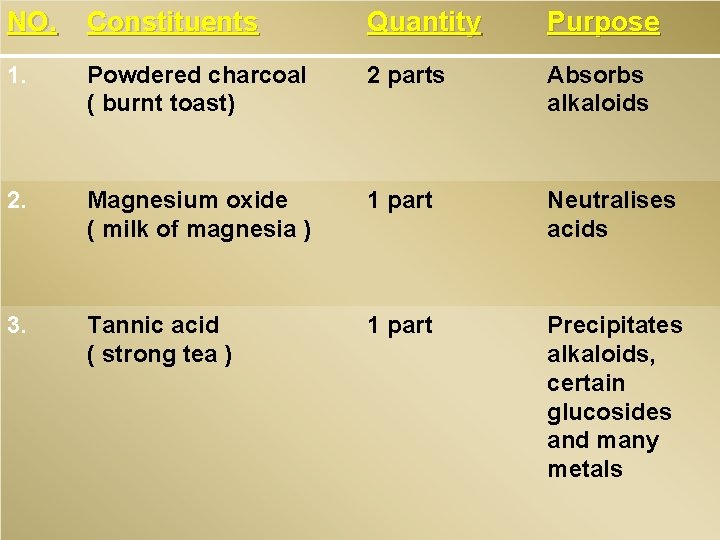
NO. Constituents Quantity Purpose 1. Powdered charcoal ( burnt toast) 2 parts Absorbs alkaloids 2. Magnesium oxide ( milk of magnesia ) 1 part Neutralises acids 3. Tannic acid ( strong tea ) 1 part Precipitates alkaloids, certain glucosides and many metals

� The mixture is given in a dose of a tablespoonful stirred up in a 200 ml of water, and may be repeated once or twice. Even when given soon after the ingestion of poison, it is not very effective.

- Slides: 17