Presentation about Good Pharmacy Practice GPP The current

Presentation about: «Good Pharmacy Practice (GPP). The current state and organization of pharmaceutical provision of the population of Ukraine» .

Good Pharmaceutical Practice In Tokyo September 5, 1993 the International Pharmaceutical Federation (FIP) adopted international guidelines for Good Pharmacy Practice. This document is intended to encourage national pharmaceutical organisations to focus the attention of pharmacists in the community and hospital pharmacy sector on developing the elements of the service they provide to meet changing circumstances. Conscious of the need to help developing countries achieve GPP, the FIP Community Pharmacy Section Executive Committee established a working group to produce guidelines in this area in 1992. The paper, entitled “GPP in Developing Countries – Guidelines for Implementation”, was endorsed by the FIP CPS Executive Committee in September 1998.

These guidelines were developed as a reference to be used by national pharmaceutical organizations, governments, and international pharmaceutical organizations to set up nationally accepted standards of Good Pharmacy Practice. The GPP Guidelines are based on the pharmaceutical care given by pharmacists. The guidelines recommend for national standards to be set: 1. the promotion of health; 2. the supply of medicines, medical devices, patient self-care; 3. improving prescribing and medicine use by pharmacists’ activities.

These guidelines have been subsequently adapted and adopted in a wide number of developed countries. In certain cases, the national professional body has strived to adapt the guidelines and developed, in collaboration with the government, specific regulation/legislation on this matter. Having realized the importance of continuing to increase awareness of GPP and stimulating its implementation, the FIP Bureau decided to request the BPP to focus on theme and to develop a specific activity. They are the ones most frequently exposed to such products which may be inefficacious or toxic products, and which threaten to erode confidence in the health care system. It was for this reason that in May 1994 the Forty-seventh World Health Assembly, in adopting resolution WHA 47. 12 on the role of the pharmacist in support of the WHO revised drug strategy, drew attention to pharmacists' responsibilities in assuring the quality of the products they dispense.

The mission of pharmacy practice is to provide medications and other health care products and services and to help people and society to make the best use of them. Comprehensive pharmacy service involves activities both to secure good health and to avoid ill-health in the population. When ill-health is treated, it is necessary to assure quality in the process of using medicines in order to achieve maximum therapeutic benefit and avoid untoward sideeffects. This presupposes the acceptance by pharmacists of shared responsibility with other professionals and with patients for the outcome of therapy. In recent years the term “pharmaceutical care” has established itself as a philosophy of practice, with the patient and the community as the primary beneficiaries of the pharmacist’s actions. The concept is particularly relevant to special groups such as the elderly, mothers and children, and chronically ill patients, as well as to the community as a whole in terms of, for example, cost containment. While the basic concepts of pharmaceutical care and good pharmacy practice are largely identical, it can be said that good pharmacy practice is the way to implement pharmaceutical care.

GPP is the practice of pharmacy that responds to the needs of the people who use the pharmacists’ services to provide optimal, evidence-based care. To support this practice it is essential that there be an established national framework of quality standards and guidelines. Good pharmacy practice requires that a pharmacist's first concern in all settings is the welfare of patients. Good pharmacy practice requires that the core of the pharmacy activity is the supply of medication and other health care products of assured quality, appropriate information and advice for the patient, and monitoring of the effects of use. Good pharmacy practice requires that an integral part of the pharmacist's contribution is the promotion of rational and economic prescribing and of appropriate use of medicines. Good Pharmacy Practice requires that the objective of each element of pharmacy service is relevant to the patient, is clearly defined and is effectively communicated to all those involved.

In satisfying these requirements, the following conditions are necessary: Professionalism should be the main philosophy underlying practice, although it is accepted that economic factors are also important. Pharmacists should have input into decisions about the use of medicines. A system should exist that enables pharmacists to report adverse events, medication errors, defects in product quality or detection of counterfeit products. This reporting may include information about drug use supplied by patients or health professionals, either directly or through pharmacists. The ongoing relationship with other health professionals, particularly physicians, should be seen as a therapeutic partnership that involves mutual trust and confidence in all matters relating to pharmacotherapeutics. The relationship between pharmacists should be as colleagues seeking to improve pharmacy service, rather than as competitors. In reality, organizations, group practices and pharmacy managers should accept a share of responsibility for the definition, evaluation and improvement of quality

The pharmacist should be aware of essential medical and pharmaceutical information about each patient. Obtaining such information is made easier if the patient chooses to use only one pharmacy or if the patient's medication profile is available. The pharmacist needs independent, comprehensive, objective and current information about therapeutics and medicines in use. Pharmacists in each practice setting should accept personal responsibility for maintaining and assessing their own competence throughout their professional working lives. Educational programs for entry to the profession should appropriately address both current and foreseeable future changes in pharmacy practice. National standards of good pharmacy practice should be specified and should be adhered to by practitioners.

Good pharmacy practice involves four main groups of activities, namely: �activities associated with the promotion of good health, the avoidance of illhealth and the achievement of health objectives; �activities associated with the supply and use of medicines and of items for the administration of medicines or for other aspects of treatment (these activities may be undertaken in the pharmacy, in an institution or in a homecare setting); �activities associated with self-care, including advice about and, where appropriate, the supply of a medicine or other treatment for symptoms of ailments that lend themselves to self-treatment; �activities associated with influencing the prescribing and use of medicines.

In addition to these groups of activities good pharmacy practice also encompasses: �establishment of arrangements with other health professional communities for health promotion activities at population level, including minimization of the abuse and misuse of medicines; �professional assessment of promotional materials for medicines and other products associated with health care; �dissemination of evaluated information about medicines and various aspects of health care; �involvement in all stages of clinical trials.

For each of the four main elements of good pharmacy practice, national standards should be established in relation to processes and facilities (table 4. 1).

For each of the four main elements of good pharmacy practice, national standards should be established in relation to processes and facilities (table 4. 1).

These standards should be promoted among members of the profession. Pharmacists have a professional responsibility to document practice experience and activities and to conduct and/or participate in pharmacy practice research and therapy research. Specific standards of good pharmacy practice can be developed only within the framework of a national organization. These guidelines are recommended as a set of professional goals in the interest of the patients or customers in the pharmacy. Responsibility for moving the project forward will rest with each national pharmaceutical organization. Achieving specific standards of good pharmacy practice for each nation within these guidelines may require considerable time and effort. As health professionals, pharmacists have a duty to begin the process without delay.
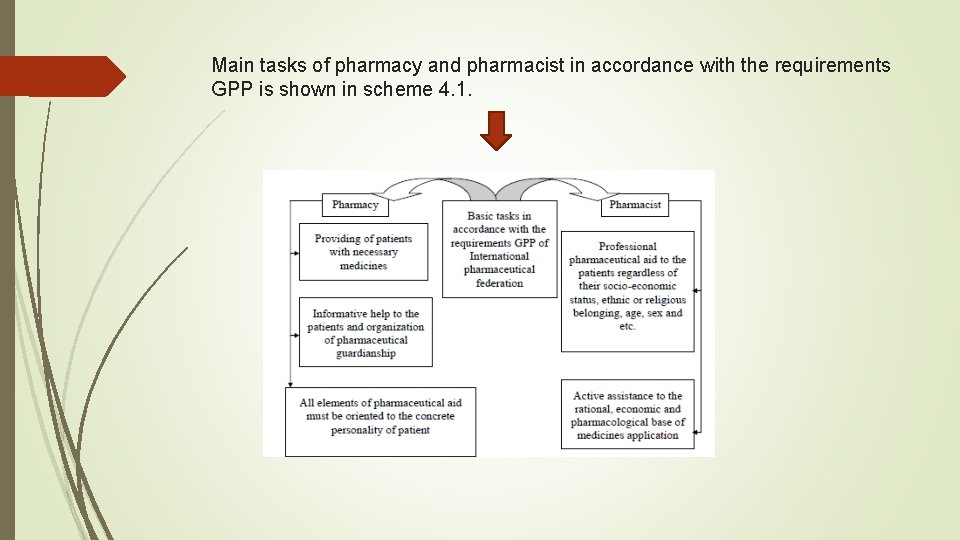
Main tasks of pharmacy and pharmacist in accordance with the requirements GPP is shown in scheme 4. 1.

Cheme 4. 1. Main tasks of pharmacy and pharmacist in accordance with the requirements GPP Thus, all international standarts describes a set of principles and procedures that when followed helps ensure that therapeutic goods and services are of high quality. References : 1. Standarts for quality of pharmacy services http: //fip. org/files/fip/Statements/latest/Dossier%20004%20 total. PDF 2. Good pharmacy practice in community and hospital pharmacy settings http: //apps. who. int/medicinedocs/documents/s 21088 en. pdf 3. Guideline for Good Clinical Practice http: //www. ich. org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Effica cy/E 6_R 1_Guideline. pdf Active assistance to the rational, economic and pharmacological base of medicines application 4. Guide to good manufacturing practice for medicinal products https: //www. tga. gov. au/pdf/manuf-pics-gmpmedicines-part 1. pdf 5. Guidelines on Good Distribution Practice of Medicinal Products for Human Use (94/C 63/03) http: //ec. europa. eu/health/files/eudralex/vol-4/gdpguidelines 1. pdf 6. Good Distribution Practice http: //ec. europa. eu/health/human- use/good_distribution_practice/index_en. htm

questions Review questions What International standards that regulate pharmaceutical activity do you know? Give a short definition (GLP, GCP, GMP, GDP, GPP). What are the definition, targets and elements of the Good pharmacy practice (GPP)? What are four basic directions of the guidance GPP?
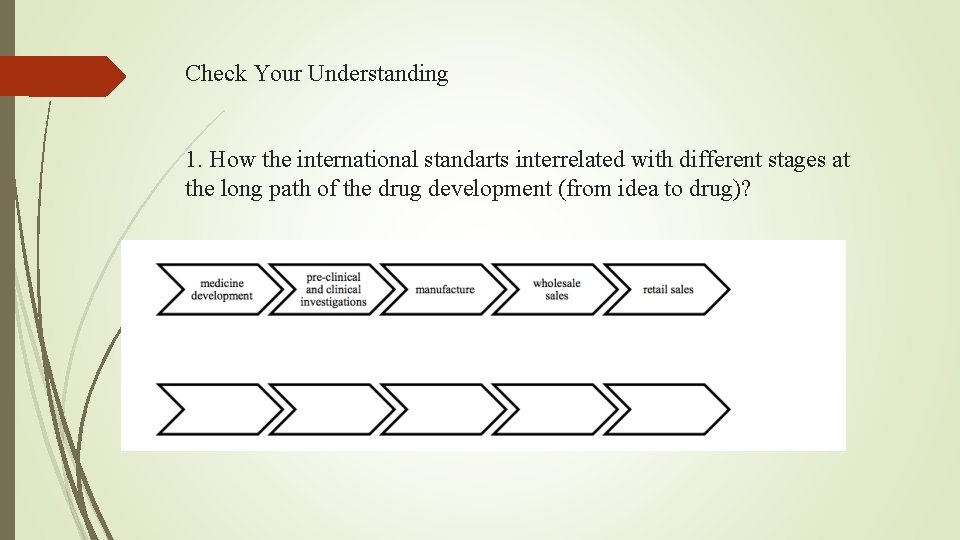
Check Your Understanding 1. How the international standarts interrelated with different stages at the long path of the drug development (from idea to drug)?

2. The guidelines recommend for national standards to be set: A the promotion of health; B advertising and marketing of the drugs; C the supply of medicines, medical devices, patient self-care; D improving prescribing and medicine use by pharmacists’ activities; E providing of the medical insurance and reimbursement for the population.

3. Align the terms of their definition:

Ukraine is gradually approaching international pharmaceutical practice standards. 1. Ukraine is gradually approaching international pharmaceutical practice standards. 2. The purpose of pharmaceutical practice is to provide the high quality pharmaceutical services that pharmaceutical workers provide to the population. 3. Scope - organizations, legal entities and individuals who carry out retail and wholesale realization in the field of drug circulation. 4. The main direction is the release of proper medical products and the provision of pharmaceutical services. 5. One of the main requirements of proper pharmacy practice is the presence of qualified staff who knows the basics of pharmacology, pharmacotherapy and deontology.

6. The Higher School of Pharmaceutical Training is the beginning of proper pharmacy practice. 7. The role of faculty members in the technology of medicine in the teaching lecture course and practical training in the discipline "Good Pharmacy and Production Practice (GPP, GMP) is important. " 8. Availability of teaching aids on the course of GPP - a guarantee of the training of a modern pharmaceutical specialist. 9. A well-defined didactic purpose and practical motivation will allow students to understand the basic requirements for all aspects of pharmaceutical preparation of dosage forms. 10. The GPP situational challenges offered by students enable them to grasp the practical skills that are essential for securing the knowledge gained during lectures. 11. Ukraine will attain the European level in meeting the requirements of proper pharmacy practice when a modern pharmaceutical specialist fully understands the knowledge provided at a high school and can realize this knowledge and skills in professional activities.

Thank you for attention!
- Slides: 22