Present in Atropa balladon Datura strammonium Chemistry Molecular
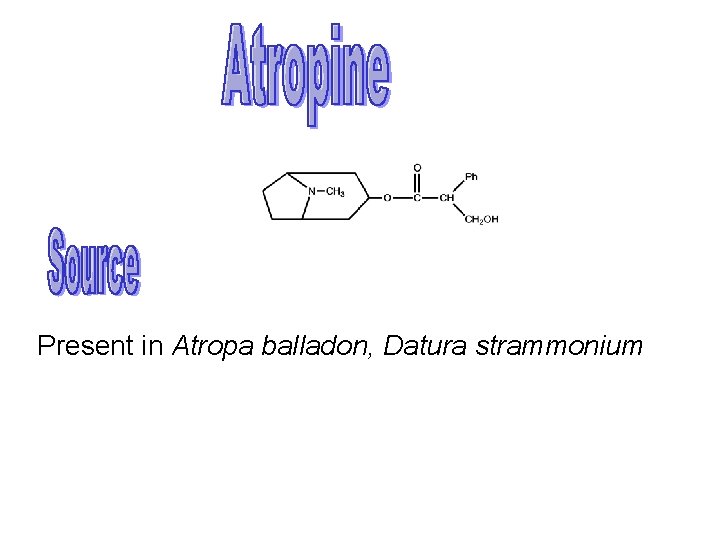
Present in Atropa balladon, Datura strammonium

Chemistry • • Molecular formula C 17 H 23 NO 3 M. Point = 118 °C Recemic mixture of d & l hyoscyamine Atropine is more stable than either of d & l isomer • Organic ester formed by aromatic acid and organic base tropine
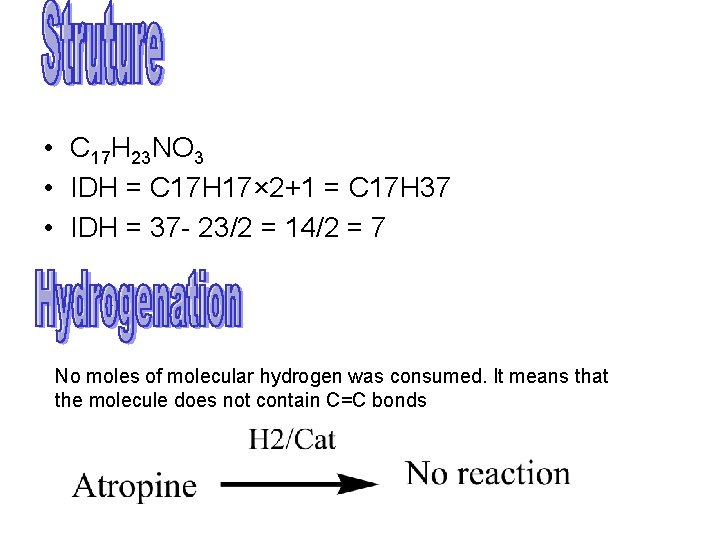
• C 17 H 23 NO 3 • IDH = C 17 H 17× 2+1 = C 17 H 37 • IDH = 37 - 23/2 = 14/2 = 7 No moles of molecular hydrogen was consumed. It means that the molecule does not contain C=C bonds
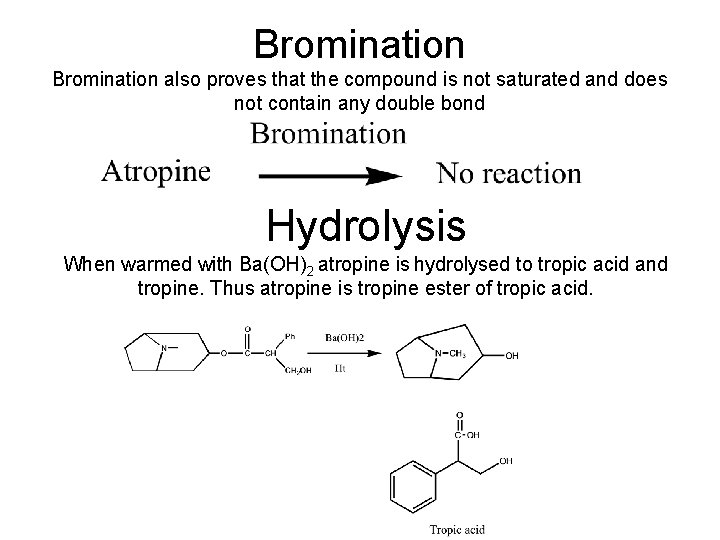
Bromination also proves that the compound is not saturated and does not contain any double bond Hydrolysis When warmed with Ba(OH)2 atropine is hydrolysed to tropic acid and tropine. Thus atropine is tropine ester of tropic acid.
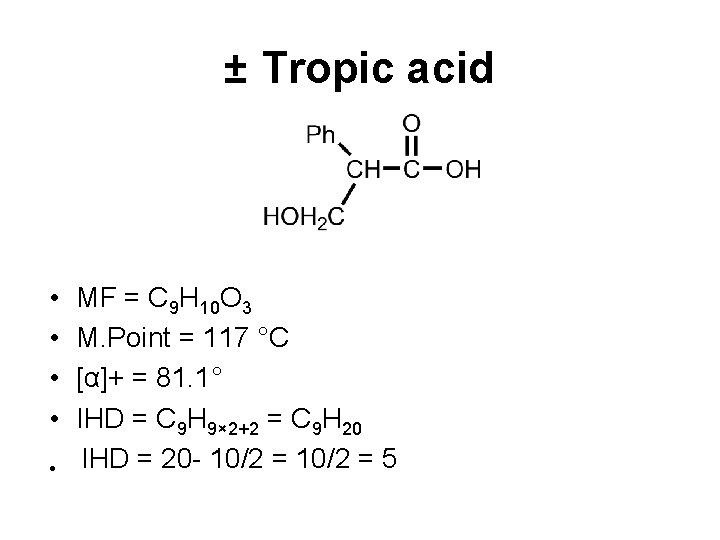
± Tropic acid • • • MF = C 9 H 10 O 3 M. Point = 117 °C [α]+ = 81. 1° IHD = C 9 H 9× 2+2 = C 9 H 20 IHD = 20 - 10/2 = 5
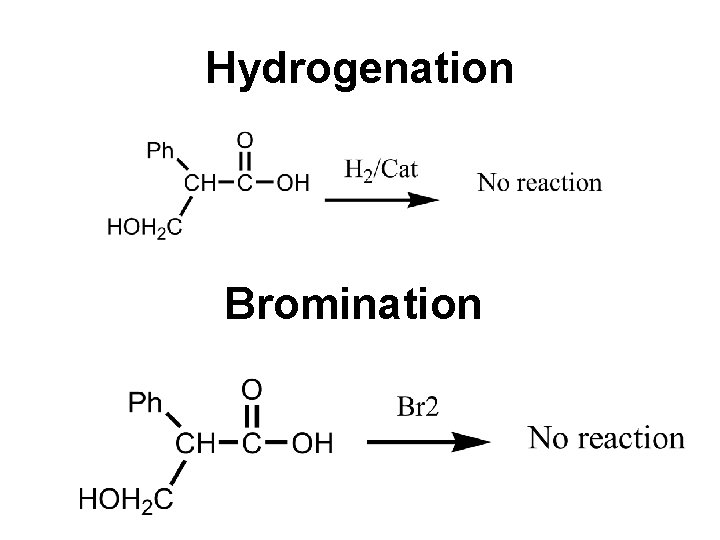
Hydrogenation Bromination
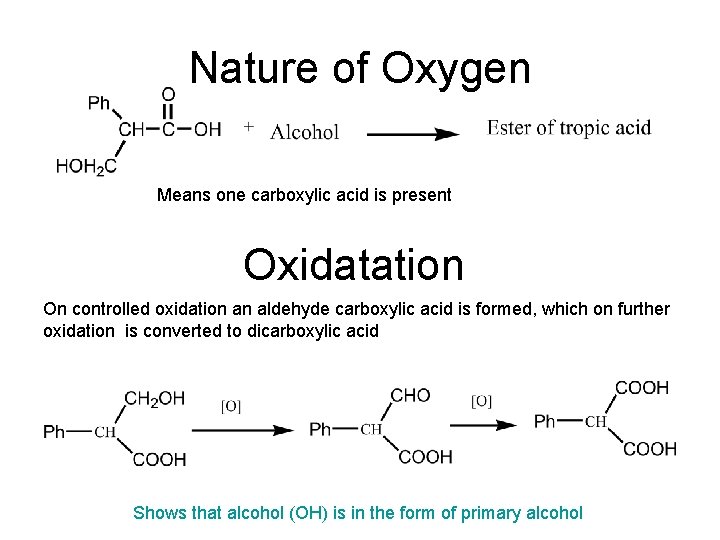
Nature of Oxygen Means one carboxylic acid is present Oxidatation On controlled oxidation an aldehyde carboxylic acid is formed, which on further oxidation is converted to dicarboxylic acid Shows that alcohol (OH) is in the form of primary alcohol

Decarboxylation • Strongly heated CO 2 evolved • Confirm the presence of COOH group • These two oxygen atoms are present in the form of carboxylic acid
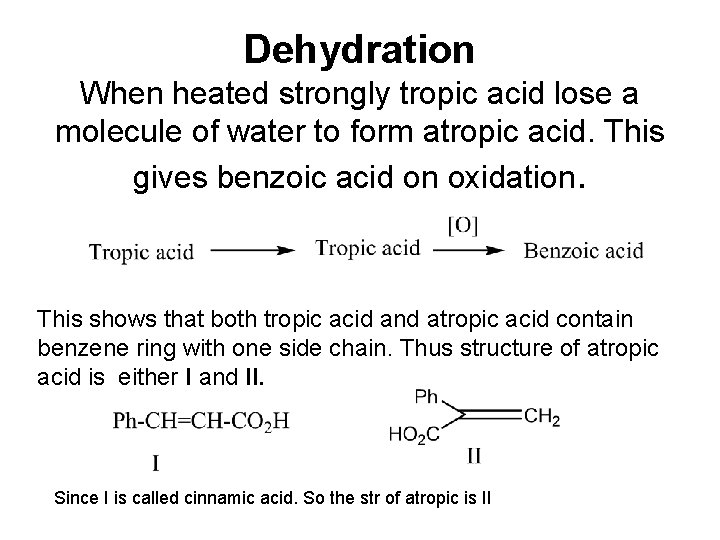
Dehydration When heated strongly tropic acid lose a molecule of water to form atropic acid. This gives benzoic acid on oxidation. This shows that both tropic acid and atropic acid contain benzene ring with one side chain. Thus structure of atropic acid is either I and II. Since I is called cinnamic acid. So the str of atropic is II
- Slides: 9